Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Medicina Interna
versão impressa ISSN 0872-671X
Medicina Interna vol.25 no.1 Lisboa mar. 2018
https://doi.org/10.24950/rspmi/original/184/1/2018
ARTIGOS ORIGINAIS / ORIGINAL ARTICLES
Vitamin D Metabolism and Pain in Rheumatoid Arthritis
Metabolismo da Vitamina D e Dor na Artrite Reumatóide
Filipe Andrade1, Albina Moreira1, Rui Môço1, Rosário Alves2, Alexandre Vasconcelos1, Eduardo Eiras1, Carlos Soares2, Anabela Ferreira1
1Department of Medicine, Hospital Pedro Hispano, Matosinhos, Portugal
2Department of Clinical Pathology, Hospital Pedro Hispano, Matosinhos, Portugal
ABSTRACT
Introduction: Vitamin D deficiency has been linked to increased susceptibility to the development of rheumatoid arthritis and with disease activity. The objective of this study was to evaluate vitamin D status in patients with RA and to assess the relationship between vitamin D levels and pain.
Methods: In a cohort of 45 patients with RA, 25-hydroxyvitamin D3 [25(OH)D3] levels, parathyroid hormone, C-reactive protein (CRP) and albumin levels were measured. Disease activity was evaluated by calculating the 28-joint Disease Activity Score (DAS28).
Results: Forty five patients were included in the study. Thirty one patients were in remission, 11 patients had painful joints and the median VAS pain score was 30. Out of the 45 patients, 41 had vitamin D levels below 30 ng/mL. Levels of 25(OH)D3 were not found to be correlated to the DAS28 score (p = 0.59), the VAS pain score (p = 0.55) or CRP (p = 0.16). Parathyroid hormone levels were positively correlated to CRP (p = 0.01).
Conclusion: In our study, it appears that vitamin D deficiency is highly prevalent in patients with RA but it is not linked to disease severity or pain experienced in RA. PTH levels are related to CRP and but not to the number of painful joints or VAS pain score. This relationship may reflect uncontrolled disease or be a reflection of physiological vitamin deficiency. Vitamin D supplementation may be needed both for the prevention of osteoporosis as well as for better disease control in patients with RA and low levels of vitamin D and high CRP.
Keywords: Arthritis, Rheumatoid; Pain; Parathyroid Hormone; Vitamin D; Vitamin D Deficiency.
RESUMO
Introdução: A deficiência de vitamina D tem sido associada ao desenvolvimento e actividade da artrite reumatóide (AR). O objetivo deste estudo foi avaliar os níveis de vitamina D em doentes com AR e avaliar a sua relação com a dor.
Métodos: Numa coorte de 45 doentes com AR, foram medidos níveis de 25-hidroxivitamina D3 [25 (OH) D3], hormona paratiróide (PTH), proteína C-reativa (CRP) e albumina. A atividade da doença foi avaliada pelo cálculo do 28-joint Disease Activity Score (DAS28).
Resultados: Dos doentes 45 foram incluídos no estudo. Dos doentes 31 estavam em remissão, 11 doentes apresentavam articulações dolorosas, a mediana de dor na escala visual analógica (EVA) foi de 30. Dos 45 doentes, 41 tinham níveis de vitamina D inferiores a 30 ng / mL. Os níveis de vitamina D não estavam associados ao DAS28 (p = 0,59), EVA (p = 0,55) ou CRP (p = 0,16). Os níveis de PTH estavam associados positivamente com a CRP (p = 0,01).
Conclusão: No nosso estudo, a deficiência de vitamina D é altamente prevalente em doentes com AR, mas não está associada à atividade da doença ou à dor. Foi encontrada uma associação entre os níveis de PTH e elevação da CRP, esta relação pode refletir doença não controlada ou ser um reflexo da deficiência fisiológica de vitamina D. A suplementação de vitamina D pode ser necessária tanto para a prevenção da osteoporose quanto para o melhor controlo da doença em doentes com AR e baixos níveis de vitamina D e elevação da CRP.
Palavras-chave:Artrite Reumatóide; Deficiência de Vitamina D; Dor; Hormona Paratiróide; Vitamina D.
