Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Portugaliae Electrochimica Acta
versão impressa ISSN 0872-1904
Port. Electrochim. Acta vol.38 no.5 Coimbra jun. 2020
https://doi.org/10.4152/pea.202005343
ARTIGOS
Corrosion Inhibition of Copper by Thioureas and N, O, S-Ligating Ring Compounds
P. Chooto*, W. Aemaek Tappachaib, S. Duangthonga and S. Manaboota
aDepartment of Chemistry, Analytical Chemistry Division, Faculty of Science, Prince of Songkla University, Hatyai, Songkhla 90112, Thailand
bDepartment of Chemistry, Inorganic Chemistry Division, Faculty of Science, Prince of Songkla University, Hatyai, Songkhla 90112, Thailand
ABSTRACT
Certain N, O, S-ligating ring compounds and thioureas were investigated to understand their role of copper corrosion inhibition in acetonitrile. For 5 quinones under study, including xanthone, xanthene, thioxanthone, acridone and 1,4-naphthoquinone, acridone was the best inhibitor, with Cu corrosion rate of 4.495 × 10-4 mm/year, whereas 1,4-naphthoquinone exhibited the lowest inhibition, due to a lower number of nitrogen groups. With the presence of sulphur, to form a stronger bond with Cu, thioureas had better inhibiting behavior than quinones. Of 4 thioureas, namely thiourea (tu), diphenylthiourea (dptu), phenylthiourea (ptu), and ethylenethiourea (etu), the fourth shows the highest inhibition – with Cu corrosion rate of 2.27 × 10-4 mm/year – and the third shows the lowest one, due to the steric effect from the phenyl group. When halide ions are present, the inhibition efficiency of thioureas decreases, due to more preferable Cu complexation to halides; the strongest copper-halide bond formation occurred by the freest iodide ion, which is consistent with the results from X-ray crystallography and electrochemistry.
Keywords: Copper, corrosion inhibition, organic inhibitor, electrochemistry.
Introduction
Copper has long been used in a number of areas, especially as a structural component, due to its advantageous properties, especially in marine systems (1-2). However, since the presence of Cl- makes it vulnerable to corrosion, corrosion inhibition is economically important. There are a variety of ways to inhibit copper corrosion; the most promising is the use of certain organic compounds, via the mechanism of adsorption, as well as by the formation of complexes (3). There are a number of researches involving the use of N-containing compounds, especially triazoles, for corrosion inhibition (5-9). The groups of compounds of interest include herein two groups: thioureas and quinones.
The mechanism of corrosion inhibition by thioureas has not been comprehensively elucidated, despite of the fact that their complexes have been extensively investigated by X-Ray crystallography (10-11) and electrochemistry, both in aqueous (12) and non-aqueous conditions (13). It is well known that thiourea forms formamidine disulfide on the copper surface, and that greater concentrations cause corrosion, due to the formation of complexes (14-15).
It is known that quinones and similar ring compounds have interactions with copper and act as corrosion inhibitors via adsorption (16), mainly because of their roles in biological cofactors (17). Quinones can also be used as a helper for corrosion inhibition on metal surfaces, by hydrazines (18). It has also been proved that quinones can inhibit mild steel and iron corrosion (18-20). To investigate their behavior, due to their insolubility, ionic liquids are used (21).
Because of the fact that there are quite a few corrosion studies in non-aqueous conditions, the corrosion inhibition behaviors of the two groups of compounds in non-aqueous solvents are the topic to be focused here. Aqueous conditions might resemble the real situation, but they can have the effect of hydrogen and oxygen species on the behavior of the inhibitors. Furthermore, there are a number of electrochemical studies in non-aqueous conditions which should shed some light in understanding Cu corrosion inhibition by thioureas and ring compounds, as well as their complexation mechanism.
