Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
GE-Portuguese Journal of Gastroenterology
versão impressa ISSN 2341-4545
GE Port J Gastroenterol vol.26 no.5 Lisboa out. 2019
https://doi.org/10.1159/000495523
CLINICAL CASE STUDY
Neuroendocrine Carcinoma and Intracystic Papillary Neoplasm: A Rare Association in the Gallbladder
Carcinoma neuroendócrino e neoplasia papilar intraquística: uma associação incomum na vesícula biliar
João Fragaa, Rui Caetano Oliveiraa, Henrique Alexandrinob,c, Maria Augusta Ciprianoa
aServiço de Anatomia Patológica, Centro Hospitalar e Universitário de Coimbra, Coimbra, Portugal; bServiço de Cirurgia, Centro Hospitalar e Universitário de Coimbra, Coimbra, Portugal; cFaculdade de Medicina, Universidade de Coimbra, Coimbra, Portugal
* Corresponding author.
ABSTRACT
Neuroendocrine carcinoma of the gallbladder is a very rare neoplasia comprising only 0.2% of all the gastrointestinal neuroendocrine tumors. We present the case of an 85-yearold male with nonspecific gastrointestinal symptoms, progressive weight loss, and deterioration in the preceding 15 days. Imaging study showed a 5-cm polypoid mass localized at the gallbladder fundus, leading to a radical cholecystectomy. Pathology revealed the known lobulated polypoid lesion, on cut section, constituted by friable tissue without evident infiltration of the gallbladder wall and a contiguous friable, brown, flat lesion. Histological evaluation displayed a small cell neuroendocrine carcinoma, consisting of intermediate and small cells, rounded or spindle, with extensive areas of necrosis, with positivity for neuroendocrine immunomarkers, associated with intracystic papillary neoplasm with high and low dysplasia. This is an uncommon association with few cases presented in the literature.
Keywords: Neuroendrocine carcinoma, Gallbladder, Intracystic papillary neoplasm
RESUMO
Carcinoma neuroendócrino da vesícula biliar é uma neoplasia muito rara, compreendendo cerca de 0.2% de todas as neoplasias neuroendócrinas do trato gastrointestinal. Apresentamos um caso de um homem de 85 anos, com sintomas gastrointestinais inespecíficos, perda de peso progressiva e deterioração do estado geral com 15 dias de evolução. Estudos de imagem demonstraram lesão polipóide com 5 cm localizada no fundo da vesícula biliar, motivando colecistectomia. Macroscopicamente observou-se lesão polipoide composta por tecido acastanhado e friável. Estudo histológico demonstrou lesão constituída por pequenas células, com extensas áreas de necrose e positivas para marcadores neuroendócrinos, associada a neoplasia papilar intraquística com displasia de baixo e alto grau. Esta associação é incomum, com apenas raros casos descritos na literatura.
Palavras-Chave: Carcinoma neuroendocrine, Vesícula biliar, Neoplasia papilar intraquística
Introduction
Neuroendocrine carcinoma (NEC) of the gallbladder is an extremely rare neoplasia, with a poor outcome [1, 2]. Symptoms are mostly unspecific, and the disease can present as an acute/chronic cholecystitis or even as a gallbladder polyp, and radiology does not possess enough accuracy to distinguish between NEC and a conventional adenocarcinoma [3]. Its origins are not clear and association with preneoplasic lesions are more uncommon [4, 5].
Due to its high mortality, the early identification of the neoplasm, on a preoperatory instance or after surgery with a correct pathologic diagnosis, is crucial for patient referral to proper treatment [3]. We present a rare case of a NEC of the gallbladder associated with intracystic papillary neoplasm (ICPN) with high and low dysplasia and explore he differential diagnosis.
Case Report
We present an 85-year-old male with a history of type 2 diabetes mellitus, arterial hypertension, dyslipidemia, and dysrhythmia, with complaints of asthenia, anorexia, and progressive weight loss – 15 kg in 1 month, corresponding to more than 10% of the total body weight – most intensively in the last 15 days. The patient also presented with intermittent epigastric pain and nausea, unrelated to meals. He denied vomiting, diarrhea, or gastrointestinal blood loss.
