Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Portugaliae Electrochimica Acta
Print version ISSN 0872-1904
Port. Electrochim. Acta vol.37 no.3 Coimbra May 2019
https://doi.org/10.4152/pea.201903179
ARTIGOS
Elaboration, Electrochemical Investigation and morphological Study of the Coating Behavior of a new Polymeric Polyepoxide Architecture: Crosslinked and Hybrid Decaglycidyl of Phosphorus Penta methylene Dianiline on E24 Carbon Steel in 3.5% NaCl
Rachid Hssissoua, Bouchra Benzidiab, Najat Hajjajib and Ahmed Elharfia
(a) Laboratory of Agroressources, Polymers and Process Engineering (LAPPE), Team of Organic Chemistry and Polymer (TOCP), Department of Chemistry, Faculty of Sciences, Ibn Tofail University, P.O. Box 133, 14000, Kénitra, Morocco
(b) Laboratory of Materials, Electrochemistry and Environment, Team of Corrosion, Protection and Environment, Department of Chemistry, Faculty of Sciences, Ibn Tofail University, P.O. Box 133, 14000, Kénitra, Morocco
ABSTRACT
We developed and applied in our work a new polyepoxide polymer, decaglycidyl phosphorus penta methylene dianiline, to study its coating behavior in a marine environment. Then, we tested the new crosslinked and formulated macromolecular binder (DGPMDAP), which is used as an anticorrosive coating on E24 carbon steel, in 3.5% NaCl. So as to evaluate and examine the DGPMDAP coating performance in the field of E24 carbon steel corrosion, we applied the different E1 (DGPMDAP/MDA) and E2 (DGPMDAP/MDA/PN) formulations on the metal substrate. The stationary and transient electrochemical studies are very interesting; and, in order to confirm the obtained results, we dispersed the charge of the natural phosphate that was incorporated in the E2 (DGPMDAP/MDA/PN) macromolecular matrix, through the use of a polarizing optical microscope.
Keywords: DGPMDAP, architecture, coating, crosslinking, formulation, carbon steel, and electrochemical studies.
Introduction
Polyepoxide polymeric architectures are very important thermosetting technological macromolecular matrices [1-3], since they have varied uses in industry, thanks to their numerous viscosimetric and rheological properties [4-6], such as aeronautical and spatial construction [7-9]. Corrosion inhibition [10] by the protection and coating of metallic objects [11] with inhibitors is important in the field of heritage conservation, due to their ease of implementation, according to their microscopic and macroscopic properties. The most common method nowadays used to protect the materials against corrosion is to apply organic polymer films [12-13].
The synthesis and formulation of a new polyepoxide architecture were done, due to its good physicochemical properties as a coating for E24 carbon steel in marine environments. We tested the decaglycidyl polymer of phosphorus pentamethylene dianiline (DGPMDAP), crosslinked and formulated by natural phosphates, among the different coating types that can play an important role in the prevention of corrosion oxidation and heritage degradation [14-15]. This is due to its good adhesion, strong acid/alkali resistance and high cross-link density, according to its macromolecular structure [16-17].
Materials and methods
Used materials
Polyepoxide decaglycidyl of phosphorus penta-methylene dianiline (DGPMDAP)
We used a new polyepoxide polymer in this work: decaglycidyl of phosphorus penta methylene dianiline (DGPMDAP), which structure is shown in Fig. 1, (methylene dianiline (MDA), natural phosphate and 3.5% NaCl). The latter products have been marketed by Aldrich Chemical Co.
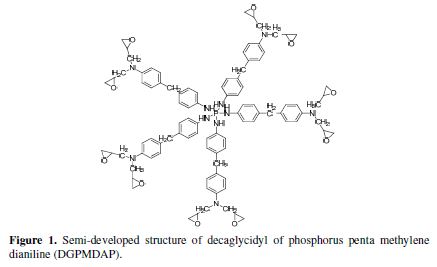
Crosslinking of decaglycidyl polyepoxide of phosphorus penta methylene dianiline using methylene dianiline
The polyepoxide macromolecular polymeric architectures can be transformed into thermosetting materials, through many chemical compounds that act as a curing agent during processing. Among the hardeners of polyepoxide systems there are two major classes: acid anhydrides and amines. The latter remain good hardeners, depending on their chemical structure. The aromatic compounds provide very good thermal stability and mechanical properties to the polymer [18], and are often used for high-tech applications. The hardener used in this study is methylene dianiline. The crosslinking reaction of the new synthesized multifunctional polymer (DGPMDAP), in the presence of methylene dianiline (MDA) as a hardener, was under the temperature effect, following the poly-condensation reaction. This leads to the three-dimensional structure shown in Fig. 2.