Materials and methods
Chemicals
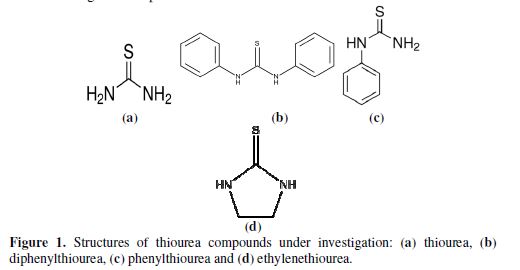
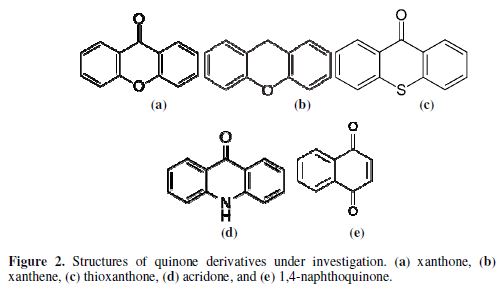
The solvent used was acetonitrile. Thioureas and ring compounds were of analytical grade and used as-received. Fig. 1 and 2 show the molecular structure of the investigated compounds.
Procedures
The electrochemical experiments were carried out using a three electrode cell assembly with Methrom AUTOLAB PGSTAT 100, by a GPES program. In the three-electrode cell, there were used copper coupons of a 2.075 cm2 exposed area as working electrode, high purity platinum wire as counter electrode and silver-silver chloride (Ag/AgCl) as reference electrode. All potentials were measured versus Ag/AgCl.
Before the experiment, the working electrode was polished with emery paper (#1000), followed by washing with acetone. Potentiodynamic polarization studies were performed in the potential range of ±2 V vs. corrosion potential, at a scan rate of 0.1799 Vs-1, to study the effect of inhibitors.
Results and discussion
Corrosion inhibition
The results from the investigation of copper corrosion in four types of ureas – namely, thiourea, diphenylthiourea, phenylthiourea and ethylenethiourea, in acetonitrile, as shown in table 1 – indicate that the metal corroded to the greatest extent in diphenylthiourea, with the corrosion rate of 4.52 ×10-4 mm/year, and that ethylenethiourea was the best inhibitor in this group (2.27×10-4 mm/year). This is due to the fact that, in terms of inductive effect, ethylene is from the electron releasing group, which explains the enhancement of its capability in forming the substances that cover the metal surface. In contrast, the steric structure of the phenyl group, despite its similar inductive effect as an electron donor, hurdles the proximity to the metal surface, where redox reactions occur to form the substances that cover it and inhibit Cu corrosion at this concentration. It has already been shown that, at lower concentrations (‹ 0.1 M), thiourea exhibits corrosion inhibiting characteristics. In contrast, Cu-thiourea suffers high corrosion, which can easily strip off the surface (3). Furthermore, both structure and substitutent groups have been proved to affect metal corrosion behavior (15). In the case of palladium (Pd), the phenyl group showed a planar structure (23), which makes it much more difficult to reach the metal surface. All of the discussed above contributors lead to the following order of corrosion inhibition: ethylenethiourea > thiourea > phenylthiourea > diphenylthiourea. The effect of concentration is not focused here, but it is generally recognized that increased concentrations of corrosion inhibitor result in greater inhibition, especially with physisorbed inhibitors (30-31).
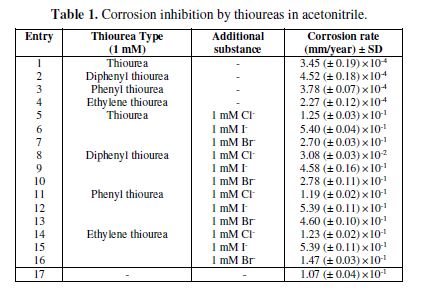
When halides are added to the solution, it is usual that copper corrosion increases, because of the formation of a stripping complex speculated to be CuCl2- (24). With the comparison of the three halides, it was found that all four types of thioureas exhibit the same trend of iodide > bromide > chloride, corresponding to electrochemical and X-ray crystallography results for copper (I) complexes (12,25). These results reveal that, for copper diphenyl thiourea-halide complexes, iodide has the longest bond distance, and only acts as a charge balancing ion; hence, it has greater freedom to form a Cu-halide complex, and directly increases copper corrosion via the formation of complexes. In their turn, bromide and chloride ions can strongly bond with copper, and cause less electron density, as well as reduce the extent of corrosion, due to the fact that they cannot directly attack the metal surface in the same way as with free ions.
