Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
GE-Portuguese Journal of Gastroenterology
versão impressa ISSN 2341-4545
GE Port J Gastroenterol vol.21 no.5 Lisboa out. 2014
https://doi.org/10.1016/j.jpg.2014.06.003
REVIEW ARTICLE
Management of rectal cancer: Times they are changing
Manejo do cancro do reto: os tempos estão a mudar
Marilia Cravoa,b,∗, Tania Rodriguesc, Susana Ourod, Ana Ferreirae, Luis Fériad, Rui Maiod,f
a Serviço de Gastrenterologia, Hospital Beatriz Angelo, Loures, Portugal
b Faculdade de Medicina de Lisboa, Lisboa, Portugal
c Serviço de Oncologia, Hospital Beatriz Angelo, Loures, Portugal
d Serviço de Cirurgia, Hospital Beatriz Angelo, Loures, Portugal
e Serviço de Imagiologia, Hospital Beatriz Angelo, Loures, Portugal
f Faculdade de Ciências Médicas, Lisboa, Portugal
*Corresponding author
ABSTRACT
Approximately one third of all colorectal malignancies are located in the rectum. It as long been recognized that rectal cancers behave differently from colonic tumors, namely in terms of local recurrence. For this reason, specific protocols have been developed to manage this disease both in staging procedures as well as in neoadjuvant and adjuvant chemoradiation treatments. Magnetic resonance imaging is now obligatory for rectal cancer staging. Also, preoperative chemoradiation is recommended in the large majority of locally advanced rectal cancers with obvious advantages in downstaging and downsizing tumors, sometimes allowing spincteric-sparing procedures. Total mesorectum excision is now the rule when operating on rectal cancer. Despite these advances, there are still unanswered questions, namely the utility of using neoadjuvant protocols in low lying, early stage tumors with the aim of performing a local excision procedure and the utility of re-staging the disease after neo-adjuvant treatment. In fact, response to neoadjuvant therapy may become a cornerstone of rectal cancer treatment and individualized therapy. Finally, there is the concern that with current protocols, we are overtreating some patients that would not need such extensive treatment.
In this review, we critically examine recent advances in staging, surgery, and chemoradiation in the management of patients with rectal cancer which have not typically been incorporated in published treatment guidelines.
Keywords: Rectal cancer; Neo-adjuvant chemoradiation; Post-treatment staging
RESUMO
Cerca de um terço de todos os tumores coloretais estão localizados no reto. Desde há longa data que é reconhecido que os tumores do reto têm um comportamento diferente dos tumores do cólon, nomeadamente em termos de recidiva local. Por este motivo, foram desenvolvidos protocolos específicos para manejar esta doença, tanto em termos de estadiamento como em termos de tratamentos neoadjuvantes e adjuvantes. A ressonância magnética é agora obrigatória como método de estadiamento. Por outro lado, a quimioradioterapia preoperatória é recomendada na grande maioria das neoplasias localmente avançadas com vantagens óbvias no downstaging e downsizing dos tumores tratados, permitindo por vezes procedimentos cirúrgicos com conservação do aparelho esfincteriano. A excisão do mesoreto é a regra na cirurgia destes tumores. Apesar destes avanços, continuam a existir questões para as quais não existe uma resposta clara, nomeadamente a utilização de protocolos neoadjuvantes em tumores do terço inferior e precoces com o intuito de realizar uma resseção local bem como a utilidade de re-estadiar estes tumores depois da terapêutica neo-adjuvante. De facto, a resposta à terapêutica preoperatória poder-se-á tornar um fator decisivo na implementação de protocolos de terapêutica individualizada. Finalmente, estudos recentes também levantam a questão de alguns dos doentes selecionados para terapêutica neo-adjuvante estarem a ser sobretratados.
Na atual revisão, tentámos rever de forma crítica os avanços recentes utilizados no estadiamento e tratamento estas neoplasias e que atualmente ainda não estão incorporados nas recomendações publicadas.
Palavras-Chave: Cancro do reto; Tratamento multimodal; Terapêutica individualizada
Introduction
Rectal cancers (RC) comprise approximately 25% of all primary colorectal cancers and follow a different natural disease course compared to colonic tumors. It is well established that surgical approach, local recurrence rates and associated complications of early stage rectal tumors are distinct from colonic cancers. This led to the establishment of specific and distinct protocols for staging and treatment of RC, namely the use of magnetic resonance imaging (MRI) for staging as well as the use of preoperative chemoradiation in selected cases.1 These advances in the management of patients with RC in the last decade contributed to a marked improvement in patients outcomes. In the United States five-year survival increased from of 49.2% in the 70s to 68.5% in the 2000-2005 period. The same trend was observed in Europe.1-3 This improvement may be related not only to disease detection at an earlier stage and widespread use of optimal surgery with total mesorectal excision (TME) but also to a multidisciplinary approach in specialized centers with an increased use of both radiotherapy and chemotherapy, ideally in a neoadjuvant context.3,4
Despite these advances, many issues remain unanswered, namely whether the surgical approach after chemoradiation can be modified based on tumor response, the wait and watch strategy for complete responders and more recently, whether preoperative radiotherapy should be selective, probably based on MRI findings.
In this review, we will review recent changes in the multimodal approach to this tumor.
Tumor staging
Pre operative
Preoperative staging of RC has two main objectives: to define the pertinent anatomy for surgical planning and to determine prognosis. Staging process begins with digital rectal examination. The accuracy of T assessment by digital examination ranges from 58% to 88%, largely depending on the surgeons experience.5 For the precise localization of tumors, especially those beyond the reach of an examining finger, rigid proctoscopy is obligatory and should be considered as the single most useful tool.