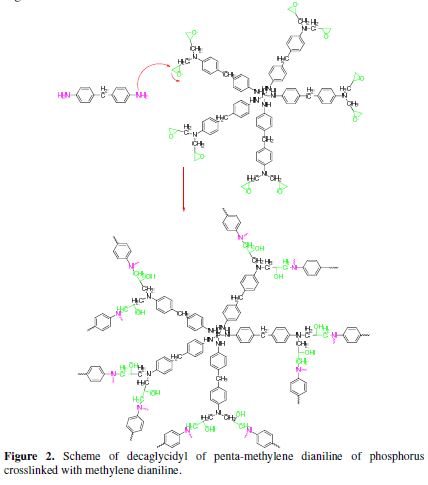
Formulation of decaglycidyl polyepoxide of phosphorus penta methylene dianiline in the presence of natural phosphate
used methods
Electrochemical cell
The electrochemical cell had three electrodes: a platinum counter-electrode, a saturated calomel reference electrode (SCE) and a rectangular E24 carbon steel working electrode. The surface of contact with the corrosive solution was 1 cm². The working electrode was polished before each test with abrasive paper of 600, 1200 and 1500 grades.
These three electrodes were immersed in a 100 mL container, in which orifices of well-designed diameters and spacing were provided, enabling the three electrodes introduction. This also made possible to receive the systems for agitation, temperature control, aeration and deaeration.
Polarizing optical microscope
The optical polarizing microscope is composed of a lens optical system, in order to obtain an enlarged image of the sample. The latter is an optical instrument equipped with an object and an eyepiece, which enables to enlarge the image of an object of small dimensions, and to separate its details, so that it can be observed by the human eye.
Ratio calculation method
In order to obtain optimum properties, it was desirable to make the prepolymer and the curing agent react in approximately stoichiometric amounts [7], after having cured the multifunctional polyepoxide macromolecular matrix in the hardener presence.
The epoxy equivalent weight (EEW) was calculated by:
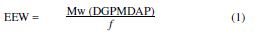
The ratio by weight of the hardener, with respect to the epoxy resin, is most commonly calculated by PHR (Parts per Hundred of Resin):
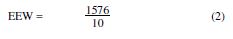
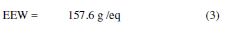
The amine hydrogen equivalent weight (AHEW) was calculated by:
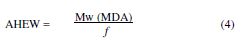
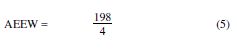
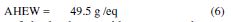
The ratio by weight of the hardener, with respect to the epoxy resin, is most commonly calculated by PHR (Parts per Hundred of Resin):
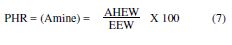
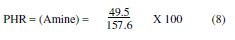
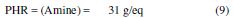
Therefore, 31 g of methylene dianiline per 100 g of DGPMDAP will have to be reacted, in order to achieve the optimum properties, when we cure the prepolymer with methylene dianiline.
The natural phosphate ratio to the weight was calculated by the following equation:
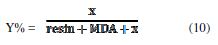
where x is the quantity of the DGPMDAP resin and y is the amount of the natural
phosphate charge.
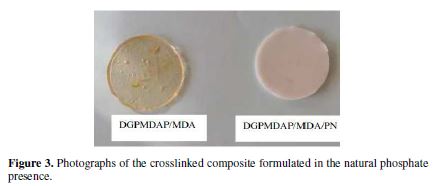
Fig. 3 shows the composite photos (DGPMDAP/MDA/PN).