Another interesting point is that, with the same halide, the corrosion inhibition trend for Cl- and I- was dptu < ptu < etu < tu. This is due to the fact that the structure of Cu(dptu)2+ is trigonal planar (25), which facilitates the absorption of the thiourea ligand on Cu. In the case of tu, the structure is tetrahedral, which is more steric than a trigonal planar, with the trend to include another additional tu ligand (26). Ptu and etu ligands have a similar coordination number to dptu, but the complexes have the same trend to become dimer (27-28), causing Cu corrosion rate to be essentially the same. For Br-, two doubly bridging etu (28) seem to influence greater adsorption on Cu, at low concentrations, and cause a different trend (etu < tu < dptu < ptu) from those of Cl- and I-. These results represent the close and subtle relationship between the structure of the complexes on the adsorption onto the surface and corrosion inhibition characteristics. However, once again, the effect of concentration has not been inhere investigated, since it is usually the case that the increase in halide concentrations causes greater corrosion rate (32).
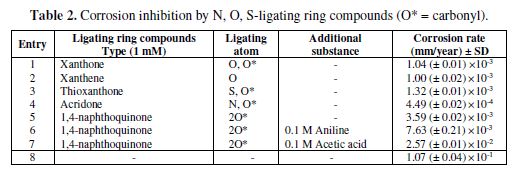
In contrast to thioureas, N, O, S-ligating ring compounds seem to form a complex with copper and attach to its surface; our results reveal that the order of capabilities of corrosion inhibition is nitrogen > oxygen > sulfur > carbonyl, reflecting greater affinity for nitrogen in forming complexes with copper as a film on its surface (29). Acridone, therefore, exhibits the greatest corrosion inhibition with Cu corrosion rate of 4.49×10-4 mm/year. In the case of oxygen, the presence of the carbonyl group has no effect on the corrosion rate (entry 1 and 2 of table 2). When there is no free ligating atom of N, O and S, such as in the case of 1,4-naphthoquinone, a weak interaction between the carbonyl group and copper results in greater corrosion. Furthermore, the corrosion rate in acidic conditions is higher than that in the basic ones, which is normally the case for acid sensitive metals such as copper (9). The inhibition mechanism for physisorbed compounds has been proofed elsewhere to be of the mixed type (30-31).
Conclusion
For thioureas, copper exhibits quite similar corrosion rates, with the lowest in the case of ethylene thioureas, compared with phenylthiourea, due to the least steric effect. With the presence of halides, iodide causes greatest corrosion, due to its freedom to attack Cu surface, in good agreement with the result by X-ray crystallography and electrochemistry. A trigonal planar structure and bridging ligands facilitate the bonding and corrosion inhibition, whereas dimer formation causes an adverse effect.
In the case of N, O, S-ligating ring compounds, the formation of bonds between copper and N can inhibit corrosion, with the capability in the order of nitrogen > oxygen > sulfur > no ligating atoms.
Future studies
- Complexes between metals and certain quinones with and without halides are being synthesized for X-Ray analysis.
- Corrosion of silver-thiourea-halide systems will be investigated in the future, as well as their cyclic voltammetric behaviors.
- Modern techniques such as electrochemical impedance spectroscopy (EIS) and surface spectroscopy, especially X-Ray Photoelectron Spectroscopy or Scanning Electron Microscope (SEM), should provide additional information to delve into a detailed mechanism, as well as copper corrosion inhibition in practice.
- More studies should also be extended to aqueous conditions.