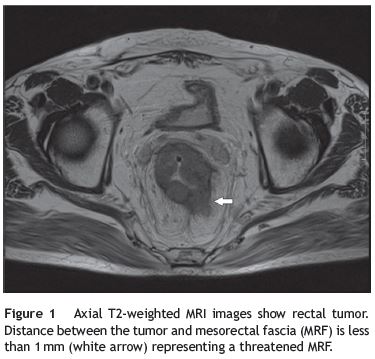
In the initial preoperative setting, superficial, RCs are probably best staged by endoscopic ultrasonography (EUS), whereas MRI should be used in all other RCs because of its proven high sensitivity and specificity in determining N-stage, extramural vascular invasion (EMVI) and circumferential resection margin (CRM).6-8 EUS more accurately determines T category as compared to MRI, although low-lying, very high or near-obstrutive tumors are major drawbacks to the use of EUS. Both MRI and EUS share the risk of understaging small lymph nodes (LN) especially when criteria to distinguish inflammatory from pathologic LN rely mainly on size, as many as 25% of positive LNs are smaller than 3mm.9 Although not included in TNM classification, tumor proximity to the mesorectal fascia (MRF) increases the risk of compromised CRM (CRM+), which is better predicted by MRI and which has been shown to be an independent risk factor of LR when determined by pathological examination.10 The MRF with tumor in close proximity (1mm on MRI) has an increased risk of having a positive CRM as is therefore called a «threatened» MRF (Fig. 1). Recently, the Mercury study was published with the aim of assessing the prognostic relevance of high resolution MRI of CRM.11 The authors concluded that this staging was superior to AJCC TNM-based criteria for assessing both local and distant recurrence. Accordingly, treatment protocols including preoperative radiotherapy should probably consider these findings. For systemic staging, CT scan of the chest, abdomen, and pelvis is usually sufficient. Thus, in practical terms, it is probably more cost-effective to perform CT scan of the chest (which does not need contrast) combined with abdominal and pelvic MRI. Rectal EUS should only be ordered if pelvic MRI is inconclusive in distinguishing T2 vs. T3N0 tumors. PET-CT imaging cannot be recommended routinely since it only changes patient management in 15% of patients.12
Post-treatment staging
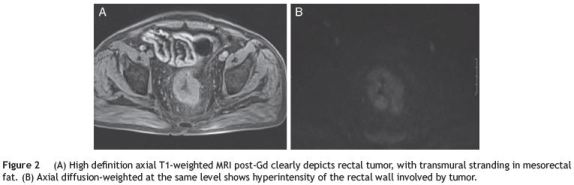
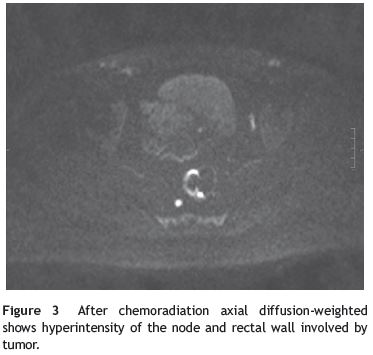
As discussed below, tumors staged as T3 N+ or higher are currently managed with neo-adjuvant (CRT). A new concept states that re-staging after CRT might help to identify complete responders and thereby modify treatment and/or surgical strategy.13 Although this might be debatable, post chemoradiation restaging is a challenge to all imaging modalities due to radiation-induced changes, namely fibrosis, edema, inflammation, and necrosis. The optimal interval between CRT and surgery has not been clearly defined. The Lyon R90-01 study compares a period of less than two weeks with six to eight weeks and found improved T and N downshift with longer intervals.14 In a recent review fromCleveland Clinic, there was a steep increase in pathologic complete response (pCR) after 7 weeks which reached a plateau only after twelve weeks.15 Therefore, an interval of seven weeks after CRT but less than twelve weeks is now recommended for post CRT restaging. In respect to the most appropriate imaging, high definition MRI has been shown to accurately distinguish patients with post-treatment tumors confined to the muscularis propria or more superficially (T0-T2N0), from those with more advanced tumors (Fig. 2).16 Emerging data suggest that reassessment using a combination of high-resolution MRI and diffusion-weighted imaging (DWI), may provide valuable prognostic information before definitive surgery,17-21 as the latter may distinguish viable tumor from fibrosis or inflammatory from neoplastic LN (Fig. 3).22
Surgery
The main aim of surgical treatment of RC is to reduce the risk of residual disease and local relapse while preserving sphincteric, urinary and sexual functions. There are a variety of surgical options in the treatment of RC, which depend not only on tumor location and stage but also on patient sphincter function. Sphincter preservation should not be attempted in those patients with incontinence unless the sphincter can be repaired.
These methods include local procedures, such as transanal local excision and transanal endoscopic microsurgery (TEM) and more invasive procedures involving a transabdominal resection (anterior resection - AR) with colorectal anastomosis, proctectomy with total mesorectal excision (TME) and colo-anal anastomosis or an abdominoperineal resection (APR) with a definitive colostomy.
