Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
GE-Portuguese Journal of Gastroenterology
versão impressa ISSN 2341-4545
GE Port J Gastroenterol vol.24 no.3 Lisboa jun. 2017
https://doi.org/10.1159/000450899
CLINICAL CASE STUDY
Multisystemic Sarcoidosis with Early Gastrointestinal Symptoms
Sarcoidose Multissistémica com Sintomatologia Digestiva Inicial
Catarina Motaa, Carlos Ferreiraa, Maria Emília Oliveirab, João Meneses Santosa, Rui M.M. Victorinoa
aMedicine II Department and bPathology Department, Hospital de Santa Maria, Centro Hospitalar de Lisboa Norte, Lisbon, Portugal
* Corresponding author.
ABSTRACT
Sarcoidosis involving the digestive tract is very rare and requires a strict differential diagnosis with other systemic granulomatous diseases. We present a case of multisystemic granulomatosis involving the stomach and colon, causing initial digestive symptoms. Pulmonary nodules, pleural effusion, ascites, hepatomegaly, splenomegaly, and mesenteric as well as retroperitoneal and axillary lymphadenopathy were subsequently identified. The reticulo-endothelial expression, the hypercalcemia and, above all, a rapid and complete resolution after corticosteroids supported the hypothesis of sarcoidosis involving the digestive tract. A brief review of digestive tract involvement in sarcoidosis and of the distinctive features of sarcoidosis with gastrointestinal involvement and Crohns disease with a rare form of pleuropulmonary involvement is presented.
Keywords: Crohns disease; Gastrointestinal diseases;·Sarcoidosis/complications
RESUMO
A sarcoidose com envolvimento do tubo digestivo é rara e exige um rigoroso diagnóstico diferencial com outras doenças granulomatosas sistémicas. Os autores apresentam um caso de granulomatose multissistémica com envolvimento do estômago e cólon, motivando sintomatologia digestiva exuberante inaugural. Subsequentemente, foram identificados nódulos pulmonares, derrame pleural, ascite, hepatoesplenomegália e adenopatias mesentéricas, retroperitoneais e axilares. A expressão reticulo-endotelial, a hipercalcémia e, sobretudo, a rápida e total resolução do quadro após corticoterapia sustentaram a hipótese de sarcoidose com envolvimento do tubo digestivo. Procede-se a uma breve revisão do envolvimento do tubo digestivo na sarcoidose e dos critérios distintivos entre sarcoidose com envolvimento gastrointestinal e doença de Crohn associada a uma forma rara de envolvimento pulmonar.
Palavras-Chave: Doença de Crohn; Doenças gastrointestinais; Sarcoidose/complicações
Introduction
Sarcoidosis, described by Hutchinson in 1877, is a chronic multisystemic granulomatous disease characterized by epithelioid granulomas that can involve multiple organs and systems, with particular predilection for the lungs, the skin, the eye, and the reticulo-endothelial system. Pulmonary involvement occurs in 90% of cases and extrapulmonary involvement exceeds 30% [1, 2]. Symptomatic participation of the digestive tract is very rare and the stomach is the organ most commonly involved, although occasional cases of esophagus, small bowel, appendix, colon, and rectum involvement have also been described [3, 4]. The diagnosis of sarcoidosis relies on a compatible clinical picture, exclusion of clinical similar entities, and histopathological observation of granulomas without caseum. Two-thirds of patients have resolution of their disease within an interval of 2 years, with spontaneous resolution in many of these cases [3, 4] .
We present a case of multisystemic sarcoidosis characterized by initial digestive symptoms, raising the question of the differential diagnosis between sarcoidosis with gastrointestinal involvement and Crohns disease with pulmonary expression.
Clinical Case
A 44-year-old woman with no relevant clinical background was hospitalized with a 2-weak history of epigastric and periumbilical pain, vomiting, abdominal distension, asthenia, and anorexia.
Physical examination revealed pallor, with reasonable general condition, blood pressure 128/73 mm Hg, pulse rate 82/min, and respiratory rate 16/min. There were no rashes or skin lesions nor palpable lymphadenopathy. Cardiovascular and pulmonary examination was normal. The abdomen was distended, diffusely tender to deep palpation, with slight splenomegaly, without hepatomegaly or palpable masses. The remaining examination was unremarkable.
