Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Acta Obstétrica e Ginecológica Portuguesa
Print version ISSN 1646-5830
Acta Obstet Ginecol Port vol.12 no.3 Coimbra Sept. 2018
ORIGINAL STUDY/ESTUDO ORIGINAL
Transvaginal repair of genital prolapse with a Prolift system: complications and outcomes after 7 years of follow-up
Tratamento cirúrgico do prolapso genital com sistema Prolift: avaliação após 7 anos de monitorização
Fernanda Santos*, Isabel Duarte**, António Correia**, António Santiago***
Centro Hospitalar de Leiria - Hospital de St. André
*Interna de Ginecologia e Obstetrícia
**Assistente Graduado de Ginecologia e Obstetrícia
***Assistente Graduado Sénior de Ginecologia e Obstetrícia
Endereço para correspondência | Dirección para correspondencia | Correspondence
ABSTRACT
Overview: Transvaginal mesh (TM) procedures has revolutionized the pelvic organ prolapse treatment. Although high success, since 2008 US Food and Drug Administration alert about potential complications that conduct to withdrawn from the market of several mesh systems.
Aim: To report long-term outcomes after a polypropylene mesh (Ethicon Gynecare ProliftTM) procedure.
Study Design: Cross-Sectional Study
Methods: It was performed a retrospective analysis of 54 women submitted to Prolift transvaginal mesh, at Urogynecology department of Centro Hospitalar de Leiria, between January 2009 to January 2017. Outcome was assessed using the Pelvic Organ Prolapse Quantification system and complications were reported with the Prosthesis Complication Classification Code designed by the International Continence Society/International Urogynecological Association. Anatomic failure was defined as recurrent prolapse (grade≥2), at the same compartment. Statistical analysis was performed with STATA 13.1 (significance if p < 0.05).
Results: Fifty-four menopausal women were followed at a median time of four years (1-7). Eighty-one percent had more than two vaginal deliveries and 70% were obese (BMI >= 30 kg/m2). Ninety-one percent had POP stage higher than 2. Total mesh was done in 10 patients (19%), isolated anterior mesh in 40 (74%) and isolated posterior mesh in 4 patients (7%). Complications happened in nine patients (17%). We reported five anatomic failures (overall success of 91%). We found statistical association between failure, POP grade and type of mesh repair. (p<0.05).
Conclusions: The Prolift transvaginal mesh system seems to be associated with mild complications and a high overall success.
Keywords: Surgical mesh; Pelvic organ prolapse; Complications; Treatment outcome.
Introduction
Pelvic organ prolapse (POP) is a herniation of the pelvic organs to or beyond the vaginal walls, which occurs when the support tissues are weak or stretched.
The different types of prolapse include: the apical prolapse (uterus or vaginal vault); the anterior vaginal wall prolapse (i.e. cystocele: bladder; urethrocele: urethra; paravaginal defect: pelvic fascia defect) and the posterior vaginal wall prolapse (i.e. enterocele: small bowel; rectocele: rectum; perineal deficiency). A woman can present with different types of prolapse, simultaneously1,2.
POP is a common condition, affecting approximately 40% of women over 50, of whom only 25% are symptomatic3.The etiology of POP is complex and multifactorial. Possible risk factors include ageing, obesity, parity, hysterectomy, menopause, congenital or acquired connective tissue abnormalities, denervation or weakness of the pelvic floor and factors associated with chronically raised intra-abdominal pressure1,2.
The severity of prolapse is graded by the Pelvic Organ Prolapse Quantitation system (POPQ), the classification of choice of the International Continence Society (ICS), the American Urogynecologic Society (AUGS), and the Society of Gynecologic Surgeons (SGS). The POPQ system is objective and a site-specific system, that involves quantitative measurements of various points representing anterior, apical and posterior vaginal prolapse, creating a “topographic” map of the vagina. For hysterectomized women there are three stages and for non-hysterectomized women there are four4.
Women with prolapse could be asymptomatic or could have a variety of pelvic floor symptoms. POP-related-symptoms may be vaginal bulge feeling, problems with voiding or incontinence for stool and/or urine, backache and pelvic heaviness. The clinical manifestations could be so serious that could impact severely the quality of life. However, only some of which are directly related to the stage or prolapsed organ5.
Treatment depends on the severity of the prolapse, its symptoms, the woman's general health, and surgeon preference and capabilities. There are different treatment options, such as pelvic floor muscle training, reduction of known risk factors, pessary therapy and surgical repair6.