Introduction
Vitamin D is a secosteroid hormone involved in bone metabolism and the regulation of calcium homeostasis.1 The hormone is synthesized in the skin by the action of ultraviolet irradiation. Overt vitamin D deficiency, which is characterized by hypocalcemia and/or hypophosphatemia and rickets in children and osteomalacia in adults, is now uncommon in most developed countries. However, subclinical vitamin D deficiency occurs even in developed countries and is associated with osteoporosis, increased risk of falls, and possibly fractures.2
In recent years numerous clinical research articles have concluded that large proportions of North American and global populations are deficient in vitamin D.1 The utility of measurement of parathyroid hormone (PTH) concentrations for identifying the optimal level of vitamin D remains controversial; the relationship between serum 25-hydroxyvitamin D3 levels and PTH is inconsistent, and no clear threshold defining sufficiency has been established.3
Rheumatoid arthritis (RA) is an autoimmune disease of unknown etiology, and vitamin D deficiency may increase the risk for the development of RA.4 The role of vitamin D deficiency in the pathogenesis of RA, as well as the relationship between vitamin D deficiency and the activity of RA is unclear.5 RA is an inflammatory disease characterized by flares and remissions, flares are characterized by pain. Vitamin D deficiency is also known to be associated with diffuse musculoskeletal pain.2 Osteomalacia is thought to be the causative factor for the primary pain mechanism in Vitamin D deficiency. The mechanism involves hydration of the demineralized gelatin matrix beneath the periosteum; the hydrated matrix pushes outward on the periosteum, causing throbbing, aching pain. Osteomalacia can often be diagnosed by using moderate force to press the thumb on the sternum or anterior tibia, which can elicit bone pain. One study showed that 93% of persons 10 to 65 years of age who were admitted to a hospital emergency department with muscle aches and bone pain and who had a wide variety of diagnosis, including fibromyalgia, chronic fatigue syndrome, and depression, were deficient in vitamin D.3
The objective of this study was to evaluate the prevalence of vitamin D deficiency in patients with RA and to find an association with pain control and acute phase inflammatory markers.
Methods
This prospective study was approved by our local Institutional Review Board (nr 137/13/OP) and informed consent was obtained from all patients. Between May 2013 and May 2014, all patients followed in our autoimmune outpatient clinic were screened for inclusion. The study cohort consisted of 45 patients with RA. The patients entered the study as they came for evaluation at the autoimmune outpatient clinic.
Study investigators collected subject data including demographic information and standard clinical data with a structured database. 25-hydroxyvitamin D3 [25(OH)D3] levels, parathyroid hormone levels, C-reactive protein (CRP) and albumin levels were measured in a standardized hospital laboratory.
All patients fulfilled the 2010 American College of Rheumatology/European League Against Rheumatism RA classification criteria.6 Disease activity was evaluated by calculating the 28-joint Disease Activity Score (DAS28). Additionally, the visual analogue scale (VAS) pain score were calculated.
The 25(OH)D3 levels were measured by a chemiluminescent microparticle immunoassay using a quantitative delayed one-step competitive procedure. First, sample, assay diluent and paramagnetic anti-vitamin D coated microparticles are combined. 25-OH vitamin D present in the sample is displaced from the vitamin D binding protein and binds to anti-vitamin D coated microparticles, forming an antigen-antibody complex. After incubation, a conjugate containing acridinium-labeled vitamin D is added to the reaction mixture and binds to unoccupied binding sites of the anti-vitamin D coated microparticles. After further incubation and washing, Pre-Trigger and Trigger Solutions are added to the reaction mixture. The resulting chemiluminescent reaction is measured as relative light units and detected by the optic system. Results are calculated automatically based on the previously established calibration curve. The measuring interval of the assay is 3.4 to 155.9 ng/mL (8.5 to 389.8 nmol/L). The sensitivity was 1.6 ng/mL and the within and between batch precision was less than 10%. Parathyroid hormone was measured by a two-step sandwich immunoassay using chemiluminescent microparticle technology. The sample, assay diluent, and anti-PTH coated paramagnetic microparticles are combined. Intact PTH present in the sample binds to the anti-PTH coated microparticles. After washing, anti-PTH acridinium-labeled conjugate is added to create a reaction mixture. Following another wash cycle, Pre-Trigger and Trigger Solutions are added to the reaction mixture. The resulting chemiluminescent reaction is measured as relative light units and detected by the optic system. The measuring interval of the assay is 3.0 to 3000.0 pg/mL. The functional sensitivity was determined to be = 5 pg/mL and the within batch precision was less than 9%. The Albumin BCG procedure is based on the binding of bromcresol green specifically with albumin to produce a colored complex. The absorbance of the complex at 628 nm is directly proportional to the albumin concentration in the sample and the measuring interval of Albumin BCG is 0.4 to 10.5 g/ dL. The PCR procedure was based in an immunoturbidimetric determination with a sensitivity of 0.1 mg/dL and a measuring interval of 0.2 to 480 mg/L.