The blood tests revealed an iron deficiency anemia with haemoglobin of 7.4 g/dL and eosinophilia with a maximum value of 1,73 × 10 9 /L. C-reactive protein and sedimentation rate were slightly increased, 2.9 g/dL and 39 mm/h, respectively. Serum protein electrophoresis showed a polyclonal hypergammaglobulinemia. Endoscopy revealed an irregular ulcer along the small gastric curvature, involving the notch. A body CT scan showed multiple peripheral and bilateral pulmonary nodules (Fig. 1), moderate left pleural effusion, bilateral axillary lymphadenopathy, slight homogeneous hepatosplenomegaly, multiple adenopathies in gastrohepatic ligament, and aortic emergence of the left renal vessels, thickening of the small gastric curve, distension of jejunoileal loops, and mild ascites. The initial presentation of a systemic condition predominantly involving the digestive tract raised the hypothesis of neoplastic disease (namely gastric adenocarcinoma and lymphoma), bacterial or fungal infection as well as other entities such as eosinophilic gastroenteritis. Given the results of endoscopy and body CT scan and while waiting for histological results, colonoscopy and bronchoscopy were done and revealed no changes. Laparoscopy was performed, showing a retroperitoneal mass above and below the stomach, without infiltration of the mesentery. Blood, bone marrow, bronchial secretions, bronchoalveolar lavage, and bronchial biopsies cultures ruled out infectious conditions. The bronchoalveolar lavage revealed the presence of a large amount of eosinophils.
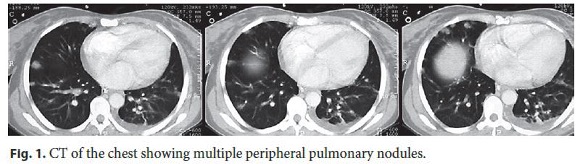
During this period, the patient was under nasogastric intubation with passive drainage, parenteral nutrition, antibiotic therapy with metronidazole, and transfusion support which resulted in progressive resolution of gastrointestinal symptoms and haemoglobin increase to 9.4 g/dL. In the absence of a conclusive diagnosis, other therapeutic interventions were postponed.
However, after the initial clinical course, the patient developed septic shock and acute respiratory distress syndrome. Enterobacter cloacae and Citrobacter freundii were isolated in blood cultures. She was admitted to the intensive care unit, and mechanical ventilation and antibiotic therapy with cefotaxime were initiated. Noteworthy, 500 mg of methylprednisolone and 200 mg of hydrocortisone were administered intravenously on the first day of sepsis, followed by hydrocortisone 100 mg/day for the subsequent 2 days.
After 8 days in the intensive care unit and readmitted to the original medicine department, there was complete resolution of the infection and, interestingly, regression of eosinophilia and hypercalcemia. Meanwhile, the results of the biopsies that were initially performed were obtained. Gastric biopsies (Fig. 2a) showed epithelioid granulomas without necrosis, and colon biopsies (Fig. 2b) revealed lymphocytic infiltration, small lymphoid aggregates, epithelioid granulomas without necrosis, and rare multinucleated giant cells. Retroperitoneal mass biopsies (Fig. 2c) showed fibrofatty and adenopathy tissue, with epithelioid granulomas without necrosis and giant cells, as well as some Langerhans cells. Fungi and bacteria were not identified. Bronchial and lung transbronchial biopsies showed no histopathological changes. The differential diagnosis comprised now a granulomatous disorder that included sarcoidosis with gastrointestinal involvement. Mild hypercalcemia was documented repeatedly, with a maximum value of 10.5 mg/dL, and the angiotensin-converting enzyme was in the upper limit of the reference range (46 U/L), consistent with the hypothesis of sarcoidosis, although the possibility of a rare form of Crohns disease with pleuro-pulmonary involvement could not be completely excluded.
