Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Portugaliae Electrochimica Acta
versão impressa ISSN 0872-1904
Port. Electrochim. Acta vol.34 no.4 Coimbra jul. 2016
https://doi.org/10.4152/pea.201604277
Alternative Electrocoagulation for Livestock Wastewater Treatment
Jose Pinedo-Hernandez* , Roberth Paternina-Uribe and Jose Marrugo-Negrete
University of Cordoba, Faculty of Basic Sciences, Department of Chemistry, Water, Applied and Environmental Chemistry Group, Monteria, Colombia
Abstract
This project assessed the technical feasibility of organic matter (COD) removal in livestock effluents, by electrocoagulation. An experimental design was used to block two factors at three levels, to evaluate the effect of the variables distance between electrodes and pH, using aluminum sacrificial electrodes. Maximum removal (90.16%) was obtained at 7 units pH, and 2.0 cm distance between electrodes. This study demonstrated the technical feasibility of electrocoagulation (EC) for the removal of organic matter as COD, present in wastewater from the livestock industry.
Keywords: Livestock effluents, Electrocoagulation, Chemical Oxygen Demand (COD).
Introduction
Colombian livestock represents a very important sector for the economy's main axis of the Caribbean region, especially in the department of Cordoba, due to certain geographical conditions characteristic of the area [1]. In livestock industry, the wastewater generated as cattle bath product represents a great threat to the environment; it contains traces of recalcitrant pesticides, toxic compounds and high levels of organic matter [2]. Electrocoagulation (EC) may be used as an alternative system of wastewater treatment, because it is inexpensive, the equipment used is simple and of easy operation, compared to the conventional methods (chemical coagulation), no chemical substances are used, and produces large and more stable flocs than those formed by chemical coagulation [3, 4]. Electrocoagulation is receiving an increasing acceptance by industry, in view of its advantages compared to other methods. The method is based on anodic dissolution of metallic aluminium and the formation of aluminium ions in the vicinity of the anode, these ions being immediately converted to the corresponding hydroxides. The hydroxide, in the process of coagulation and flocculation, has highly adsorptive and adherent qualities, and is colloidally dispersed. When a current is passed through Al anodic dissolution, it takes place according to the following reaction:
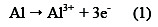
Simultaneously, water is reduced at the cathode to hydrogen gas and hydroxyl ion (OH-)
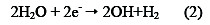
Thus, electrocoagulation introduces metal cations in situ, electrochemically, using sacrificial anodes. Al3+ hydrolyzes in water, forming the corresponding hydroxide; Eqs. (3) - (5) illustrate this, in the case of aluminium anode charge on the organic matter, which is responsible for the stability:
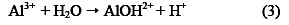
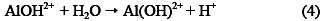
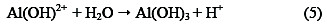
The main objective of this study was to evaluate the removal of organic matter (COD) in livestock effluents by electrocoagulation, controlling pH and distance variables between electrodes.
On this basis, it was expected that using Al anode would improve the process of electrocoagulation, through enhancing the rate of mass transfer of Al3+ from the anode surface to the bulk solution, to be applied to continuous flow process. This would reduce the concentration of polarization, and hence, reduce the passivation tendency of the anode, which adversely affects the process of electrocoagulation.
Materials and methods
Sample collection and preservation
Wastewater sample under study was taken from a cattle farm in the town of Monteria, Cordoba -Colombia, in a day of cattle bath. Effluents were collected in plastic tanks, sufficient for processing in the EC system. The processing was performed before and after pH treatment, and the COD analysis was based on Standard Methods 4500H B and 5220 D [5] procedures references. Initial pH and COD were 6.5 units and 680 mg L-1.
Electrocoagulation cell
Fig. 1 shows the experimental scheme of the EC process.
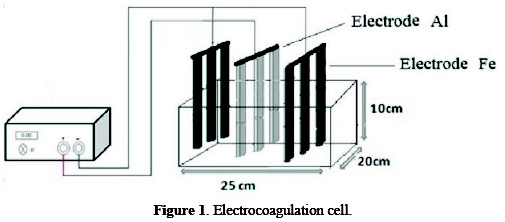
Volume was used for each 4.0 L test sample. Six iron electrodes in the reactor were used as cathodes, and three aluminium anodes with effective dimensions of 18 × 2.5 × 0.5 cm were connected to DC -PHYWE 0-50 V fluent potential. The entire submerged surface of every electrode was 25 cm2.