Generally, conservative treatments are reserved for women with a non-bothersome POP, for those who wish to conceive and for those who refuse or have contraindications to surgery1.
Concerning to the surgical approach, there are a wide variety of abdominal and vaginal techniques, which means a lack of consensus or guidelines to the optimal treatment1,7. The POP surgeries are one example of surgical treatments for non-life-threatening conditions, which means that they are characterized by the main goal of improving the patient's quality of life. Therefore, POP surgery could be reconstructive (normal anatomy is restored) or obliterative (remove partial or total of the prolapsed organ; i.e. Lefort colpocleisis)7. Traditionally, surgical repair has been done using patient's native tissue (such as anterior and/or posterior colporrhaphy and apical suspensions). Although generally safe, the failure rate for primary repair of POP is reported as being high (approximately 20-30%)8. Due to high recurrence rate, alternative procedures have been developed, involving transvaginal placement of synthetic mesh and biological grafts. These procedures were believed to be potentially less invasive and more effective with lower risk of recurrence, such as the application of tapes for continence surgery and meshes for herniorrhaphies. However, new mesh kits were brought to market faster than short-term safety data was obtained and much sooner than long-term safety data could be obtained. Under a poor premarket clinical evaluation, concerns have been raised on the safety of transvaginal meshes after serious adverse events had been reported3,9. In 2008 and 2011 the United States (US) Food and Drug Administration (FDA) issued public health warnings regarding the frequency of complications associated with use of transvaginal mesh for pelvic organ prolapse. The most reported complications were infection, erosion, mesh exposure, perforation, dyspareunia, acute or chronic pain, urinary problems and vaginal stricture10. Given the seriousness and the number of complications reported, several mesh systems were withdrawn from the market.
European, Canadian, and US regulatory boards have released statements regarding the use of synthetic mesh in gynecologic surgery. The European Commission's Scientific Committee on Emerging and Newly Identified Health Risks stated in 2015 that “The implantation of any mesh for the treatment of POP via the vaginal route should only be considered in complex cases, in particular, after failed primary repair surgery” 11. In 2016 FDA finalized regulations that reclassified surgical mesh for transvaginal POP repair as a class III (high risk) device and required manufacturers to submit premarket approval applications to support the safety and effectiveness of synthetic mesh use8-11.
Since 2009, selected patients with primary (grade ≥ 2) or recurrent prolapse in our unit were offered the polypropylene mesh kit (Ethicon Gynecare Prolift). This mesh was chosen, as there were long-term data associated with its use as mid-urethral sling for stress urinary incontinence (SUI) treatment (Gynecare Tension free vaginal tape (TVT) Obturator system). Due to FDA warnings, the department choose to maintain clinical follow-up of women submitted to POP repair with a Prolift transvaginal mesh (Prolift™).
Based on what is stated above, the aim of this study will be to report long-term outcomes after POP repair with a Prolift™ kit.
Methods
It was performed a cross-sectional, retrospective and analytical study of 54 women surgically treated with Prolift™, at Urogynecology department of Centro Hospitalar de Leiria, between January 2009 to January 2017. The last Gynecare Prolift mesh was applied at January 2014.
The patients were selected to a mesh repair based on individual risk factors for surgery, on the POP grade (equal or higher than 2) and on the assumption of low risk of relapse. In order to identify an occult stress urinary incontinence, besides history and physical examination all women proposed to POP surgical treatment were preoperatively evaluated by urodynamic tests.
All women involved were enlightened about the different surgical techniques, the rates of success and potential risks. All signed a written informed consent.
This study is in accordance with the Declaration of Helsinki, as revised in 2013.
The following variables were analyzed: age at surgery, body mass index (BMI-kg/m2), parity, menopause, comorbidities (i.e. arterial hypertension, constipation), previous surgeries, urinary incontinence symptoms, anatomical classification of prolapse (according to vaginal walls), POP grade (by POPQ system), surgical procedures (anterior, posterior or total mesh repair), acute and late complications (reported by the Prosthesis Complication Classification Code designed by the International Continence Society/International Urogynecological Association (ICS/IUGA)), anatomic failure (defined as recurrent pelvic organ prolapse grade≥2, at the same compartment) and time of follow-up.
Statistical analysis was performed using the STATA program (version 13.1), with a statistically significant result if p£0.05 and a 95% confidence level. To evaluate the relationship between nominal variables, Fisher's exact or chi-square test were used. To compare measures of central tendency were used student's t-test or Wilcoxon's test (according to the presence or absence of normal distribution). The adjustment of confounding factors was done through the application of a logistic regression. Time to event (relapse or complications) was analysed by Kaplan-Meier curves and comparison between procedures by log rank test. Hazard ratios were obtained by cox proportional regression models.