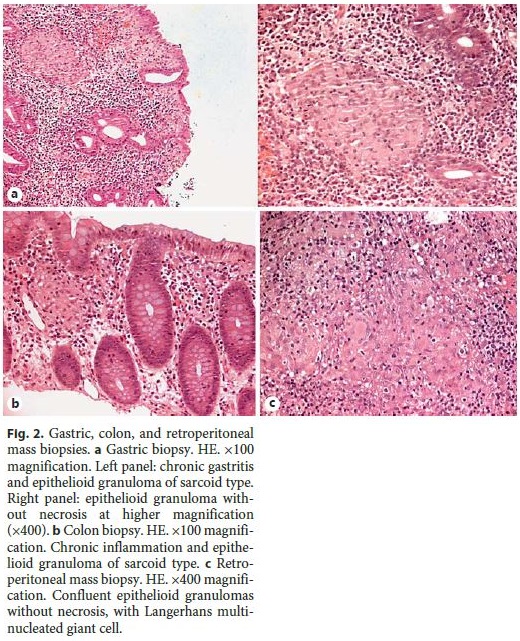
In endoscopy, performed 15 days after readmission to the medicine department, there was regression of gastric lesions and the patient started enteral feeding. Thirty days after readmission to the medicine department, a body CT scan revealed regression of pulmonary nodules, pleural effusion, adenopathy, and hepatosplenomegaly, no distension of the intestinal loops, as well as significant improvement of ascites. Oral feeding was successfully started. The patient was discharged with the diagnosis of multisystemic sarcoidosis, with follow-up in an internal medicine outpatients clinic.
Discussion
This patient had a severe systemic granulomatous disease, which remitted after high-dose but short-duration corticosteroid therapy and raised the diagnostic challenge of distinguishing between sarcoidosis with symptomatic expression of the digestive tract and Crohns disease with pleuro-pulmonary involvement. The similarity between the 2 entities is well known, with the presence of granulomas without caseum in the different organs involved in both. Symptomatic gastrointestinal involvement in sarcoidosis has a prevalence estimated between 0.1 and 0.9%, with evidence of higher subclinical disease in post-mortem studies [4–6]. The incidence of pulmonary complications in inflammatory bowel disease is less than 1%, with most cases occurring in ulcerative colitis [7]. The value of the angiotensin-converting enzyme is a relevant element in the differential diagnosis of these conditions. According to the literature, the angiotensin-converting enzyme is elevated in 60–75% of the patients with untreated sarcoidosis [4, 8]. In our case, the angiotensin-converting enzyme value was in the upper limit of the reference range and thus it was of little help in the differential diagnosis, although in Crohns disease it tends to be frequently decreased, particularly during periods of disease activity. The presence of hypercalcemia was considered as an element in favor of sarcoidosis, since it is a well-recognized metabolic feature of this entity (10–20% of sarcoidosis cases), and it is not expected in the context of Crohns disease [9, 10]. Another element that favors the diagnosis of sarcoidosis in our patient was the presence of splenomegaly and exuberant axillary, mesenteric, and retroperitoneal lymphadenopathy, which are a frequent finding in sarcoidosis [4, 11] and typically absent in the context of Crohns disease [12]. Finally, the clear and complete resolution of all clinical, radiological, and endoscopic manifestations following 500 mg of methylprednisolone and a 3-day course of hydrocortisone in a total dose of 400 mg are more consistent with the expected response in cases of sarcoidosis rather than Crohns disease, where sustained corticosteroid therapy is necessary for remission (Table 1).

As stated above, the digestive tube involvement in sarcoidosis is rare, particularly with symptomatic manifestations. The stomach, especially the antrum, is the organ most often involved. In 2010, Afshar et al. [13], through a MEDLINE search, reported only 26 cases of symptomatic gastric sarcoidosis with well-documented histological evidence of noncaseating granulomas. The most common symptoms of gastric sarcoidosis are abdominal pain, hematemesis, nausea, vomiting, and weight loss [3, 5, 13]. Endoscopy may reveal gastritis, curvature deformation, and ulcers of benign or malignant appearance [3, 13]. Nodular lesions, polyps, and thickening of the folds, with possible deformation and pyloric stenosis, can also be observed. In the literature, there are few case reports of small bowel sarcoidosis [14], with diarrhea, abdominal pain, bleeding, and malabsorption. The involvement of the colon, occasionally described in the literature, is often manifested by abdominal pain, constipation, and bleeding. Colonoscopy may show nodules, polyps, strictures, and erosions [15]. In the reported case, we assume that the clinical presentation surely resulted from sarcoidosis gastric involvement, according to the usual findings previously described in the literature. Regarding small bowel involvement, the investigation performed and the clinical evolution of the patient did not allow us to be conclusive. Interestingly, our patient had no clinical symptoms of colon disease, and the colonoscopy was completely normal. Additionally, colon biopsies revealed the presence of granulomas, illustrating the previously recognized possibility of subclinical involvement of the gut.