Vitamin D deficiency was defined as a 25-hydroxyvitamin D level of less than 20 ng per milliliter and Vitamin D insufficiency was defined as a 25-hydroxyvitamin D level between 21 and 30 ng per milliliter. PTH level was defined as normal between 10 and 65 pg/mL. Death or osteoporotic fractures were ascertained by review of electronic medical records 36 months after inclusion.
STATISTICAL ANALYSIS
Continuous variables are presented as median (interquartile range), and categorical variables as absolute and relative frequencies. Comparisons between groups of quantitative and qualitative variables were performed using Pearson test, Spearman test and Mann–Whitney tests. Before variance analysis, tests of normality and homogeneity of variance were applied. Mann–Whitney U tests were used if the normality and homogeneity of variance assumptions were not satisfied. Data handling and analysis were performed with the Statistical Package for Social Sciences version 24 (SPSS Inc., Chicago, IL).
Results
Data are presented as median (interquartile range) or frequency (%). Forty-five patients were included in the study. The characteristics of the 45 included patients are summarized in Table 1. The sample was mostly female (68.8%) with a median age of 60 (55 - 70) years old.
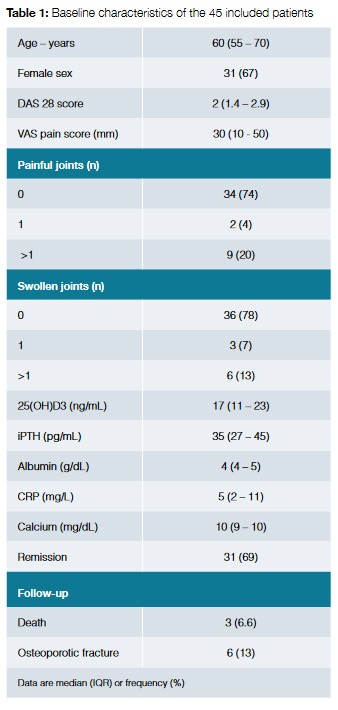
Briefly, the number of patients in remission was 31 (68.8%). Eleven patients (24.4%) had painful joints and median VAS pain score was 30 (10 – 50).
Concerning 25(OH)D3 levels out of the 45 patients, 8 (17.7%) met the criteria for deficiency, 33 (73%) met the criteria for insufficiency and four (9%) patients had normal 25(OH) D3 level. Four patients had elevated PTH levels.
In our sample levels of 25(OH) D3 were not found to be correlated to the DAS28 score (r = -0.083, n = 44, p = 0.59), the VAS pain score (r = 0.096, n = 42, p = 0.55) or CRP (r = -0.214, n = 43, p = 0.16). Splitting the sample into two groups depending on remission and non-remission and rerunning the analysis for each group results followed the same pattern, and no significant correlations between the variables and 25(OH)D3 level were found.
Parathyroid hormone levels, on the contrary were found to be correlated to CRP (r = -0.385, n = 44, p = 0.01) but not to the to the DAS28 score (r = 0.030, n = 45, p = 0,84) or the VAS pain score (r = -0.153, n = 43, p = 0.33). A Mann-Whitney test indicated that the parathyroid hormone levels werent significantly greater in those with one or more painful joint (mean rank = 26.7) than for those with no painful joint (mean rank = 21.8), U = 146.5, p = 0.285. Moreover, when splitting the sample into two groups depending on remission and non-remission, a Mann-Whitney test indicated similar results.