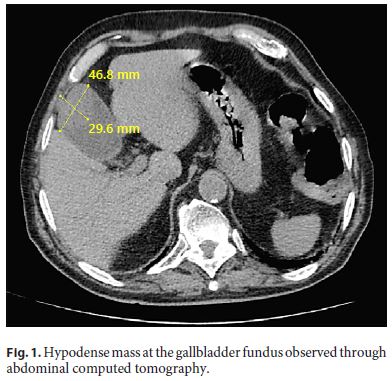
Physical examination revealed tenderness in the upper right quadrant of the abdomen, without guarding, palpable masses, or organomegaly. Blood tests showed a total bilirubin level of 2.10 mg/dL, a direct bilirubin level of 0.90 mg/dL, an aspartate aminotransferase level of 26 IU/L, an alanine aminotransferase level of 24 IU/L, and an alkaline phosphatase level of 82 IU/L. CA19-9 and CEA were normal and complete blood count was unremarkable. Upper endoscopy and total colonoscopy revealed antral gastritis, sigmoid diverticulosis, and a sessile polyp at 80 cm. Ultrasound study displayed moderate distension of the gallbladder, without evident parietal thickening, but with an echogenic heterogeneous content, partially vascularized in the Doppler study, suspicious for neoplasia. Chest and abdomen computed tomography displayed a hypodense mass at the gallbladder fundus with 46.8 × 29.6 mm (Fig. 1), without any signs of metastatic spread. The patient was then proposed for cholecystectomy.
Surgery consisted of exploratory laparotomy, radical cholecystectomy (en bloc with a rim of liver parenchyma), and hepatic pedicle lymph node dissection. During surgery, frozen section evaluation of the surgical margin of the cystic duct was negative for neoplasia. Gross examination presented a gallbladder with 10 × 6.5 × 4 cm, with a 5-cm lobulated, greenish polypoid lesion located at the fundus (Fig. 2a). On cut section, it consisted of friable tissue, with firm and whitish areas, without evident infiltration of the wall; there was another flat, slightly polypoid 2-cm lesion, brown and friable, also apparently noninvasive (Fig. 2b).

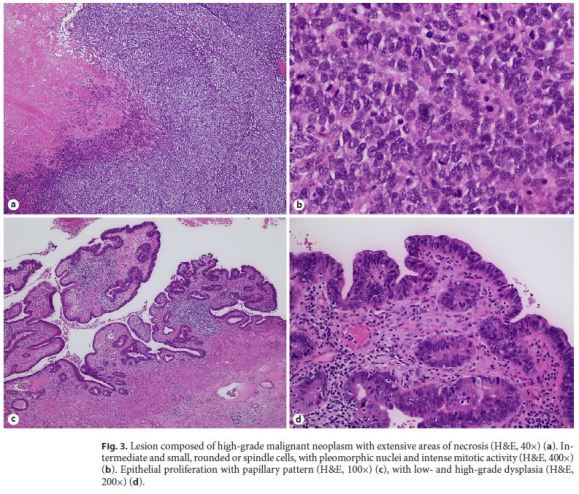
Histological examination was performed on hematoxylin and eosin (H&E)-stained slides observed in light microscope (Nikon Eclipse 50i) and images obtained using a Nikon-Digital Sight DSFi1 camera. On histological study, the largest polypoid lesion was composed of high-grade malignant neoplasm with extensive areas of necrosis, consisting of intermediate and small cells, rounded or spindle (Fig. 3a). The tumor had a predominantly polypoid growth pattern; however, there was infiltration of the muscularis propria layer. The tumor surface was extensively ulcerated, partly covered by the columnar epithelium of the mucosa. The cells had pleomorphic nuclei and intense mitotic activity – 84 mitoses/10 high-power fields (3 mm2) (Fig. 3b). Vascular invasion was not present. The adjacent flat lesion presented areas of epithelial proliferation with papillary pattern (Fig. 3c), with low- and high-grade dysplasia (Fig. 3d). Conjugation of morphological characteristics and location of the lesion provided the following differential diagnosis: poorly differentiated carcinoma/NEC primary or secondary (most likely from lung), lymphoma, and melanoma.