Method for the E24 carbon steel coating by decaglycidyl of crosslinked and formulated phosphorus penta methylene dianiline
The surface plate was polished with abrasive paper of increasingly fine granulometry (600, 1200 and 1500) before each test, in order to obtain reliable and reproducible results. The used experimental protocol was as follows:
Formulation 1 (DGPMDAP/MDA): we mixed 1 g of DGPMDAP with 0.31 g of MDA as a hardener, and then we stirred the mixture for about 30 min, to give a single fluid phase. The formulation was applied to the E24 carbon steel with a film-producing rod. This coated plate was placed in the oven at 70 °C for 24 hours, to crosslink the polymer which was deposited on the substrate.
Formulation 2 (DGPMDAP/MDA/PN): we followed the same previous protocol (formulation 1) with the addition of 5% of natural phosphate as filler.
Results and discussions
Polarization curves
The stationary measurements were carried out in potentiodynamic mode, through the use of a SP-200 "Biologic Science Instruments" potentiometer/galvanostat. The working electrode was previously immersed at the free corrosion potential for 30 min. The scanning speed was 1 mV/s, and the determination of the electrochemical parameters (icorr, Ecorr, βa and βc) from the polarization curves was done using a non-linear regression by the Ec-Lab software. Thus, the coating efficiency was calculated from the following formula:
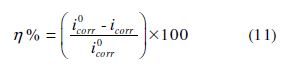
where i°corr and icorr are, respectively, the corrosion current densities (A.cm-2), without and with the coating polymer, with different formulations.
βa and βc are, respectively, the Tafel constant of the anodic and cathodic reactions (V-1). These constants are related to the Tafel β (V dec-1) slope by the form on the logarithmic scale given by:
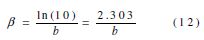
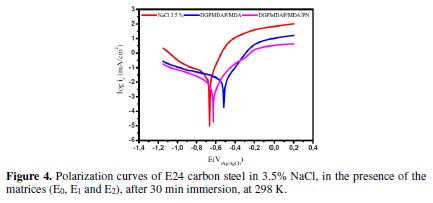
The potentiodynamic polarization curves, which describe the protective effect of the (E0), E1 (DGPMDAP/MDA) and E2 (DGPMDAP/MDA/PN) formulations on E24 carbon steel in a marine environment, are presented in Fig. 4.
The electrochemical parameters of the E0, E1 (DGPMDAP/MDA) and E2 (DGPMDAP/MDA/PN) coating matrices are summarized in Table 1.
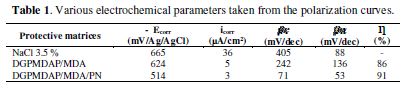
In the control E0 matrix, which does not have the substrate coating, the C1- and hydroxyl OH- chloride anions coming from the corrosive medium directly attack the substrate, inducing direct corrosion. The results in Table 1 show that the addition of the E1 and E2 formulations to the corrosive solution leads to a change in the corrosion potential (Ecorr), in the cathodic and anodic domains towards the positive values, ??and to a marked decrease in the current density.
In this part of our work, we studied the protection mechanism of E24 carbon steel by the following new matrices (E1 and E2), mainly composed of the new polymeric polyepoxide architecture (DGPMDAP), the hardener (MDA) and the natural phosphate (PN) charge. The results interpretation, according to the constitution of the multifunctional macromolecular matrix, is ??given as follows.
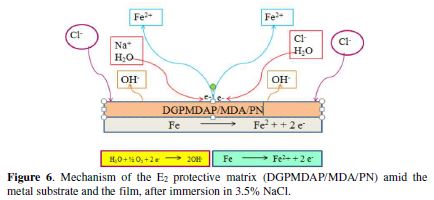
Figs. 5 and 6 show, respectively, the protection mechanism of E24 carbon steel by the E1 (DGPMDAP/MDA) and E2 (DGPMDAP/MDA/PN) formulations, with and without natural phosphate.
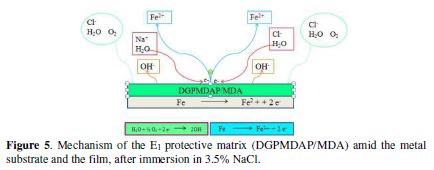
In the E1 protective matrix (DGPMDAP/MDA) presence on the metal substrate (E24 carbon steel) immersed in the sorptive solution for 30 min, we observed a relatively large diffusion of the chloride and hydroxyl ions through the protection film, which could present micropores by inducing primary corrosion translated by icorr = 5 μA/cm², and an efficiency of ?% = 86%. This is explained by the acceleration of the cathodic and anodic metal dissolution [19]. The protection mechanism of the E1 matrix is shown in Fig. 5.