Local excision methods
Local excision methods are performed transanally with a deep margin outside the muscularis propria into the mesorectal fat and a mucosal margin with 1 cm or more around the target lesion. These procedures are reserved for selected cases with a low likelihood of nodal metastasis. This probability depends on the depth of tumor invasion (T stage), tumor differentiation and LVI. For tumors confined to the submucosa, associated nodal metastasis have been reported in 6-11 percent of patients, while cancers invading the muscularis propria have a 10-20 percent risk of nodal metastases and this risk increases to 33-58 percent in tumors extending into the perirectal fat.23 The incidence of LN metastases also increases dramatically with grade of tumor differentiation with up to 50% of poorly differentiated tumors exhibiting lymph nodes metastasis.24
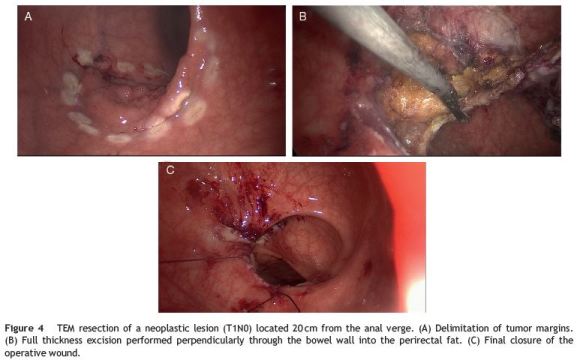
Early RC (confined to the rectal wall without nodal or distant disease - T1N0M0 - with no lymphovascular or perineural invasion, well differentiated and mobile) can be treated with local excision through the Parks transanal local excision or transanal endoscopic microsurgery (TEM).25 Parks transanal local excision is appropriate for selected T1N0M0 early RC less than 3 cmin diameter, located in the 8 cm distal rectum, and occupying less than 40% of the circumference of the rectal wall. TEM is a minimally invasive surgical technique originally described by Buess et al. in the 80s,26-28 which uses a transanal approach with a set of endoscopic surgical instruments that can reach further into the rectum (until 20 cm from the anal verge), along with a form of enhanced or assisted vision.
Both techniques require a full thickness excision performed perpendicularly through the bowel wall into the perirectal fat, with negative (>3mm) deep and mucosal margins, while avoiding fragmentation (Fig. 4).25 However, anatomic considerations may prevent local excision even if tumor staging is appropriate. In large lesions, full thickness excision and primary closure can lead to loss of rectal volume and strictures, creating poor functional results particularly if combined with pelvic radiation.
Local therapies are appealing because of their technical ease, low complication rate, rapid post operative recovery with minimal mortality and morbidity, and above all because they avoid the need for a permanent stoma in early, distally located RCs.26 The major drawback to local procedures, include the absence of pathological staging of nodal involvement, mainly because there is evidence that LN micrometastases also exist in early RC and are unlikely to be identified by endorectal ultrasound. If unfavorable features are observed on pathological examination (high grade, positive or indeterminate margins, perineural or lymphovascular invasion) a radical excision is warranted.25
Although more controversial, T2 lesions can also be successfully treated with local excision, especially if combined with neo-adjuvant CRT, although long term outcomes are unknown. The on-going study ACOSOG trial Z604129 which is a single-arm study evaluating the oncologic outcome of patients with T2N0M0 distal rectal cancers treated with CRT followed by local excision, may shed some light on this issue. Moreover, the observation that a complete mucosal response often corresponds to negative LNs, also supports the strategy of less aggressive surgical treatments in patients submitted to CRT and with a complete clinical and radiological response.13 Close follow-up after this strategy is mandatory.
Radical resections
Local recurrence is a major drawback of isolated locoregional treatments such as surgery. In the late 1970s, Heald et al30 developed the technique of total mesorectal excision (TME) demonstrating that, in some cases, nests of tumor cells outside lymph nodes could be found in the mesorectum and would be left behind by a conventional anterior resection. Using TME alone, Heald et al.31 achieved local recurrence rates of less than 5% and emphasis became focused on the CRM.32-35 Over the last two decades, TME has brought a dramatic improvement in the outcome of surgery for rectal cancer. Anterior resection (AR) is indicated for tumors in the two proximal thirds of the rectum but can also be performed in distal rectal tumors with no involvement of the sphincter. In AR there should be a 5 cm oncological margin from the distal end of the tumor for more proximal tumors but 1-2 cm margins are acceptable for very distal tumors, especially after neo-adjuvant CRT, thereby allowing a sphincter-sparing procedure to be performed. When resection with safe margin carries the loss of continence (direct involvement of the sphincter or levators) or when preoperative continence function is already compromised, an APR is indicated with a definitive colostomy. Although it has been the gold standard of distal rectal cancer surgical therapy, it is nowadays performed in less than 5% of all cases.
Retrospective comparative studies of patients treated with AR and APR, revealed that APR has higher values of local recurrence and reduced survival. This difference in outcome may be explained by the fact that tumors below the peritoneal reflection are usually at a higher stage and have a different lymphatic drainage which might not be included in the package of the TME, with higher incidence of lateral pelvic lymph node involvement.36
In the distal third of the rectum the mesorectum disappears at the top of the sphincter. Below this level, the sphincter constitutes the CRM. Distal rectal tumors have a shorter distance to cross until the CRM as compared to more proximal tumors, protected by a thicker mesorectum. Based on the study of the morphometry of the surgical specimen, West et al.37,38 demonstrated that APR specimens have less tissue volume around the tumor when compared with AR, which was associated with a greater CRM involvement, local recurrence and less overall survival. This problem could be overcome with a new APR, introduced by Holm et al.,39 more cylindrical and closer to the original Miles description, with removal of more tissue around the tumor, reducing the probability of CRM involvement.37,38 This operation involves an abdominal dissection with removal of the rectum and mesorectum down to the levators and a wider perineal dissection, in prone position, with removal of the anal canal, levators and coccyx from below. The perineal defect can be closed with flaps.