Results
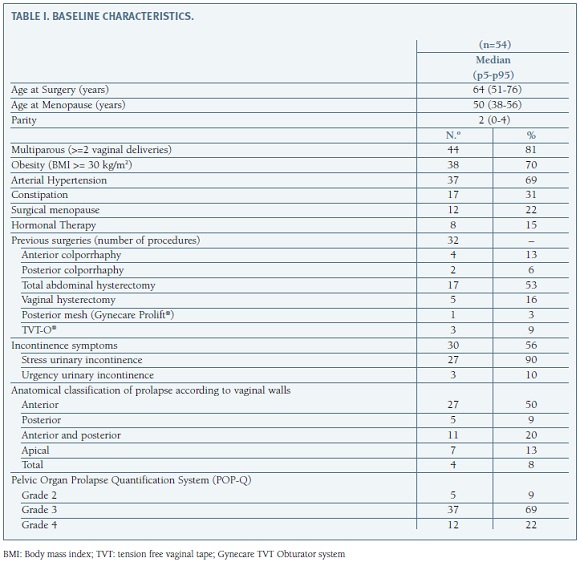
Fifty-four menopausal women were submitted to POP repair through a transvaginal placement of a synthetic mesh (Ethicon Gynecare Prolift). The median age at surgery was 64 years (range:51-76), 81% (n=44) were multiparous (more than two vaginal deliveries), 70% (n=38) were obese (BMI >= 30 kg/m2) and 31% (n=17) referred constipation (no bowel movements at least during three days). Twenty three (59%) woman had already been submitted to gynecological surgeries. Twenty two to hysterectomy, from that, two to mid-urethral sling (Gynecare TVT Obturator system: TVT-O), two to anterior colporrhaphy and other two to posterior and anterior colporrhaphy, all at the same surgical time. One woman was submitted to posterior POP repair by a posterior Prolift and to stress urinary incontinence repair by a TVT Obturator system. In other words, five women were previous submitted to POP repair (two: anterior colporrhaphy; two: anterior and posterior colporrhaphy and one to posterior Prolift). All women previously submitted to anterior or posterior colporrhaphy had POP relapse at the same compartment. The one submitted to a posterior mesh repair did not suffer recurrence on the operated area. Fifty six percent (n=30) had urinary incontinence symptoms, 90% of that stress urinary incontinence. (Table I).
The commonest prolapse was identified at the anterior vaginal compartment (it was identified in 42 women, alone or in association with prolapses at other compartments). Thirty-seven patients (69%) had a grade 3 prolapse and twelve a grade 4 (22%) (Table I).
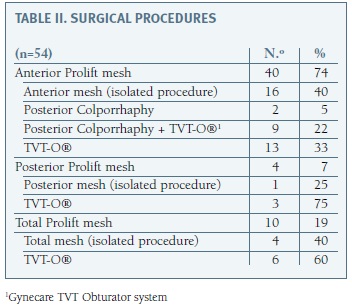
Total mesh was used in 10 patients (19%), an isolated anterior mesh in 40 (74%) and an isolated posterior mesh in 4 patients (7%) (Table II).
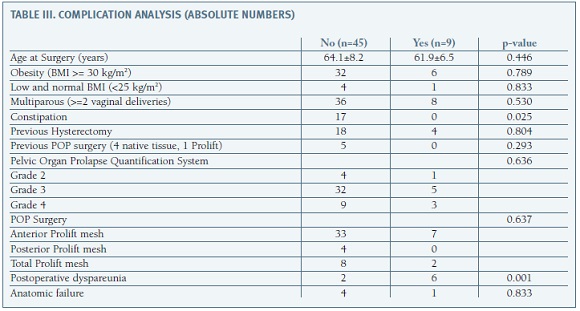
There were no intraoperative complications identified like bladder injury or heavy hemorrhage (bleeding equal or higher than 250mL). Postoperatively, it was identified 17% (n=9) complications, four prominences, four exposures and one contraction. According to Category (C), Time (T) and Site (S) Classification, four patients were classified as T1 (two: CA1a and two: CB1b), three as T2 (three: CB2b), one as T3 (CB3b) and one T4 (CA2a), all S1. One mesh exposure was successfully managed with mesh resection under anesthesia, four months after POP surgery, the other three partial mesh exposures occurred at a median time of 60 months after surgery (range: 60-70 months). The threads exposed were resected at medical office. The other complications did not need surgical intervention.