References
(1) Liao X, Cao F, Zheng L, et al. Corrosion behaviour of copper under chloride-containing thin electrolyte layer. Corros Sci. 2011;53:3289-3298. Available from: doi:https://doi.org/10.1016/j.corsci.2011.06.004. [ Links ]
(2) Zhang J, Qiao Q, Hu S, et al. Theoretical evaluation of corrosion inhibition performance of imidazoline compounds with different hydrophilic groups. Corros Sci. 2011;53:147-152. Available from: doi:https://doi.org/10.1016/j.corsci.2010.09.007. [ Links ]
(3) Zhao J, Cui G. Study on Adsorption and Complexation Behavior of Thiourea on Copper Surface. Int J Electrochem Sci. 2011;6:4048-4058. [ Links ]
(4) Tian H, Li W, Cao K, et al. Potent inhibition of copper corrosion in neutral chloride media by novel non-toxic thiadiazole derivatives. Corros Sci. 2013; 73:281-291. Available from: https://doi.org/10.1016/j.corsci.2013.04.017. [ Links ]
(5) Pan YC, Wen Y, Guo XY, et al. 2-Amino-5-(4-pyridinyl)-1,3,4-thiadiazole monolayers on copper surface: Observation of the relationship between its corrosion inhibition and adsorption structure. Corros Sci. 2013;73:274-280. Available from: https://doi.org/10.1016/j.corsci.2013.04.016. [ Links ]
(6) Tansug G, Tuken T, Giray ES, et al. A new corrosion inhibitor for copper protection. Corros Sci. 2014;84:21-29. Available from: https://doi.org/10.1016/j.corsci.2014.03.004. [ Links ]
(7) Khan PF, Shanti V, Babu RK, et al.. Effect of benzotriazole on corrosion inhibition of copper under flow conditions. J Environ Chem Eng. 2015;3(1):10-19. Available from: https://doi.org/10.1016/j.jece.2014.11.005. [ Links ]
(8) Sharif ESM, Eramus RM, Comins JD. Effects of 3-amino-1,2,4-triazole on the inhibition of copper corrosion in acidic chloride solutions. J Colloid Interface Sci. 2007;311:144-151. Available from: doi:https://doi.org/10.1016/j.jcis.2007.02.064. [ Links ]
(9) Sudheer, Quraishi MA. Electrochemical and theoretical investigation of triazole derivatives on corrosion inhibition behavior of copper in hydrochloric acid medium. Corros Sci. 2013;70:161-169. Available from: https://doi.org/10.1016/j.corsci.2013.01.025. [ Links ]
(10) Taylor IF, Weininger MS, Amma EL. Preparation, crystal structure, and bonding in the dimers of tris(thiourea) coppers(I) tetrafluoroborate and tris(s-dimethylthiourea) copper(I) tetrafluoroborate. Inorg Chem. 1974;13:2835–2842. Available from: DOI: https://doi.org/10.1021/ic50142a014.
(11) Bowmaker GA, Pakawatchai C, Skelton BW, et al. Synthesis and Characterization of Coordination Compounds of Silver(I) Nitrite with Ligands Ethylenethiourea and N,N'-diethylthiourea. Z Anorg Chem. 2008; 14:2583-2588. Available from: DOI: https://doi.org/10.21776/ub.jpacr.2016.005.03.253. [ Links ]
(12) Cofre P, Bustos A. Voltammetric behaviour of the copper(II)—thiourea system in sulphuric acid medium at platinum and glassy carbon electrodes. J Appl Electrochem. 1994;24(6):564-568.
(13) Chuaysong R, Chooto P, Pakawatchai C. Electrochemical properties of copper(I) halides and substituted thiourea complexes. Science Asia. 2008; 34(3):440-442. Available from: doi: https://doi.org/10.2306/scienceasia1513-1874.2008.34.440. [ Links ]
(14) Gomez LH, Suarez C, Valanzuela A. Effect of thiourea concentration on the electrochemical behavior of gold and copper electrodes in presence and absence of Cu(II) ions. J Chil Chem Soc. 2009;54(4):439-444. Available from: http://dx.doi.org/10.4067/S0717-97072009000400026. [ Links ]
(15) Mostafa HA, Zaghloul EI, Moussa MN, et al. Corrosion Inhibition of Copper by some thiourea Derivatives. Portugaliae Electrochimica Acta. 2002;20(2): 63-75. [ Links ]
(16) Sayak R, Biprajit S, Denis B, et al. Stabilizing the Elusive ortho-Quinone/Copper(I) Oxidation State Combination through π/π Interaction in an Isolated Complex. J Am Chem Soc. 2008;130(46):15230–15231. Available from: DOI: http://dx.doi.org/10.1021/ja804429v.
(17) Wang CH, Chung SY, Jao JH, et al. Surface-Confined Pyrroloquinoline Quinone: Characterizations and Interactions with Copper Ions. J Phys Chem. 2011;115:1978-1984. Available from: DOI: http://dx.doi.org/10.1021/jp1081774. [ Links ]
(18) US Patent 3551349 A.