Intersphincteric resection
Tumors below 5 cm from the anal verge were not usually considered for a sphincter-sparing surgery because it was not possible to obtain a distal margin of 2 cm through a conventional laparotomy. In this context, the intersphincteric resection (ISR) was introduced as a form of treatment for distal rectal tumors, considering that the mesorectum terminates at the top of the sphincter complex.40
ISR is indicated for well differentiated tumors located below 5 cm from the anal verge with predictably negative CRM in MRI. Involvement of the internal sphincter is not a contraindication. In contrast, ISR should not be performed in fixed tumors, involving the external sphincter or levators as well as in patients with poor preoperative continence.38,40 Limitations for sphincter-sparing procedures are beginning to be regarded as mostly functional and not just oncological.41,42
Despite laparoscopic approach of colon cancer is now universally accepted, the extension of this approach to RC is still controversial. There is an evident lack of data and the CLASICC study remains the only randomized controlled multicentre trial comparing the results of classic and laparoscopic approach to rectal cancer surgery. Some groups still express oncological concerns based on the first results of this study, which reported higher rates of CRM involvement and a trend for worst sexual male function in the laparoscopic group. These results were not reproduced and at 3 and 5 years there are no significant differences between both approaches,43-45 thereby encouraging the use laparoscopic approach in RC.46
Chemoradiation treatment
Previous studies have consistently shown that postoperative 5-fluorouracil (5FU)-based chemoradiation significantly improves local control and survival compared with surgery alone.47-49 When radiotherapy was compared to concurrent CRT, the German Rectal Cancer Trial50 confirmed that CRT delivered preoperatively, results in a significant decrease in acute and late toxicities, concomitantly with a better local control of disease and a higher chance of sphincter preservation. Since then, the standard treatment for locally advanced, clinically resectable (T3 and/or N+) rectal cancer is preoperative CRT.
Although 5-fluorouracil continuous infusion (5FU-CI) is the conventional regimen used,47-49 two recently published studies showed that capecitabine has similar rates of pCR, sphincter-sparing surgery, and toxicity,51,52 and so both agents can be used in the neo-adjuvant setting.
In patients with pretreatment stage I disease (T2N0), neoadjuvant CRT therapy may be considered in distally located tumors with the aim of downsizing, thereby increasing the chances of a sphincter sparing procedure53,54; however the benefits of this strategy remains unproven.
Preoperative vs. Postoperative chemoradiation
Two randomized trials compared preoperative vs. postoperative chemoradiation for clinically resectable rectal cancer. The German trial55 completed the planned accrual of more than 800 patients with rectal cancers less than 16 cm from the anal verge who were randomized to preoperative CRT vs. postoperative CRT. Patients who received preoperative therapy had a significant decrease in local recurrence (6% vs. 15%; P = .006), acute toxicity (27% vs. 40%; P = .001), and chronic toxicity (14% vs. 24%; P = .012) when compared with postoperative therapy. In addition, there was a significant increase in sphincter preservation surgeries (39% vs. 20%; P = .004). No differences were observed in 5-year survival. At 10 years the local control benefit of preoperative vs. postoperative therapy was still observed. In contrast to these results which clearly favorable to preoperative treatment, in the NSABP R-03 trial56 this benefit was not as obvious. However, the results of the NSABP trial should be interpreted with caution because only 267 of the 900 planned patients were accrued, limiting the statistical power to detect differences. Based on these results, preoperative chemoradiation remains the standard of care.
Short-course radiotherapy vs. long-course chemoradiation
The two main strategies of preoperative radiotherapy are long-course chemoradiation and short-course radiation. The first, involves the delivery of a long course of preoperative radiotherapy using conventional doses of 1.8-2Gy per fraction over 5-6 weeks, with a total dose of 45-50.4 Gy. This approach typically involves the administration of concurrent 5FU or capecitabine-based chemotherapy and is the most accepted approach worldwide.52
The rationale for giving chemotherapy concurrently with radiotherapy is that it potentiates local radiotherapy sensitization and has the potential to induce tumor downsizing and/or downstaging, hopefully improving rates of sphincter sparing procedures and increasing rates of pathological complete response (pCR).36 The second, traditionally used in Scandinavia, consists of short-course preoperative radiotherapy (SCPRT) delivering a total dose of 25 Gy over 5 days (5 fractions) without chemotherapy, followed by surgery within 10 days of the first session of radiotherapy.57-59 The rationale for this regimen is that the short time period for delivery of the dose may counteract the effects of accelerated cellular repopulation, a phenomenon characteristic of tumor cells exposed to radiotherapy.
In patients with T3/4 rectal cancer, the delivery of a long course of preoperative radiotherapy concurrent with chemotherapy is associated with a relative risk reduction in local recurrence of approximately 50%, whereas shortcourse radiotherapy does not result in apparent downstaging of tumors in terms of nodal status.58
Two large RCT studied the effect of SCPRT in both local recurrence and 5-year survival.57,58 Although the results of both of these trials favor SCPRT, both were performed before the widespread introduction of TME surgery and, therefore, it remains to be proven whether this beneficial effect would also be observed if TME had been performed.
Therefore on the basis of available evidence, longcourse chemoradiation appears preferable, particularly for patients with distal tumors or threatened margins.