Immunohistochemistry studies were performed on one representative block of the lesion, performed on Ventana Marker Platform Bench Mark ULTRA IHC/ISH, with an indirect multimeric detection system, biotin free, peroxidase conjugated, and showed diffuse positivity for synaptophysin (MRQ-40, Cell Marque, CA, USA), heterogeneous for neuron-specific enolase (NSE) (MRQ55, Cell Marque, CA, USA) and focal for chromogranin A (LK2H10, Ventana, AZ, USA), CK 8/18 (B22.1/B23.1, Ventana, AZ, USA), and CD99 (O13, Ventana, AZ, USA), and faintly diffuse positivity for vimentin (V9, Ventana, AZ, USA). P53 (DO-7, Ventana, AZ, USA) was diffusely positive (Fig. 4). Staining with Cam5.2 (Cam5.2, Ventana, AZ, USA) showed focal positivity; staining for CKAE1/3 (PCK26, Ventana, AZ, USA), LCA (2B11&PD7/26, Ventana, AZ, USA), epithelial membrane antigen (EMA, E29, Ventana, AZ, USA), α-smooth-actin (1A4, Ventana, AZ, USA), melanosome (HMB45, Ventana, AZ, USA), CK7 (SP52, Ventana, AZ, USA), and thyroid transcription factor (TTF1, SP141, Ventana, AZ, USA) was negative.
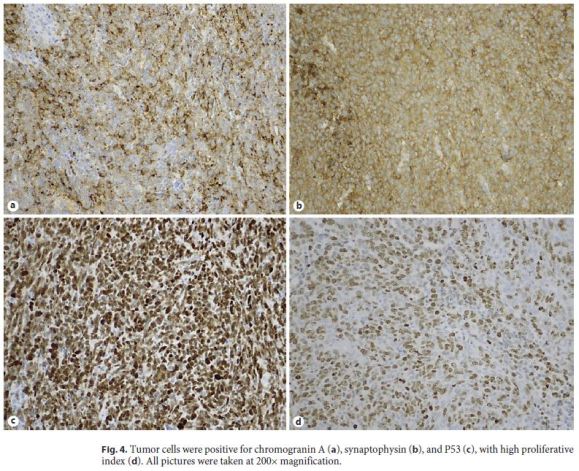
The Ki67 (30-9, Ventana, AZ, USA) proliferative index evaluation was also studied, with a high result (80%). Absence of staining for TTF1 argued against secondary disease of lung lesion, which was also supported by the ICPN of the gallbladder, usually associated with invasive tumors [6]. The ICPN revealed diffuse positivity for MUC1 (H23, Ventana, AZ, USA) and focal positivity for MUC5AC (MRQ-19, Cell Marque, CA, USA) and MUC6 (CLH5, Leica, Germany), with absence of expression for MUC2 (MRQ-18, Ventana, AZ, USA), thus being classified as ICPN of pancreatobiliary type.
The absence of expression for LCA excluded lymphoma, and no staining for HMB45 was against melanoma. The scarce positivity for epithelial markers (Cam5.2 and CK 8/18) cannot exclude poorly differentiated carcinoma, but the expression of neuroendocrine markers together with the morphology was in favor of neuroendocrine tumor (NET), and according to the histological morphology, high Ki67 proliferation rate, and high mitotic index, it was classified as a small cell NEC, G3.
Surgery was the treatment of choice. The patient was discharged after 6 days. He did not perform chemotherapy due to frailty. The patient died after 3 months due to disease progression. According to the familys wishes, no autopsy was performed.
Discussion
Embryologically, the gallbladder originates in the endoderm, but the origin of neuroendocrine cells is controversial. In the ventral wall of the early primitive midgut, the hepatic diverticulum, by the fourth week of development, originates two buds: the cranial area forms the liver and extrahepatic bile ducts and the caudal area gives rise to the ventral pancreas inferiorly and the gallbladder and cystic duct superiorly [7].
Neuroendocrine cells have a controversial derivation. These were initially identified by Kultschitsky in 1897 [4], in the colic mucosa. He proposed a different polarity of these cells in relation to the other intestinal secretory ones and suggested that its secretion would be released into the bloodstream. Pearse [8] stated that neuroendocrine cells would have an origin in neural crest migrant cells, and Fontaine and Le Douarin [9] confirmed the same structure in gastrointestinal ganglion cells, paraganglionic, melanocyte, and thyroid C-cell. Cheng and Leblond [10] are of the opinion that intestinal endocrine cells have the same endodermal origin as the remaining gastrointestinal mucosa. Neuroendocrine cells have been identified in the gallbladder, most commonly in association with foci of intestinal metaplasia [1]. Progenitor cells play an important role during embryogenesis and also in tissue maintenance, which support the hypothesis that stem/ progenitor cells may constitute a precursor to tumors with a mixed phenotype [5, 11, 12].