Furthermore, through the E2 protective matrix (DGPMDAP/MDA/PN) on the E24 carbon steel immersed in the sorptive solution for 30 min, in the presence of natural phosphate as filler, we found that the corrosive species (chloride ions Cl- and hydroxyl OH-) diffusion is slowed down due to the layer formed by natural phosphate [20-21]. This indicates the decrease in the dissolution rate of the substrate coated by the E2 formulation. Thus, the corrosion electrochemical parameters are icorr = 3 μA/cm², and the coating efficiency reaches a maximum value of ? = 91%. The E2 matrix protection mechanism is shown in Fig. 6.
The corrosion potential globally moves towards the anode direction, after the E1 (DGPMDAP/MDA) and E2 (DGPMDAP/MDA/PN) protective matrices addition, and the addition of 5% of natural phosphate is accompanied by a marked decrease in the corrosion current, in both the cathodic and anodic domains. This allowed us to conclude that the natural phosphate addition to the E2 protective matrix makes it well cured and to act as a mixed inhibitor.
Electrochemical impedance
The electrochemical impedance spectroscopy measurements were carried out by using the same apparatus with a signal amplitude of 10 mV, and the explored frequency range varied from 100 kHz to 10 mHz. The results were then analyzed by using an equivalent electrical circuit, and the Bouckamp simulation program [22]. The effectiveness of the coating protection was calculated by the following equation:
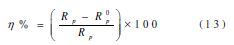
where Rop and Rp are, respectively, the polarization resistance in the absence and presence of the coating polymer with different formulations.
The results of the electrochemical impedance spectroscopy are presented in the form of Nyquist diagrams. In order to study the coating behavior mechanism in detail, we carried out the impedance diagrams of the E24 carbon steel immersed in the NaCl 3.5% corrosive solution for 30 min, with respect to the assembly of the protective matrices (E0, E1 and E2). The latter are presented in Fig. 7, which confirms the results obtained by the potentiodynamic polarization curves.
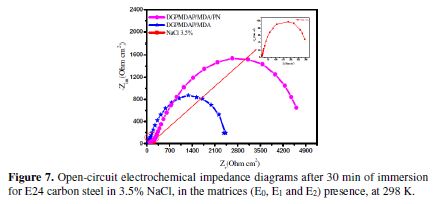
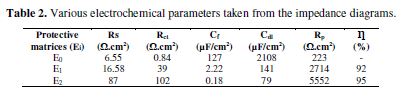
The analysis of the different obtained results from the protective matrices (E0, E1 and E2) shows, respectively, that the latter are formed by a high-frequency capacitive loop attributed to the film effect, which is due to the diffusion phenomenon. On the other hand, the more the semi-circle diameter increases, the better is the protective film corrosion resistance [22].
The latter depends on the E1 and E2 coating formulations, and increases with the presence of the natural phosphate charge. This indicates that the coated substrate impedance increases with the formulations, which also increases the coating behavior effectiveness. These results confirm those obtained by the stationary method.
However, the E1 and E2 formulations addition improves the Rtc values, ??and decreases the Cdt values, because these observations clearly point out that: the E24 carbon steel corrosion in 3.5% NaCl is controlled by a charge transfer process; and that the coating behavior occurs through the adsorption of the new polyepoxide DGPMDAP polymeric architecture onto the E24 carbon steel surface. The decrease in the Cdt values ??may result from a decrease in the local dielectric constant, and/or an increase in the current density. The electrical double layer thickness suggests that the DGPMDAP macromolecular matrix functions by adsorption onto the metal solution substrate interface [23]. The effectiveness of a compound as a corrosion coating primarily depends on both the size and active centers of the DGPMDAP compound. The DGPMDAP coating improved performance can be attributed to the epoxide group’s structure, and to the larger size of the DGPMDAP macromolecular matrix. Indeed, the larger semi-circle in the Nyquist diagram of the E2 coating system indicates, more precisely, the corrosion resistance. This can be explained by the natural phosphate presence, responsible for the higher corrosion resistance. Therefore, we conclude from these results that the coating film polarization resistance values for the E0, E1 and E2 protective matrices confirm the results of the parameters from the potentiodynamic polarization curves.