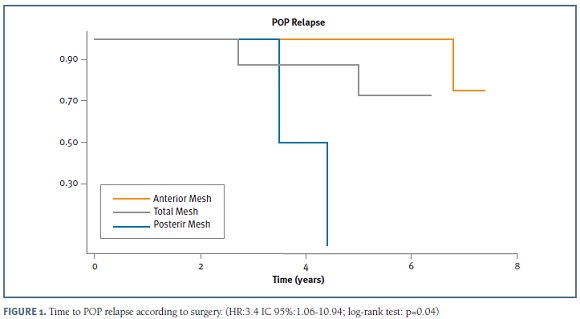
It was identified an anatomic failure rate of 9% (n=5) occurring after a median time of four years (range: 3-7 years). Thus, we report an overall success of 91% (Figure 1).
Six patients (11%) presented de novo prolapse in untreated vaginal compartments and six (11%) de novo urinary incontinence symptoms (three (5.5%) stress and three (5.5%) urge incontinence). Twenty-three patients had pre-operative sexual activity (43%) and the de novo dyspareunia rate was 11%.
Until now, 69% (n = 37) of patients are under surveillance, 24% (n = 13) abandoned the medical department and 7% (n = 4) died for reasons not related to POP surgery. The median time of follow up was four years (range: 1-7years). For those who die the median time of follow up was three years (range: 1- 6 years).
We found statistical association between grade of POP and number of vaginal deliveries (p=0.05), even after adjusting for age, comorbidities and previous surgeries. It was not identified association among age, BMI, POP grade, comorbidities and type of POP mesh repair and development of complications (p=0.801). There was relation between complications and complain of dyspareunia (p=0.001). It was not identified relation among complications and failure (p=0.833). (Table III) Through unadjusted study, it was identified statistical relation between grade of POP and relapse (p=0.05). Although, after adjusting for age, type of mesh repair, BMI and comorbidities, that association was lost (p=0.120). There was significant association between age and failure (HR: 1.16; IC 95%:1.01-1.34; p=0.04), association that was dissipated after adjusting for comorbidities and type of POP mesh repair (p=0.670). Even after adjusting for age, comorbidities, previous surgeries and grade of POP, it was identified statistical association within relapse and type of mesh repair (HR:3.4; IC 95%:1.06-10.94; p=0.04). The relapses were more frequent and earlier among total Prolift mesh procedures. (Figure 1) We do not find statistical association between POP surgery and de novo POP in the non-operated compartment (p=0.681). It was also no relation between type of mesh repair and de novo complains of incontinence (p=0.833).
Discussion
Through this retrospective analysis we found an overall success of 91%, once we identified five relapses at the same vaginal compartment (anatomic failure=9%). Multiple studies are in agreement with our study. Feiner et al reviewed POP mesh repairs done on 2653 women and found an objective overall success of 87% for the Prolift system12. The 2016 Cochrane Review compiled data from 37 randomized controlled trials (4023 women) of transvaginal polypropylene mesh compared to native tissue repair and demonstrated, through low to moderate quality evidence, that there are advantages to use transvaginal permanent mesh, including lower rates of awareness of prolapse, repeat surgery for prolapse, and recurrent prolapse on examination. The evidence suggests that if 19% of women are aware of prolapse after native tissue repair, between 10% and 15% will be aware of prolapse after permanent mesh repair. If the rate of recurrent prolapse on examination after a native tissue repair is assumed to be 38%, the risk would be between 11% and 20% after a repair with transvaginal permanent mesh2. Other author's advocates that the advantage is restricted to one compartment. At one study, the Elevate® transvaginal mesh kit (n=100) was compared to a native tissue repair cohort (n=100), and it was identified significantly higher success for mesh at the anterior compartment (98 vs. 87%, p=0.006), without difference in outcome at the apical (99 vs. 96%, p=0.317) or posterior compartments (100 vs. 97%, p=0.567)13. Some studies reported different outcomes, an example is PROSPECT study, a multicenter, randomized controlled study, which include 1352 woman, and assesses benefits and possible harm of primary anterior or posterior transvaginal repair enhanced by mesh or graft against standard native tissue repair. This has shown that, in the first two years after surgery, women do not benefit from having their first anterior or posterior prolapse repair reinforced with synthetic mesh or biological graft either in terms of prolapse symptoms or anatomical cure6.