Change of surgical strategy based on post-treatment staging
Restaging after neo-adjuvant CRT might help to identify responders to therapy in whom planned treatment based on the original presentation might no longer be indicated. As discussed earlier, the post-treatment assessment often enables sphincter preservation due to tumor downsizing and T or N downshifting.13 A natural assumption would be that tumors initially staged as T3N0 who after CRT had a downshilt to T0/T1N0 could be safely managed by local excision. Further supporting this practice, in the German Rectal Cancer Study Group,48-50 the surgeons pretreatment surgical recommendation was compared with the surgical procedure after neo-adjuvant CRT. Forty percent of patients originally thought to need APR actually underwent a sphincter-preserving procedure without oncologic compromise at a median follow-up of 45 months. However, there are no prospective clinical trials supporting this strategy. The on-going ACOSOG trial Z6041 which is a single-arm study evaluating the outcome of patients with T2N0M0 distal RC treated with CRT followed by local excision procedures, will certainly help to clarify this issue.29
CRT causes tumor necrosis, which is then replaced by inflammatory tissue and ultimately fibrosis. Pathologists can quantify the ratio of viable tumor cells to fibrosis to generate a tumor regression grade (TRG).60 In regard to lymph node response to CRT the only accurate method is pathologic examination of the surgical specimen, but previous observations strongly support the hypothesis that there is a close relationship between primary tumor post-treatment T stage and risk of persistent lymph node metastasis.61 For this reason, Kosinsky et al.13 consider that mucosal response can be viewed as a proxy for LN response. Using staging and neoadjuvant CRT protocols discussed above, we may expect rates of pathologic response ranging from 5% to 42%.62 For this reason, some authors now propose a new algorithm in which surgical approach is based on response to neoadjuvant treatment.13 Although not validated, it provides a framework for the incorporation of treatment response in operative planning and sets the stage for considering less radical operative strategies or even a wait and watch strategy in which highly selected RCs are not operated immediately.13 This strategy is really a «no-immediate» surgical approach, recommended only in highly selected patients who require intensive follow-up with rectal and endoscopic examinations, especially during the first year. Full excisional biopsy is performed in equivocal cases. Disease recurrence in patients previously identified as having had a complete clinical response, requires surgical salvage which has been shown not to compromise outcome as compared with patients who received immediate surgery after neoadjuvant CRT.63
Postoperative adjuvant chemotherapy after neoadjuvant chemoradiation and surgery
The neoadjuvant CRT approach commits patients to the entire three component package of CRT, surgery and adjuvant therapy. Beets et al.53 performed a pooled analysis of 2724 patients who received preoperative chemoradiation. Overall, 41% received postoperative chemotherapy and there was no benefit in disease-free survival in the subset of patients with ypT0N0 or ypT3-4Nx disease. Patients with ypT1-2N0 disease had the greatest benefit, probably because patients who were responders to CRT were also selected. Thus, although its benefit remains controversial, most investigators feel that it is reasonable to use the same adjuvant chemotherapy for colon cancer.54 For patients selected to receive postoperative adjuvant chemotherapy, 4 months (8 cycles) of FOLFOX/CAPOX or capecitabine monotherapy is recommended although carrying the risk of potentially overtreating some patients.
Novel approaches to neo-adjuvant treatment - the PROSPECT study
As stated before, contemporary management of locally advanced rectal cancer involves preoperative chemoradiation, followed by surgery and then adjuvant systemic chemotherapy. However, although before the advent of TME, LR was a major problem, nowadays the vast majority of rectal cancer deaths are from disseminated metastatic disease, which reinforces the importance of systemic treatment.64 The problem with the current strategy is that neoadjuvant CRT utilizes either 5-fluorouracil or capecitabine solely as sensitizing agents. Effective chemotherapy with FOLFOX or CAPOX will only start 20-24 weeks from diagnosis, allowing for possible dissemination of micrometastases. As a result, the Alliance for Clinical Trials in Oncology launched the PROSPECT trial early in 2012 - Preoperative Radiation or Selective Preoperative radiation and Evaluation before Chemotherapy and TME,65 with the aim of moving systemic therapy more proximally in the total treatment course. It is a phase II/III randomized trial to evaluate the impact of selective use of radiation in the era of TME and high-quality MRI imaging. Therefore, in the intervention arm, patients would first start with systemic treatment (FOLFOX×6 cycles) with restaging of primary tumor after that. If any progression was observed or regression was lower than 20%, the classic CRT protocol would be performed. If not, the patient would proceed immediately to low anterior resection with TME, eventually followed by an additional 6 cycles. Patients with unexpected positive surgical margins would receive postoperative radiation. This novel approach incorporate selective rather than consistent use of radiation in the treatment of mid RC and customizes subsequent treatment based on response to neoadjuvant FOLFOX.
Conclusions
Multimodal treatment of RC with preoperative CRT in clinical T3N1 cases has improved local recurrence rates and, in the some cases, has allowed a sphincter preservation procedure. TME is now part of an optimal radical resection for RC with the emphasis on CRM.
However, recent studies start to question this classic approach because of a number of issues. First, there is clear evidence that pathologic stage after neo-adjuvant CRT more accurately indicates prognosis than initial clinical stage. However, NCCN and ESMO treatment guidelines, besides not recommending restaging after neoadjuvant therapy, still consider cTNM staging as an indicator for such therapy whereas recent studies demonstrate that high definition MRI with accurate staging of CRM, may be a better predictor for both local and distant recurrence. Also, preoperative CRT might also be considered in patients with T2N0 distally located tumors and, in very carefully selected responders, a wait and watch strategy may be recommended. Finally, there are now concerns that by submitting to CRT all patients clinically staged as T3N+, we might be (i) overtreating some patients (ii) delaying systemic treatment to 4-5 months after diagnosis thereby increasing the risk to systemic dissemination.
Therefore, management of RC is clearly going to change in a near future and it is of paramount importance that these patients are referred to specialized centers where these multiple and possible strategies are extensively discussed in a multidisciplinary team. Gastroenterologists should definitely be part of this team!