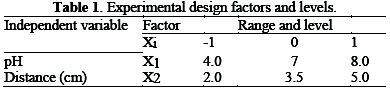
Experimental design
Statgraphics Centurion 15.2.06 software was used for the statistical design of experiments and data analysis. The two most important operating variables, initial wastewater pH (x1) and distance (x2), were optimized for both wastewaters. Their range and levels are shown in Table 1.
Levels of every factor were evaluated in triplicate; there were 27 essays in the whole process. Percentage of COD removal was established as the response variable. Time and potential were 30 min and 50 V. Aluminium was used as sacrificial electrode. Fixed variables sets were selected based on tests conducted by Mestra and Pineda [6].
Statistical analysis
The data were submitted to ANOVA test, and mean comparisons were performed when needed using Tukey tests. Response Surface Methodology (RSM) was applied to evaluate the simple and combined effects of three independent parameters on removal and optimizing of the operating conditions. Statgraphics Centurion 15.2.06 statistical software was used for all analysis. A significance level of 0.05 was selected.
Results and discussion
COD removal function of pH and distance
The removal of COD in function of pH and distance between electrodes is shown in Fig. 2.
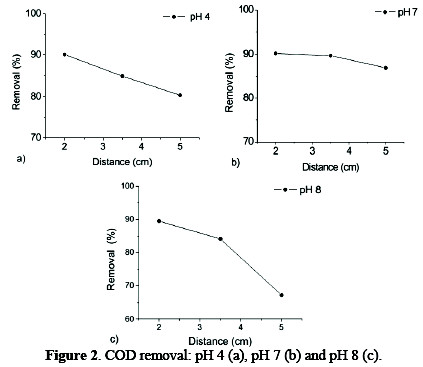
Removal values are higher than 80% at initial pH of 4 and 7, independently of distance. However, initial pH of 8 and 5 cm distance show 67.21% removal (Fig. 2c). It is observed that decreasing the distance between electrodes increases the removal of COD, reaching maximum values (> 80%) at a distance of 2 cm for different pH values. This is possibly due to the electrostatic field formed during the electrocoagulation process, which depends on the distance between electrodes [7-8], and causes the metal ions production by the anode (sacrificial electrode): its function is to destabilize loads possessing contaminant particles present in water, neutralize the systems that keep the particles in suspension - allowing the formation of aggregates of contaminants, and initiating the coagulation process in less time, with higher removal [9].
Fig. 2b shows that, at pH 7, COD removal decreases according to the distance, with no statistically significant difference (p > 0.05), unlike pH 4 and 8, which have a statistically major difference (p < 0.05). This can be attributed to pH < 7, because the formed hydroxides are not stable enough to react with the aluminum cation, not allowing coagulant formation [10-12]. A pH near 7 facilitates the generation of hydroxyl radicals and, in its turn, the formation of agglomerates, which are ultimately removed from the solution [13]. With pH values > 7 the oxidative potential in question decreases; this justifies the reduction in the removal of reactions involved in the process, and therefore, a decrease in pollutant removal [14].
Optimization of the treatment conditions
The application of RSM based on the parameters estimation generates the second order regression model, where the removal percentage of COD (y) and the independent variables studied are related (Eq. 6).
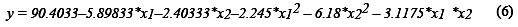
The coefficient of determination (r2) was 0.843, which implies that 84.3 % of the variations in COD removal are explained through independent variables, and 15.7 % of variations cannot be explained by the model. According to Montgomery [15], it is satisfactory that (r2) is at least 75%, when considering proceeding with the methodology. The model generates the optimum values for the maximum COD removal, as a function of pH and distance. The response surface calculated on the basis of the model (Fig. 3) allows visualizing the behavior of the response variable, and clearly indicates the factors levels combination that leads to a maximum value.
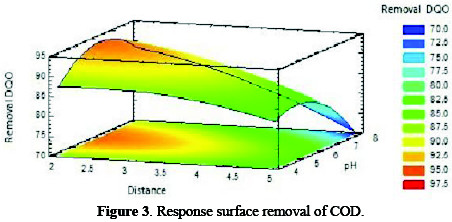
In this study, it is observed that the best results are in the orange region, where factors interaction leads to results between 92.5 and 95 %.
Significant differences were not observed (p < 0.05) when the response was compared to the corresponding experimental value, which confirms that RSM can be used to optimize the process parameters (Table 2).

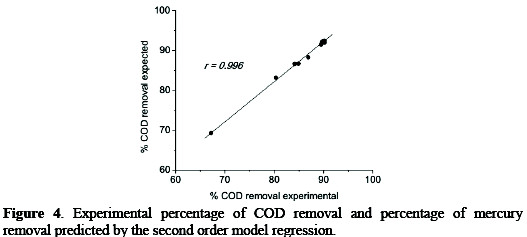
Additionally, it is observed that the results of the second order regression model present significant correlations with the experimentally obtained results (r=0.996, p=0.01, n=27; Fig. 4).