(19) Muralidharan S, Phani KLN, Pitchumuni S, et al. Polyamino-Benzoquinone Polymers: A New Class of Corrosion Inhibitors for Mild Steel. J Electrochem Soc. 1995;5:1478-1483. [ Links ]
(20) Musa AY, Jalgham RTT, Mohamad AB. Molecular dynamic and quantum chemical calculations for phthalazine derivatives as corrosion inhibitors of mild steel in 1 M HCl. Corros Sci. 2012;56:176-183. Available from: doi:http://dx.doi.org/10.1016/j.corsci.2011.12.005. [ Links ]
(21) Jeyaprabha C, Sathiyanarayanan S, Phani KLN, Venkatachari G. Influence of poly(aminoquinone) on corrosion inhibition of iron in acid media. Appl Surf Sci. 2005;252:966-975. Available from: DOI: http://dx.doi.org/10.1016/j.apsusc. 2005.01.098. [ Links ]
(22) Puerto ED, Cuesta A, Sanchez-Cortez S, et al. Electrochemical SERS study on a copper electrode of the insoluble organic pigment quinacridone quinone using ionic liquids (BMIMCl and TBAN) as dispersing agents. Analyst. 2013;138:4670-4676. [ Links ]
(23) Anderson JA, Athawale A, Imrie FE, et al. Aqueous phase hydrogenation of substituted phenyls over carbon nanofibre and activated carbon supported Pd. J Catalysis. 2010;270:9-15. Available from: doi:http://dx.doi.org/10.1016/j.jcat.2009.11.028 [ Links ]
(24) Finsgar M, Merl DK. Application of corrosion inhibitors for steels in acidic media for the oil and gas industry: A review. Corros Sci. 2014;80:82-95. Available from: https://doi.org/10.1016/j.corsci.2014.04.044. [ Links ]
(25) Singhagomol L. Crystal structure and thermal analysis of copper (I) diphenylthiourea complexes. MSc thesis, Prince of Songkla University. 1999.
(26) Okaya Y, Knobler CB. Refinement of the crystal structure of tris(thiourea)-copper(I) chloride. Acta Cryst. 1964;17:928-930. Available from: doi: https://doi.org/10.1107/S0365110X64002407. [ Links ]
(27) Battaglia LP, Corradi AB, Nardelli M, et al. X-Ray crystal structures of tetrakis(imidazolidine-2-thionato)copper(I) nitrate and dichloro-µ-imidazolidine-2-thionato-tris(imidazolidine-2-thionato)dicopper(I). J Chem Soc Dalton Trans. 1976;2:143-146. [ Links ]
(28) Bowmaker GA, Hanna JV, Pakawatchai C, et al. Crystal structures and vibrational spectroscopy of copper(I) thiourea complexes. Inorg Chem. 2009; 48(1):350-368. Available from: DOI: https://doi.org/10.1021/ic801310r. [ Links ]
(29) Milan MA, Snezana MM, Marija BP. Films formed on copper surface in chloride media in the presence of azoles. Corros Sci. 2009;51:1228-1237. Available from: doi:https://doi.org/10.1016/j.corsci.2009.03.026. [ Links ]
(30) Chooto P, Manaboot S. Electrochemical, spectrochemical and quantum chemical studies on dimedone as corrosion inhibitor for copper in acetonitrile. Journal of Scientific Research & Reports. 2017;15(2):1-13. Available from: DOI: https://doi.org/10.9734/JSRR/2017/34929. [ Links ]
(31) Gurten AA, Keles H, Bayol E, et al. The effect of temperature and concentration on the inhibition of acid corrosion of carbon steel by newly synthesized Schiff base. Journal of Industrial and Engineering Chemistry. 2015;27:68–78. Available from: https://doi.org/10.1016/j.jiec.2014.11.046.
(32) Emran KM. Effects of concentration and temperature on the corrosion properties of the Fe–Ni–Mn alloy in HCl solutions. Res Chem Intermed. 2015;41:3583–3596. Available from: DOI https://doi.org/10.1007/s11164-013-1473-9.
Received August 6, 2017; accepted March 19, 2018
* Corresponding author. E-mail address: pipat.c@psu.ac.th
Acknowledgements
We wish to thank Assistant Professor Dr. Chaweng Pakawatchai, for X-Ray analysis and Associate Professor Dr. Wilawan Mahabusarakham, for xanthone derivatives. Research scholarship from Graduate School, Prince of Songkla University, is deeply appreciated. All facilities and partial financial support were provided by Department of Chemistry, Faculty of Science, Prince of Songkla University.