References
1. American Cancer Society. Colorectal Cancer Facts & Figures 2008-2010. http://www5.cancer.org/downloads/STT/F861708 [ Links ]
2. van Gijn W, Krijnen P, Lemmens VE, den Dulk M, Putter H, van de Velde CJ. Quality assurance in rectal cancer treatment in the Netherlands: a catch up compared to colon cancer treatment.Eur J Surg Oncol. 2010;36:340-4. [ Links ]
3. Verdecchia A, Guzzinati S, Francisci S, De Angelis R, Bray F, Allemani C, et al. Survival trends in European cancer patients diagnosed from 1988 to 1999. Eur J Cancer. 2009 Apr;45(6):1042-66. [ Links ]
4. Påhlman L, Bohe M, Cedermark B, Dahlberg M, Lindmark G, Sjödahl R, et al. The Swedish rectal cancer registry. Br J Surg. 2007 Oct;94(10):1285-92. [ Links ]
5. Schaffzin DM, Wong WD. Endorectal ultrasound in the preoperative evaluation of rectal cancer. Clin Colorectal Cancer. 2004;4:124-32. [ Links ]
6. Glynne-Jones R, Kronfli M. Locally advanced rectal cancer: a comparison of management strategies. Drugs. 2011;71:1153-77. [ Links ]
7. Augestad KM, Lindsetmo RO, Stulberg J, Reynolds H, Senagore A, Champagne B, et al., International Rectal Cancer Study Group (IRCSG). International preoperative rectal cancer management: staging, neoadjuvant treatment, and impact of multidisciplinary teams. World J Surg. 2010 Nov;34(11):2689-700. [ Links ]
8. MERCURY Study Group. Extramural depth of tumor invasion at thin section MR in patients with rectal cancer: results of the mercury study. Radiology. 2007;243:132-9. [ Links ]
9. Brown G, Radcliffe AG, Newcombe RG, Dallimore NS, Bourne MW, Williams GT. Preoperative assessment of prognostic factors in rectal cancer using high-resolution magnetic resonance imaging. Br J Surg. 2003 Mar;90(3):355-64. [ Links ]
10. Quirke P, Steele R, Monson J, Grieve R, Khanna S, Couture J, et al. Effect of the plane of surgery achieved on local recurrence in patients with operable rectal cancer: a prospective study using data from the MRC CR07 and NCIC-CTG C016 randomised clinical trial. Lancet. 2009 Mar;373(9666):821-8. [ Links ]
11. Taylor FG, Quirke P, Heald RJ, Moran BJ, Blomqvist L, Swift IR, et al. Preoperative magnetic resonance imaging assessment of circumferential resection margin predicts disease-free survival and local recurrence: 5-year follow-up results of the MERCURY Study. J Clin Oncol. 2013 Jun;32(1):34-43. [ Links ]
12. Vriens D, de Geus-Oei LF, van der Graaf WT, Oyen WJ. Tailoring therapy in colorectal cancer by PET-CT. Q J Nucl Med Mol Imaging. 2009;53:224-44 [ Links ]
13. Kosinsky L, Habr-Gama A, Ludwig K, Perez R. Shifting concepts in rectal cancer management. A review of contemporary primary rectal cancer treatment strategies. CA Cancer J Clin. 2012;62:173-202. [ Links ]
14. Francois Y, Nemoz CJ, Baulieux J, Vignal J, Grandjean JP, Partensky C, et al. Influence of the interval between preoperative radiation therapy and surgery in downstaging ad on the rate of sphincter-sparing surgery for rectal cancer: the Lyon R90-01 randomized trial. J Clin Oncol. 1999 Aug;17(8):2396. [ Links ]
15. Kalady MF, de Campos-Lobato LF, Stocchi L, Geisler DP, Dietz D, Lavery IC, et al. Predictive factors of pathologic complete response after neoadjuvant chemoradiation for rectal cancer. Ann Surg. 2009 Oct;250(4):582-9. [ Links ]
16. Engelen SM, Beets-Tan RG, Lahaye MJ, Lammering G, Jansen RL, van Dam RM, et al. MRI after chemoradiatiotherapy of rectal cancer: a useful tool to select patients for local excision. Dis Colon Rectum. 2010 Jul;53(7):979-86. [ Links ]
17. Pomerri F, Pucciarelli S, Maretto I, Zandonà M, Del Bianco P, Amadio L, et al. Prospective assessment of imaging after preoperative chemoradiotherapy for rectal cancer. Surgery. 2011 Jan;149(1):56-64. [ Links ]
18. Barbaro B, Fiorucci C, Tebala C, Valentini V, Gambacorta MA, Vecchio FM, et al. Locally advanced rectal cancer: MR imaging in prediction of response after preoperative chemotherapy and radiation therapy. Radiology. 2009 Mar;250(3):730-9. [ Links ]
19. Dresen RC, Beets GL, Rutten HJ, Engelen SM, Lahaye MJ, Vliegen RF, et al. Locally advanced rectal cancer: MR imaging for restaging after neoadjuvant radiation therapy with concomitant chemotherapy. Part I. Are we able to predict tumor confined to the rectal wall? Radiology. 2009 Jul;252(1):71-80. [ Links ]
20. Koh DM, Chau I, Tait D, Wotherspoon A, Cunningham D, Brown G. Evaluating mesorectal lymph nodes in rectal cancer before and after neoadjuvant chemoradiation using thin-section T2-weighted magnetic resonance imaging. Int J Radiat Oncol Biol Phys. 2008 Jun;71(2):456-61. [ Links ]
21. Dzik-Jurasz A, Domenig C, George M. Diffusion MRI for prediction of response of rectal cancer to chemoradiation. Lancet. 2002;27:307-8. [ Links ]
22. Lambregts DM, Vandecaveye V, Barbaro B, Bakers FC, Lambrecht M, Maas M, et al. Diffusion-weigthed MRI for selection of complete responders after chemoradiation for locally advanced rectal cancer: a multicenter study. Ann Surg Oncol. 2011 Aug;18(8):2224-31. [ Links ]