Albumin levels were found to be correlated with the VAS pain score (r = -0.385, n = 43, p = 0.01). A Mann-Whitney test indicated that albumin levels were significantly greater in those with remission (mean rank = 26.6) than in those with no remission (mean rank = 14.5), U = 104.5, p = 0.006. Albumin level was also found to be significantly greater in those with no painful joints (mean rank = 26.2) than in those with one or more painful joints (mean rank = 13.1), U = 78.5, p = 0.003.
After a mean follow-up of 36 months there were three casualties (lung adenocarcinoma, cytomegalovirus enterocolitis and one septic arthritis) and six osteoporotic related fractures. A Spearmans correlation was run to assess the relationship between levels of 25(OH)D3 and death or osteoporotic fractures. There was no correlation found to be statistically significant, rs = -0.181, p = 0.239 and rs = -0.130, p = 0.399 respectively. Osteoporotic fractures were associated with RA disease activity as assessed by the DAS28 activity score (rs = 0.332, p = 0.003).
Dicussion
Our findings suggest hypovitaminosis D is common in RA patients. Over 90% had vitamin D levels below the currently accepted threshold of sufficiency (i.e. 30 ng/mL). In spite of the high number of patients with low vitamin D levels we only found 4 patients with hyperparathyroidism, this can identifies a distinct group of patients with hypovitaminosis D and a blunted PTH response as reported by Sahota et al.7 Others have also reported high prevalence rates of vitamin D deficiency in RA and in a recent meta-analysis, Lee et al reported a significantly higher prevalence of vitamin D deficiency in the RA group than in the control group (55.2% vs 33.2%).8
Contrary to other reports no significant associations were evident between vitamin D levels and disease activity scores or pain. This may be related to the very high prevalence of vitamin D deficiency and insufficiency present in our sample (91.1%).8,9
Concerning albumin levels, we observed a strong negative association with tender joint count and VAS pain score.
In contrast to Oelzner et al10 that reported a negative correlation in RA patients between disease activity and parathyroid hormone levels, we have not found such an association. Instead, we found a positive association between parathyroid hormone level and CRP levels. Interestingly, this association was also present in patients in remission, which can represent subclinical disease activity. Parathyroid hormone stimulates interleukin-6 (IL-6) production by osteoblasts and liver cells.11,12 In turn, IL-6 may modulate acute-phase protein synthesis in the liver.13 This relationship may reflect uncontrolled disease or reflect physiological vitamin D deficiency.
These results may have therapeutic implications, vitamin D supplementation may be needed for the prevention of osteoporosis and better disease control in RA patients with low levels of D3 [25(OH)D3] and high CRP despite remission as assessed by the DAS28 score.
The Womens Health Initiative study showed that serum levels of 25-hydroxyvitamin D had little effect on the risk of fracture when levels were 26 ng per milliliter (65 nmoL per liter) or less. However, women who were taking calcium and vitamin D3 had a 29.0% reduction in hip fracture.3,14 In our study after a mean follow-up of 36 months, low levels of D3 [25(OH) D3] were not associated to mortality or osteoporotic related fractures. One possible explanation for this can be the lack of information related to the evolution of vitamin D status during the follow-up period, namely the vitamin D supplementation done during this period.
There were several limitations in our study. This is a small study, and certainly future studies may be warranted to confirm our results in larger, more representative populations. Additional information including use of vitamin D agonists, associations with BMI and osteoporosis would have been desirable.
In conclusion, vitamin D deficiency appears to be common in RA even in a southern European country. Parathyroid hormone levels appear to be associated with inflammation, and either the parathyroid hormone levels directly impact RA symptoms and disability, or the reverse may be true. Larger prospective cohort and interventional studies are required to explore these relationships. As vitamin D deficiency is easily corrected in most individuals, clinicians should consider screening RA patients to identify and address suboptimal vitamin D levels and elevated Parathyroid hormone levels.
Acknowledgments
We thank Vitor Santos for his help in all the laboratory work.
References
1. Holick MF. Vitamin D: evolutionary, physiological and health perspectives.Curr Drug Targets. 2011;12:4-18. [ Links ]
2. Hirani V, Blyth FM, Naganathan V, Cumming RG, Le Couteur DG, Handelsman DJ, et al. Active vitamin D (1,25 dihydroxyvitamin D) is associated with chronic pain in older australian men: the Concord Health and Ageing in Men Project. J Gerontol Ser A Biol Sci Med Sci. 2015;70:387-95. [ Links ]
3. Holick MF. Vitamin D deficiency. N Engl J Med. 2007;357:266-281. [ Links ]
4. Merlino LA, Curtis J, Mikuls TR, Cerhan JR, Criswell LA, Saag KG, et al. Vitamin D intake is inversely associated with rheumatoid arthritis: Results from the Iowa Women’s Health Study. Arthritis Rheum. 2004;50:72-7.