Most NEC are found in gastrointestinal (66%) and respiratory (31%) tracts. In the first one, most of them are localized in the rectum, jejuno-ileum, and pancreas; NEC of the gallbladder is very rare, comprising 0.2% of all gastrointestinal NET, with the first small cell carcinoma described in 1981 by Albores-Saavedra et al. [2]. Since then, some cases have been described, with less than 100 cases in the literature [3, 13, 14]. It is not possible to differentiate preoperatively between gallbladder adenocarcinoma and NEC with imaging techniques [10]. The diagnosis of gallbladder NEC is rarely made preoperatively because this entity generally presents with nonspecific symptoms [3].
The current WHO classification divides neuroendocrine neoplasms of the gallbladder into the categories of NET (G1, G2), small cell NEC and large cell NEC (G3), mixed neuroendocrine-non-neuroendocrine neoplasms, goblet cell carcinoid, and tubular carcinoid. Primary NETs of the gallbladder are more frequent in females (68%) and the age at presentation ranges from 25 to 85 years, peaking in ages 75–79 years. Differential diagnosis between NET and NEC is performed with mitotic count in 10 high-power fields and with Ki67 proliferative index; in some cases, the P53 may be added to support NEC. NEC can be subdivided into small cell and large cell carcinomas, based on morphology, and in some cases, NEC are combined with other histological components, including adeno-, adenosquamous, and mucinous carcinoma, and are classified as MANEC according to WHO 2010 [15].
Adsay et al. [16] reported ICPN in 14 out of 3,265 cholecystectomies (0.4%). This frequency might be higher among invasive carcinomas, and 39 out of 606 (6.4%) carcinoma cases had ICPN component. Conversely, invasive carcinoma was seen in 68 out of 123 (55%) ICPN cases [16]. Papillary neoplasms arising from the biliary system histologically resemble intraductal papillary mucinous neoplasms (IPMNs) of the pancreas. The new WHO classification, revised in 2010, uses the term intracystic (gallbladder) or intraductal (bile duct) papillary neoplasms to classify such lesions and to clarify the terminology [2, 13]. Similar to IPMNs, ICPNs often show greater architectural complexity and more cytological atypia and mitotic figures than adenomas in the gallbladder. Similar to IPMNs, some of them develop into invasive cancer, and high MUC-1 expression has been reported to be associated with carcinogenesis to tubular adenocarcinoma and poorer survival [5, 17, 18].
The prognosis of NEC of the gallbladder is very poor, with a median overall survival of 8 months. Aggressive surgical resection in combination with chemotherapy could improve the survival outcome if the tumor is resectable [1, 2, 19].
In short, we present a case of a primary NEC of the gallbladder, an extremely rare neoplasia, associated with ICPN, with a vast differential diagnosis – poorly differentiated carcinoma, lymphoma, and melanoma, which has a poor prognosis.