23. Spratt JS. Adenocarcinoma of the colon and rectum. In: Neoplasms of the colon, rectum and anus. Philadelphia: WB Saunders; 1984. p. 206-13. [ Links ]
24. Brodsky J, Cohen R, Minsky B. Variables correlated with the risk of lymph node metastasis in early rectal cancer. Cancer. 1992;69:322-6. [ Links ]
25. Tjandra JJ, Kilkenny JW, Buie WD, Hyman N, Simmang C, Anthony T, et al. Practice parameters for the management of rectal cancer (revised). Dis Colon Rectum. 2005 Mar;48(3):411-23. [ Links ]
26. Cataldo P. Transanal endoscopic microsurgery. Surg Clin North Am. 2006;86:915-25. [ Links ]
27. Buess G, Theiss R, Gunther M, et al. Endoscopic operative procedure for the removal of rectal polyps. Coloproctol. 1984;184:252-61. [ Links ]
28. Suppiah A, Maslekar S, Alabi A, et al. Transanal endoscopic microsurgery in early rectal cancer: time for a trial? Colorectal Dis. 2008;10:314-32. [ Links ]
29. Ota DM, Nelson H, ACOSOG Group Co-Charis. Local excision of rectal cancer revisited: ACOSOG protocol Z6041. Ann Surg Oncol. 2007;14:271. [ Links ]
30. Heald R, Husband E, Ryall R. The mesorectum in rectal cancer surgery: the clue to pelvic recurrence? Br J Surg. 1982;69(10):613-6. [ Links ]
31. Heald R, Ryall R. Recurrence and survival after total mesorectal excision of rectal cancer. Lancet. 1986;1(8496):1479-82. [ Links ]
32. Nagtegaal I, Quirke P. What is the Role for the circumferential margin in the modern treatment of rectal cancer? J Clin Oncol. 2008;26(2):303-12. [ Links ]
33. Wibe A, Rendedal PR, Svensson E, Norstein J, Eide TJ, Myrvold HE, et al. Prognostic significance of the circumferential resection margin following total mesorectal excision for rectal cancer. Br J Surg. 2002 Mar;89(3):327-34. [ Links ]
34. Barabouti D, Wong W. Current management of rectal cancer: total mesorectal excision (nerve sparing) technique and clinical outcome. Surg Clin North Am. 2005;14:137-55. [ Links ]
35. Nicholls R, Tekkis P. Multidisciplinary treatment of cancer of the rectum: a European approach. Surg Clin North Am. 2008;17:533-51. [ Links ]
36. NCCN Clinical Practice Guidelines in Oncology. Rectal cancer. Fort Washington, PA: National Comprehensive Cancer Network; 2012. [ Links ]
37. West NP, Finan PJ, Anderin C, Lindholm J, Holm T, Quirke P. Evidence of the oncological superiority of cylindrical abdominoperineal excision for low rectal cancer. J Clin Oncol. 2008 Jul;26(21):3517-22. [ Links ]
38. Marr R, Birbeck K, Garvican J, Macklin CP, Tiffin NJ, Parsons WJ, et al. The moderns abdominoperineal excision. The next challenge after total mesorectal excision. Ann Surg. 2005 Jul;242(1):74-82. [ Links ]
39. Holm T, Ljung A, Häggmark T, Jurell G, Lagergren J. Extended abdominoperineal resection with gluteus maximus flap reconstruction of the pelvic floor for rectal cancer. Br J Surg. 2007 Feb;94(2):232-8. [ Links ]
40. Schiessel R, Novi G, Holzer B, Rosen HR, Renner K, Hölbling N, et al. Technique and long-term results of intersphincteric resection for low rectal cancer. Dis Colon Rectum. 2005 Oct;48(10):1858-67. [ Links ]
41. Wallace M, Glynne-Jonest R. Saving the sphincter in rectal cancer: are we prepared to change practice? Colorectal Dis. 2007;9:302-9. [ Links ]
42. Rullier E. Sphincter saving is the primary objective for local treatment of cancer of the lower rectum. Lancet. 2006;7:775-7. [ Links ]
43. Jayne DG, Guillou PJ, Thorpe H, Quirke P, Copeland J, Smith AM, et al. Randomized trial of laparoscopic-assisted resection of colorectal carcinoma: 3 year results of the UK MRC CLASICC Trial Group. J Clin Oncol. 2007 Jul;25(21):3061-968. [ Links ]
44. Jayne DG, Thorpe HC, Copeland J, Quirke P, Brown JM, Guillou PJ. Five-year follow up of the Medical Research Council CLASICC trial of laparoscopically assisted versus opena surgery for colorectal cancer. Br J Surg. 2010 Nov;97(11):1638-45. [ Links ]
45. Guillou PJ, Quirke P, Thorpe H, Walker J, Jayne DG, Smith AM, et al. Short-term endpoints of conventional versus laparoscopicassisted surgery in patients with colorectal cancer (MRC CLASICC trial): multicentre, randomised controlled trial. Lancet. 2005 May;365(9472):1718-26. [ Links ]
46. McKay GD, Morgan MJ,Wong SK, Gatenby AH, Fulham SB, Ahmed KW, et al. Improved short-term outcomes of laparoscopic versus open resection for colon and rectal cancer in an area health service: a multicenter study. Dis Colon Rectum. 2012 Jan;55(1):42-50. [ Links ]
47. Fisher B, Wolmark N, Rockette H, Redmond C, Deutsch M, Wickerham DL, et al. Postoperative adjuvant chemotherapy or radiation therapy for rectal cancer: results from NSABP protocol R-01. J Natl Cancer Inst. 1988 Mar;80(1):21-9. [ Links ]
48. Gastrointestinal Tumor Study Group. Prolongation of the disease-free survival in surgically treated rectal carcinoma. N Engl J Med. 1985;312:1465-72. [ Links ]