Figs. 8 and 9, respectively, show the phase angle and Bode diagrams of the E24 carbon steel coating behavior in a marine environment, which confirm the electrochemical impedance spectroscopy study. The diagrams plots of different E0, E1 and E2 formulations are illustrated in the figures below.
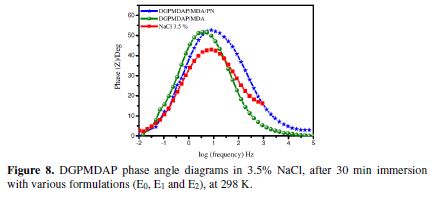
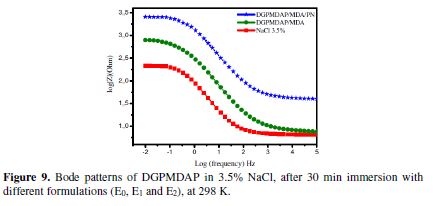
Fig. 8 shows the increase in the phase angle, according to the frequency, with different coating formulations. This increase in the phase angles confirms a higher protection by stronger coating formulations (E0, E1 and E2); also, from this diagram, we have noticed that there are three frequency domains: low frequencies, intermediate frequencies and high frequencies. The phase angle values are approximately equal to zero at high frequencies, which indicates that the electrode behavior corresponds to the solution resistance [24]. For the intermediate frequencies, the phase angle is close to 60 °, and there is a linear relation between log | Z | according to log (f), with a slope close to -1, which indicates the coating capacitive behavior at intermediate frequencies.
For low frequencies, the increase in the impedance absolute values (Fig. 9) confirms the greatest protection of E24 carbon steel by the different coating formulations.
In the literature, a capacitor behavior would be ideal if the slope value reached -1, and a phase angle value reached 90 °. The phase angle values ??were calculated from the phase curves (Z), according to log (frequency). These α values range from -0.60 to -0.80 for the various coating formulations, which is related to the non-ideal structure of the metal/solution interface. These results confirm that of the electrochemical impedance spectroscopy.
According to the presented curve shapes, the Bode and Nyquist diagrams show a capacitive behavior at the interface, and the existence of an equivalent electrical circuit (Fig. 10), which is due to the interpretation of the information on the behavior properties of the metal surface coating or corrosion. This circuit is composed of: electrolyte resistance (Rs), coating film capacity (Cf), coating film resistance (Rp), load transfer resistor (Rct) and double layer capacity (Cdl).
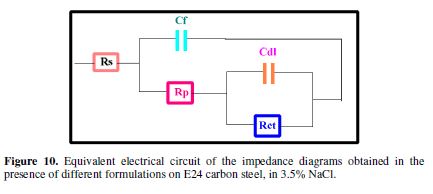
The electrochemical impedance ??and corrosion inhibition efficiency values resulting from each protection matrix (E0, E1 and E2) are summarized in Table 2.
Optic polarizer microscope
We showed in this part of our work the dispersion of natural phosphate charge immersed in the E2 macromolecular matrix (DGPMDAP/MDA/PN) on the E24 carbon steel surface, using a polarizing optical microscope to confirm the previously obtained results. The surface results obtained from the different samples (E0, E1 and E2) of the standard prepared substrate, crosslinked and formulated with the natural phosphate charge, are shown in Fig. 11.

The surface morphology of different formulations (E0, E1 and E2) was observed by a field emission polarizing optical microscope. The comparison of different surface morphologies based on two different E1 (PGEPBAP/MDA) and E2 (PGEPBAP/MDA/PN) formulations has shown that the formulation type is a key factor in controlling the morphology and good dispersion of the charge in the matrix onto the substrate. According to these observations of the surface morphology of the elaborated new E1 and E2 matrices, we have clearly shown the effective dispersion of the natural phosphate charge incorporated in the E2 matrix. This confirms the results which were, respectively, obtained by the stationary method and the transient method.