References
1 Eltawil KM, Gustafsson BI, Kidd M, Modlin IM. Neuroendocrine tumors of the gallbladder: an evaluation and reassessment of management strategy. J Clin Gastroenterol. 2010 Nov-Dec;44(10):687–95. [ Links ]
2 Albores-Saavedra J, Cruz-Ortiz H, AlcantaraVazques A, Henson DE. Unusual types of gallbladder carcinoma. A report of 16 cases. Arch Pathol Lab Med. 1981 Jun;105(6):287–93. [ Links ]
3 Chen C, Wang L, Liu X, Zhang G, Zhao Y, Geng Z. Gallbladder neuroendocrine carcinoma: report of 10 cases and comparision of clinicopathologic features with gallbladder adenocarcinoma. Int J Clin Exp Pathol. 2015 Jul;8(7):8218–26. [ Links ]
4 Rosai J. The origin of neuroendocrine tumors and the neural crest saga. Mod Pathol. 2011 Apr; 24(S2 Suppl 2):S53–7. [ Links ]
5 Meguro Y, Fukushima N, Koizumi M, Kasahara N, Hydo M, Morishima K, et al. A case of mixed adenoneuroendocrine carcinoma of the gallbladder arising from an intracystic papillary neoplasm associated with pancreaticobiliary maljunction. Pathol Int. 2014 Sep;64(9):465–71. [ Links ]
6 Hoang MP, Murakata LA, Katabi N, Henson DE, Albores-Saavedra J. Invasive papillary carcinomas of the extrahepatic bile ducts: a clinicopathologic and immunohistochemical study of 13 cases. Mod Pathol. 2002 Dec;15(12):1251–8. [ Links ]
7 Ando H. Embryology of the biliary tract. Dig Surg. 2010;27(2):87–9. [ Links ]
8 Pearse AG. The cytochemistry and ultrastructure of polypeptide hormone-producing cells of the APUD series and the embryologic, physiologic and pathologic implications of the concept. J Histochem Cytochem. 1969 May;17(5):303–13. [ Links ]
9 Fontaine J, Le Douarin NM. Analysis of endoderm formation in the avian blastoderm by the use of quail-chick chimaeras. The problem of the neurectodermal origin of the cells of the APUD series. J Embryol Exp Morphol. 1977 Oct;41:209–22. [ Links ]
10 Cheng H, Leblond CP. Origin, differentiation and renewal of the four main epithelial cell types in the mouse small intestine. V. Unitarian Theory of the origin of the four epithelial cell types. Am J Anat. 1974 Dec;141(4):537–61. [ Links ]
11 Oshiro H, Matsuo K, Mawatari H, Inayama Y, Yamanaka S, Nagahama K, et al. Mucin-producing gallbladder adenocarcinoma with focal small cell and large cell neuroendocrine differentiation associated with pancreaticobiliary maljunction. Pathol Int. 2008 Dec;58(12):780–6. [ Links ]
12 Vadlamani I, Brunt EM. Hepatocellular progenitor cell tumor of the gallbladder: a case report and review of the literature. Mod Pathol. 2005 Jun;18(6):864–70. [ Links ]
13 Lee H, Choi HJ, Park IY. Small cell carcinoma of the gallbladder: a case report. Ann Hepatobiliary Pancreat Surg. 2017 Aug;21(3):168–71. [ Links ]
14 Carrera C, Kunk P, Rahma O. Small Cell Carcinoma of the Gallbladder: Case Report and Comprehensive Analysis of Published Cases. J Oncol. 2015;2015:304909. [ Links ]
15 Buscemi S, Orlando E, Damiano G, Portelli F, Palumbo VD, Valentino A, et al. Pure large cell neuroendocrine carcinoma of the gallbladder. Report of a case and review of the literature. Int J Surg. 2016 Apr;28 Suppl 1:S128–32. [ Links ]
16 Adsay V, Jang KT, Roa JC, Dursun N, Ohike N, Bagci P, et al. Intracholecystic papillarytubular neoplasms (ICPN) of the gallbladder (neoplastic polyps, adenomas, and papillary neoplasms that are ≥1.0 cm): clinicopathologic and immunohistochemical analysis of 123 cases. Am J Surg Pathol. 2012 Sep;36(9):1279–301. [ Links ]
17 Diagnostic criteria of pancreaticobiliary maljunction. J Hepatobiliary Pancreat Surg. 1994;1(3):219–21. [ Links ]
18 Barton JG, Barrett DA, Maricevich MA, Schnelldorfer T, Wood CM, Smyrk TC, et al. Intraductal papillary mucinous neoplasm of the biliary tract: a real disease? HPB (Oxford). 2009 Dec;11(8):684–91. [ Links ]
19 Adachi T, Haraguchi M, Irie J, Yoshimoto T, Uehara R, Ito S, et al. Gallbladder small cell carcinoma: a case report and literature review. Surg Case Rep. 2016 Dec;2(1):71. [ Links ]
Statement of Ethics
The authors have no ethical conflicts to disclose.
Disclosure Statement
The authors have no conflicts of interest to disclose.
* Corresponding author.
Rui Caetano Oliveira
Pathology Department CHUC
Praceta Mota Pinto
PT–3000-001 Coimbra (Portugal)
E-Mail ruipedrocoliveira@hotmail.com
Received: November 11, 2018; Accepted after revision: November 13, 2018