In spite of these major differences, there are some unanimity when the concern are the complications. We found a rate of complications of 17% (n=9), 67% related with mild symptoms and all characterized by a simple resolution (only one needed anesthesia). Research and media publications have demonstrated the frequent occurrence of serious complications, arguing no differences in spite of mesh type or surgical technique12. In 2008 FDA issued a first warning and in 2011 update the previous communication, reporting higher rate of adverse events and stating absence of clear evidence that mesh use improves clinical outcomes in comparison to traditional nonmesh repair. This led to a reappraisal of the role of transvaginal mesh in POP surgery. In this context, it was done an evaluation about the trends in the surgical management of POP in Portugal. All records of women diagnosed with genital prolapse at public hospitals, from 1 January 2000 to 31 December 2012, were included. They identified an increase of POP diagnosis of 105%, from 2000 to 2012 (2368 to 4941 cases). It was observed that the first registration of POP mesh repair was at 2007, since that it was recorded 1468 POP cases. The use of vaginal meshes was low compared with the total number of anterior/posterior repairs (10% of all cystocele, 7% rectocele, 3% cystocele plus rectocele). After the 2011 FDA communication, the author's verified that, against all expectations, the use of transvaginal mesh has increased14. In this document there was no references about complications or relapses.
Currently, over 30 000 cases due to mesh-related complications and lawsuits are brought to courts. Reacting to this, several products have been withdrawn from the market by the manufacturers15. Gynecare Prolift, was one of the systems removed from the American market by Johnson & Johnson, in June 2012. However, the sales persisted at other countries, which justifies the presence of studies concerning the Prolift system, after that period of time. Kasyan et al analyzed the biggest series of 152 complications (22.5%) following Prolift transvaginal mesh for POP. The most frequent complications were erosions (21%), followed by dyspareunia (11%), mesh shrinkage (4.4%), pelvic abscess (2.7%), and fistula (1.3%). They also reported that the higher risk for the development of complications was a younger age, less prominent prolapse, development of hematomas, and concomitant hysterectomies15. El-Khawand et al conducted a retrospective cohort study with the aim of identifying risk factors for mesh exposure after polypropylene transvaginal mesh placement, and concluded that BMI and concurrent total hysterectomy were significantly associated with mesh exposures. They concluded that the lower the BMI, the higher the mesh exposure rate, which was 0.8% with no hysterectomy and 23.5% when a concurrent hysterectomy was performed12. At our study only constipation was identified as a potential risk factor for the development of complications. (Table III) Concerning complications management, Hampel states that erosions with consecutive infection, pain and dyspareunia, often required surgical revision. Current studies report that the postoperative POP mesh removal is about 3%15.
At literature is also argued that there is an increased risk of de novo urinary incontinence, once the operation inherently alters the position of the urethra and bladder. The risk has been estimated to occur in 22 to 41% of patients16. At our study we identified 11% of de novo urinary incontinence symptoms (three (5.5%) stress and three (5.5%) urge incontinence).
Withagen et al, after a secondary analysis also mentioned that mesh-augmented prolapse repair in only one vaginal compartment is associated with a higher de novo prolapse rate in the untreated compartments compared with conventional vaginal native tissue repair in women with recurrent pelvic organ prolapse (47% versus 17%)17. We do not find statistical association between POP surgery and de novo POP in the non-operated compartment.
According to Scientific Committee on Emerging and Newly Identified Health Risks (SCENIHR), the clinical outcome following mesh implantation is influenced by several aspects such as material properties, product design, overall mesh size, route of implantation, patient characteristics, associated procedures (e.g. hysterectomy) and the surgeon's experience. So, SCENIHR advise that all these aspects should be evaluated previous to surgery, adding that the implantation of any mesh for the treatment of POP via the vaginal route should be only considered in complex cases in particular after failed primary repair surgery18.
Based on what was exposed, we identify as strengths of our study its clinical relevance, the long-time of follow up, the inexpensive execution and the statistical study, which took into account the presence of potential confounding factors. Although it also has some limitations, namely the retrospective analysis, which is endowed with a lower level of evidence and greater susceptibility to information or selection bias, and the number of cases, which limits the extrapolation of results. It was also not used standardized questionnaires to access patient's symptoms and quality of life and the considerable number of patients that were lost to follow-up (17 patients: four died for reasons not related to POP and 13 abandoned the medical department).
In conclusion, after a long period of follow-up we could demonstrated that the Prolift procedure was successful and that it was mostly related with the development of mild complications.