Conclusion
After the synthesis of the novel polymeric polyepoxide decafunctional architecture, decaglycidyl phosphorus penta methylene dianiline, we applied it for E24 carbon steel protection by the coating behavior process derived from the standard epoxy prepolymer formulations, methylene dianiline and natural phosphate used as a load. The electrochemical studies results, namely, the potentiodynamic polarization curves, and the electrochemical impedance spectroscopy, are quite in reasonable agreement, as the addition of natural phosphate to the formulated E2 matrix (DGPMDAP/MDA/PN) has a very good protection effect. The potentiodynamic polarization that reflects the performed method initially shows the current density diminution in the cathodic and anodic domains. However, the electrochemical impedance spectroscopy reflecting the transient method shows the best results for the coating by the charged matrix (E2), followed by the retuned matrix (E1), and the control. The phase angle and Bode diagrams confirm the electrochemical impedance spectroscopy results, as well as the formulated E2 matrix morphological study using the polarizing optical microscope. In prospect, we will carry out the theoretical study with the DFT method. This will be done to confirm the adhesion sites on the metal support.
References
- Fetouaki S, Toufik M, Meghraoui H, Elharfi A. Phys. Chem. Ne. J. 2006; 27: 131-140.
- Hsissou R, Elharfi A. International Journal of Chem.Tech. Research, 2017; 10 (2); 891-902.
- Hsissou R, El Rhayam Y, Elharfi A. Inter. J. Innov. Appli. Stud. 2014; 7 (2); 674-682.
- Hsissou R, Dagdag O, Elharfi A. Mor. J. Chem. 3 (4), (2015) 791-797.
- Hsissou R, Elharfi A. Mor. J. Chem. 2016 ; 4 (2); 315-323.
- Hsissou R, Bekhta A, Elharfi A. J. Mater. Environ. Sci., 2017; 8 (2); 603-610.
- Grich M, Meghraoui H, Ziraoui R, El Gouri M, Elharfi A. Ann. Chim. Sci. Mat., 2011; 36 (1); 1-10.
- Hsissou R, El Bouchti M, Elharfi A. J. Mater. Environ. Sci., 2017; 8 (12); 4349-4361.
- Gohardani O, Williamson DM, Hammond DW. J. Wear., 2012; 294–295; 336-346.
- Dagdag O, El Gouri M, Galai M, Ebn Touhami M, Essamri A, Elharfi A. Der Pharma Chemica, 2015 ; 7(4) ; 114-122.
- Hsissou R, Benassaoui H, Benhiba F, Hajjaji N, Elharfi A. Journal of Chemical Technology and Metallurgy. 2017; 52 (3); 431-438.
- Cantor A, Bushman J, Glodoski M, Kiefer E, Bersch R, Wallenkamp H. Mater. Performance, 2006; 45; 38–41.
- Antonijevic MM, Petrovic MB. Int. J. Electrochem. Sci., 2008; 3; 1–28.
- Petitjean J, Aeiyach S, Lacroix JC, Lacaze PC. J. Electroanal. Chem., 1999; 478; 92–100.
- Panah NB, Danaee I. Prog. Org. Coat., 2010; 68; 214–218.
- Ferreira CA, Aeiyach S, Delamar M, Lacaze PC. Chem. Interfacial Electrochem., 1990; 284; 351–369.
- Hsissou R, Bekhta A, El Hilal B, El Aazaoui J, Dagdag O, Abouelouafa K, Elharfi A. Inter. J. Sci. Eng. Res., 2017; 8(1); 1181-1188.
- Hsissou R, Rafik M, Hegazi SE, Elharfi A. Ara. J. Che. Env. Res., 2016; 3 (1); 35-50.
- Zubielewicz M, Gnot W. Prog. Org. Coatings. 2004; 49; 358-371.
- Rossenbeck B, Ebbinghaus P, Stratmann M, Grundmeier G. Corros. Sci. 2006; 48; 3703
- Liu S, Zhong Y, Jiang R, Zeng Z, Feng Z, Xiao R. Corros. Sci. 2011; 53; 746.
- Ananda Kumar S, Balakrishnan T, Alagar M, Denchev Z. Progress in Organic Coatings, 2006; 55; 207-217.
- Benali O, Larabi L, Traisnel M, Gengenbre L, Harek Y. Appl. Surf. Sci. 2007; 253; 6130–6139.
- Guillaumin V, Landolt D. Corr. Sci., 2002; 44 (1); 179.
E-mail address:r.hsissou@gmail.com