5. Matsumoto Y, Sugioka Y, Tada M, Okano T, Mamoto K, Inui K, et al. Relationships between serum 25-hydroxycalciferol, vitamin D intake and disease activity in patients with rheumatoid arthritis –TOMORROW study. Mod Rheumatol. 2015;25:246-50.
6. Aletaha D, Neogi T, Silman AJ, Funovits J, Felson DT, Bingham CO 3rd, et al. 2010 Rheumatoid arthritis classification criteria: An American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 2010;62:2569-81. [ Links ]
7. Sahota O, Mundey M, San P, Godber I, Lawson N, Hosking D. The relationship between vitamin D and parathyroid hormone: calcium homeostasis, bone turnover, and bone mineral density in postmenopausal women with established osteoporosis. Bone. 2004;35:312-9. [ Links ]
8. Lee YH, Bae S-C. Vitamin D level in rheumatoid arthritis and its correlation with the disease activity: a meta-analysis. Clin Exp Rheumatol. 2016;34:827-33. [ Links ]
9. Haque UJ, Bartlett SJ. Relationships among vitamin D, disease activity, pain and disability in rheumatoid arthritis. Clin Exp Rheumatol. 2010;28:745-7. [ Links ]
10. Oelzner P, Müller A, Deschner F, Hüller M, Abendroth K, Hein G, et al. Relationship between disease activity and serum levels of vitamin D metabolites and pth in rheumatoid arthritis. Calcif Tissue Int. 1998;62:193-8. [ Links ]
11. Cheng SP, Liu CL, Liu TP, Hsu YC, Lee JJ. Association between parathyroid hormone levels and inflammatory markers among US adults. Mediators Inflamm. 2014;2014:709024. [ Links ]
12. Mitnick MA, Grey A, Masiukiewicz U, Bartkiewicz M, Rios-Velez L, Friedman S, et al. Parathyroid hormone induces hepatic production of bioactive interleukin-6 and its soluble receptor. Am J Physiol Endocrinol Metab. 2001;280:E405-12. [ Links ]
13. Castell JV, Gómez-Lechón MJ, David M, Fabra R, Trullenque R, Heinrich PC. Acute-phase response of human hepatocytes: regulation of acute-phase protein synthesis by interleukin-6. Hepatology. 1990;12:1179-86. [ Links ]
14. Jackson RD, LaCroix AZ, Gass M, Wallace RB, Robbins J, Lewis CE, et al. Calcium plus vitamin D supplementation and the risk of fractures. N Engl J Med. 2006;354:669-83. [ Links ]
Correspondência/Correspondence: Filipe Andrade filipeteixeiraandrade@gmail.com
Department of Medicine, Hospital Pedro Hispano, Matosinhos, Portugal
Rua Dr. Eduardo Torres, 4464-513 Senhora da Hora, Senhora da Hora
Conflicts of interest: The authors have no conflicts of interest to declare.
Conflitos de Interesse: Os autores declaram a inexistência de conflitos de interesse na realização do presente trabalho.
Financing Support: This work has not received any contribution, grant or scholarship.
Fontes de Financiamento: Não existiram fontes externas de financiamento para a realização deste artigo.
Confidentiality of data: The authors declare that they have followed the protocols of their work center on the publication of data from patients.
Direito à Privacidade e Consentimento Informado: Os autores declaram que nenhum dado que permita a identificação do doente aparece neste artigo.
Protection of human and animal subjects: The authors declare that the procedures followed were in accordance with the regulations of the relevant clinical research ethics committee and with those of the Code of Ethics of the World Medical Association (Declaration of Helsinki).
Proteção de Seres Humanos e Animais: Os autores declaram que não foram realizadas experiências em seres humanos ou animais.
Recebido: 14/09/2017
Aceite: 02/11/2017