In conclusion, we presented a case of multisystemic sarcoidosis where the presenting features were digestive symptoms with documentation of gastric and colon involvement. This case illustrates the difficulties in the differential diagnosis between sarcoidosis with gastrointestinal involvement and Crohns disease with a rare form of pleuropulmonary involvement.
References
1 Baughman RP, et al: Clinical characteristics of patients in a case control study of sarcoidosis. Am J Respir Crit Care Med 2001;164:1885–1889. [ Links ]
2 Rizzato G, et al: The organ-specific extrapulmonary presentation of sarcoidosis: a frequent occurrence but a challenge to an early diagnosis. A 3-year-long prospective observational study. Sarcoidosis Vasc Diffuse Lung Dis 2004;21:119–126. [ Links ]
3 Ebert EC, Kierson M, Hagspiel KD: Gastrointestinal and hepatic manifestations of sarcoidosis. Am J Gastroenterol 2008;103:3184–3192. [ Links ]
4 Iannuzzi MC, Rybicku BA, Teirstein AS: Sarcoidosis. N Engl J Med 2007;357:2153–2165. [ Links ]
5 Vahid B, et al: Sarcoidosis of gastrointestinal tract: a rare disease. Dig Dis Sci 2007;52:3316–3320. [ Links ]
6 Tokala H, Polsani K, Kalavakunta JK: Gastric sarcoidosis: a rare clinical presentation. Case Rep Gastrointest Med 2013;2013:260704. [ Links ]
7 Levenbrown Y, et al: Granulomatous lung disease as the initial presentation of Crohns disease. J Pediatr Gastroenterol Nutr 2009;48:487–490. [ Links ]
8 Studdy PR, Bird R: Serum angiotensin converting enzyme in sarcoidosis – its value in present clinical practice. Ann Clin Biochem 1989;26:13–18. [ Links ]
9 Sharma OP: Hypercalcemia in granulomatous disorders: a clinical review. Curr Opin Pulm Med 2000;6:442–447. [ Links ]
10 Ioachimescu AG, Bauer TW, Licata A: Active Crohns disease and hypercalcaemia treated with infliximab: case report and literature review. Endocr Pract 2008;14:87–92. [ Links ]
11 Sreelesh KP, Arun Kumar ML, Anoop TM: Primary splenic sarcoidosis. Proc (Bayl Univ Med Cent) 2014;27:344–345. [ Links ]
12 Rozen P, et al: Variability of splenomegaly in Crohns disease. Am J Gastroenterol 1977;67:493–498. [ Links ]
13 Afshar K, BoydKing A, Sharma OP, Shigemitsu H: Gastric sarcoidosis and review of the literature. J Natl Med Assoc 2010;102:419–422. [ Links ]
14 Esmadi M, et al: Sarcoidosis: an extremely rare cause of granulomatous enterocolitis. J Gastrointestin Liver Dis 2012;21:423–425. [ Links ]
15 Daldoul S, et al: Unusual presentation of a colonic sarcoidosis. Case Rep Med 2012;2012:169760. [ Links ]
Statement of Ethics
This study did not require informed consent nor review/approval by the appropriate ethics committee.
Disclosure Statement
The authors have no conflicts of interest to declare.
* Corresponding author.
Dr. Catarina Mota
Medicine II Department, Hospital de Santa Maria
Av. Professor Egas Moniz
PT–1649-028 Lisbon (Portugal)
E-Mail catarina.mot@gmail.com
Received: April 9, 2016; Accepted after revision: September 4, 2016
Acknowledgements
We are indebted to Dr. Marisa Teixeira da Silva for fruitful discussions. Grant support was not provided for this article.