BIBLIOGRAPHY
1. Rogers R., Fashokun T. Pelvic organ prolapse in women: Epidemiology, risk factors, clinical manifestations, and management [Internet]. Uptodate.com. 2017 Available at: http://www.uptodate.com (cited 27 August 2017). [ Links ]
2. Maher C, Feiner B, Baessler K, Christmann-Schmid C, Haya N, Marjoribanks J Transvaginal mesh or grafts compared with native tissue repair for vaginal prolapse. Cochrane Database Syst Rev 2016. [ Links ]
3. Khan Z., Thomas L., Emery S. Outcomes and complications of trans-vaginal mesh repair using the ProliftTM kit for pelvic organ prolapse at 4 years median follow-up in a tertiary referral center. Arch Gynecol Obstet. 2014. 290:1151-1157. [ Links ]
4. Persu C, Chapple CR., Cauni V., Gutue S., Geavlete P. Pelvic Organ Prolapse Quantification System (POP-Q) - a new era in pelvic prolapse staging. J Med Life. 2011. 4:75-81. [ Links ]
5. Farthmann J., Watermann D., Zamperoni H., Wolf C., Fink T., Gabriel B. Pelvic organ prolapse surgery in elderly patients. Arch Gynecol Obstet. 2017. 295: 1421-1425. [ Links ]
6. Reisenauer C, Koelbl H. Three studies in the focus of pelvic organ prolapse research Arch Gynecol Obstet. 2017. 296:139-141. [ Links ]
7. Eric Jelovsek J. Pelvic organ prolapse in women: Choosing a primary surgical procedure. [Internet]. Uptodate.com. 2017 Available at: http://www.uptodate.com (cited 27 August 2017). [ Links ]
8. Chapple CR, Cruz F, Deffieux X, Milani AL, Arlandis S, Artibani W, et al. Consensus statement of the European Urology Association and the European Urogynecological Association on the use of implanted materials for treating pelvic organ prolapse and stress urinary incontinence. Eur Urol. 2017. 72:424-431. [ Links ]
9. Bontje HF, G. Van de Pol, Van der Zaag-Loonen H, Spaans W. Follow-up of mesh complications using the IUGA/ICS category-time-site coding classification Int Urogynecol J. 2014. 25:817-822
10. U.S. Food and Drug Administration. Concerns about Surgical Mesh for POP [Internet]. Available at: www.fda.gov (cited 26 August 2017). [ Links ]
11. Trabuco E, Gebhart J. Overview of transvaginal placement of mesh for prolapse and stress urinary incontinence. [Internet]. Uptodate.com. 2017 Available at: http://www.uptodate.com (cited 27 August 2017). [ Links ]
12. Iver S, Botros S. Transvaginal mesh: a historical review and update of the current state of affairs in the United States. Int Urogynecol J. 2016. 28:527-535. [ Links ]
13. Gaines N, Gupta P, Sirls L. Pelvic Prolapse Repair in the Era of Mesh. Curr Urol Rep. 2016. 17:20. [ Links ]
14. Mascarenhas T, Mascarenhas Saraiva M, Ricon-Ferraz A, Nogueira P, Lopes F, Freitas A. Pelvic organ prolapse surgical management in Portugal and FDA safety communication have an impact on vaginal mesh. Int Urogynecol J. 2014. 26: 113-122. [ Links ]
15. Barski D, Deng D. Management of Mesh Complications after SUI and POP Repair: Review and Analysis of the Current Literature. Biomed Res Int. 2015: 831285.
16. Altman D, Mikkola T, Bek KM, Rahkola-Soisalo P, Gunnarsson J, Engh ME et al. Pelvic organ prolapse repair using the UpholdTM Vaginal Support System: a 1-year multicenter study. Int Urogynecol J. 2016. 27: 1337-1345. [ Links ]
17. Withagen M, Milani A, Leeuw J, Vierhout M. Development of de novo prolapse in untreated vaginal compartments after prolapse repair with and without mesh: a secondary analysis of a randomised controlled trial. BJOG 2012. 119:354-360. [ Links ]
18. SCENIHR (Scientific Committee on Emerging and Newly Identified Health Risks). The safety of surgical meshes used in urogynecological surgery. 2015. [ Links ]
Endereço para correspondência | Dirección para correspondencia | Correspondence
Fernanda Santos,
Centro Hospitalar Leiria EPE
Leiria, Portugal
E-mail: fernandapatricia.santos@yahoo.com
Recebido em: 31/08/2017
Aceite para publicação: 10/04/2018