49. Krook JE, Moertel CG, Gunderson LL, Wieand HS, Collins RT, Beart RW, et al. Effective surgical adjuvant therapy for high-risk rectal carcinoma. N Engl J Med. 1991 Mar;324(11):709-15. [ Links ]
50. Sauer R, Becker H, Hohenberger W, Rödel C, Wittekind C, Fietkau R, et al. Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med. 2004 Oct 21;351(17):1731-40. [ Links ]
51. Roh MS, Yothers GA, OConnell MJ, et al. The impact of capecitabine and oxaliplatin in the preoperative multimodality treatment in patients with carcinoma of the rectum NSABP R-04. J Clin Oncol. 2011;29, abstract 3503 http://www.asco.org (accessed 5.4.11). [ Links ]
52. Hofheinz R, Wenz FK, Post S, et al. Capecitabine (Cape) versus 5-fluorouracil (5-FU) based (neo)adjuvant chemoradiotherapy (CRT) for locally advanced rectal cancer (LARC): long-term results of a randomized, phase III trial. J Clin Oncol. 2011;29, abstract 3504 http://www.asco.org (accessed 5.4.11). [ Links ]
53. Beets GL, Mass M, Nelemans PJ, et al. Evaluation of response after chemoradiation for rectal cancer as a predictive factor for the benefit of adjuvant chemotherapy: a pooled analysis of 2724 individual patients. J Clin Oncol. 2011;29, abstract 361 http://www.asco.org (accessed 6.4.12). [ Links ]
54. Valentini V, Aristei C, Glimelius B, Minsky BD, Beets-Tan R, Borras JM, et al. Multidisciplinary Rectak Cancer Management : 2nd European Rectal Cancer Consensus Conference (EURECA-CC2). Radiother Oncol. 2009 Aug;92(2):148-63. [ Links ]
55. Roh MS, Colangelo LH, OConnell MJ, Yothers G, Deutsch M, Allegra CJ, et al. Preoperative multimodality therapy improves disease-free survival in patients with carcinoma of the rectum: NSABP R-03. J Clin Oncol. 2011 Nov;29(31):5126-30. [ Links ]
56. Roh MS, Colangelo LH, OConnell MJ, et al. Preoperative multimodality therapy improves disease-free survival in patients with carcinoma of the rectum: NSABP-R03. J Clin Oncol. 2009;27:5124-30. [ Links ]
57. Swedish Rectal Cancer Trial. Improved survival with preoperative radiotherapy in resectable rectal cancer. N Engl J Med. 1997;336:980-7. [ Links ]
58. Kapiteijn E, Marijnen CA, Nagtegaal ID, Putter H, Steup WH, Wiggers T, et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer. N Engl J Med. 2001 Aug;345(9):638-46. [ Links ]
59. Sebag-Montefiore D, Stephens RJ, Steele R, Monson J, Grieve R, Khanna S, et al. Preoperative radiotherapy versus selective postoperative chemoradiotherapy in patients with rectal cancer (MRC CR07 and NCIC-CTG C016): a multicenter, randomized trial. Lancet. 2009 Mar 7;373(9666):811-20. [ Links ]
60. Dworak O, Keilholz L, Hoffman A. Pathological features of rectal cancer after preoperative radiochemotherapy. Int J Colorectal Dis. 1997 Feb;12(2):19-23. [ Links ]
61. Pucciarelli S, Capirci C, Emanuele U, Toppan P, Friso ML, Pennelli GM, et al. Relationship between pathologic T-stage and nodal metastasis after preoperative chemoradiotherapy for locally advanced rectal cancer. Ann Surg Oncol. 2005;12:111-6. [ Links ]
62. Sanghere P, Wong DW, McConkey CC, Geh JI, Hartley A. Chemoradiotherapy for rectal cancer: an updated analysis of factors affecting pathological response. Clin Oncol. 2008;20:176-86. [ Links ]
63. Habr-Gama A, Perez RO, Proscurshim I, Nunes Dos Santos RM, Kiss D, Gama-Rodrigues J, et al. Interval between surgery and neoadjuvant chemoradation therapy for distal rectal cancer: does delayed surgery have an impact on outcome? Int J Radiat Oncol Biol Phys. 2008 Jul;71(4):1181-8. [ Links ]
64. Schrag D. Evolving role of neoadjuvant therapy in tectal cancer. Curr Treat Options Oncol. 2013;14:350-64. [ Links ]
65. Schrag D, Weiser MR, Goodman KA, Gonen M, Hollywood E, Cercek A, et al. Neoadjuvant chemotherapy without routine use of radiation therapy for patients with locally advanced rectal cancer: a pilot trial. J Clin Oncol. 2014 Feb;32(6):513-8. [ Links ]
*Corresponding author
E-mail address: marilia.cravo@sapo.pt (M. Cravo).
Ethical disclosures
Protection of human and animal subjects. The authors declare that no experiments were performed on humans or animals for this investigation.
Confidentiality of data. The authors declare that they have followed the protocols of their work center on the publication of patient data and that all the patients included in the study have received sufficient information and have given their informed consent in writing to participate in that study.
Right to privacy and informed consent. The authors must have obtained the informed consent of the patients and/or subjects mentioned in the article. The author for correspondence must be in possession of this document.
Conflicts of interest
The authors have no conflicts of interest to declare.
Received 12 February 2014; accepted 17 June 2014
Acknowledgement
We are grateful to Professor Antonio Alberto Santos for the critical review of the manuscript and helpful suggestions.