The variance analysis is shown in Table 3.
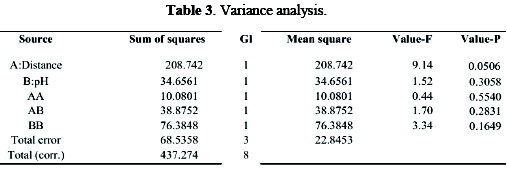
It is observed that the independent variables are not statistically significant (p > 0.05). However, the distance factor has a value close to 0.05, which can reveal the removal difference based on this factor.
To assess the adequacy of the developed model, the difference between Experiment and Predicted (waste) response is used to graphically analyze the effectiveness of the model. Waste is considered as unexplained variations by the model, and they will have a normal distribution if the model accurately predicts normal residue probability graph and normal distribution. This should give a linear fashion, and graphic of residuals versus predicted values should represent a random pattern of residues around zero [16]. Fig. 5a shows the normal probability graph waste for use as Al anode electrode in the optimization model for COD removal from effluents of livestock industry, where it meets the criteria of statistical normality by its linear trend.
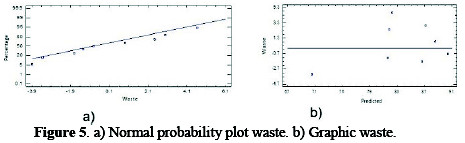
Fig. 5b shows the graph of actual versus predicted residuals for the removal of COD, exhibiting a random pattern around zero waste, which represents a normal distribution.
The electrical energy consumption was calculated in terms of KHz. E (kWh) = U × I × t /1000, where U is cell voltage (V), I is current (A), t is the time of EC (h). The implementation of EC unit in large scale level mainly depends on the treatment process cost. In order to find out the economy of the proposed treatment method, the economic evaluation was made in optimum operating conditions. It was found that the energy supplied to wastewater treatment by EC is 0.03 KHz, given that KHz price in Colombia ($ 349.7; $US 0.13) is $ 4.37 for volume of solution (0.004 m3), i.e., 1092.8 $/m3 (0.39 $US/m3). Related to the consumption of Al per test (0.0643 g / L) by m3 (64.25 g / m3), the A1 material price ($ 4313.7 / kg) is obtained by m3 ($ 277.2 / m3; $ 0.11 US / m3). This result illustrates the economical feasibility of the proposed treatment in the on-field implementation, i.e., wastewater treatment plants (WTPâs). pH and temperature should be taken into account to develop processes to improve electrocoagulation process. It has been determined in some cases that greater removal of a contaminant occurs within a specific pH range. This range can be even wider. In general, the literature indicates that the best removals were obtained for pH values close to 7, which improves electrocoagulation process [18].
The pre treatment temperature was 28 °C, and post treatment temperature increased 2 °C. Chen [19] indicates that the system temperature must be lower than or equal to 60 °C, to achieve greater removal.
Conclusions
Electrocoagulation (EC) and effluent treatment bath livestock is a technically viable alternative for the removal of organic matter as COD. The results showed that, for a distance between electrodes of 2 cm, COD removal percentage was significantly higher. The applied electrochemical treatment allowed the removal of COD in 90.16%, under conditions of optimal electrocoagulation: pH (7.0), distance (2 cm), voltage (50 V), time (30 min). These results indicated that electrochemical treatment process is an effective treatment method, in terms of removal efficiency, with reasonable operating costs.
References
1. Lombana J, Martinez D, Valverde M, et al. Caracterization the livestock sector of the Colombian Caribbean. Editorial Universidad del Norte; 2012. [ Links ]
2. Barrios J, Yepez J. Evaluation model of solar photocatalytic degradation kinetic in a cpc reactor of pesticide used in the bathroom of cattle. Graduation Project: Universidad de Cartagena, Facultad de Ingenieria, Cartagena-Colombia; 2010. [ Links ]
3. Rios GB, Almerava F, Herrera MT. Port Electrochim Acta. 2005;23:17. [ Links ]
4. Holt PK, Barton GW, Mitchell CA. Chemosphere. 2005;59:355. [ Links ]
5. APHA, Standard Methods for the Examination of Water and Wastewater. 21th ed. Washington, DC: American Public Health Association, American Water Works Association, Water Pollution Control Federation; 2005. [ Links ]
6. Mestra D, Pineda R. Electrocoagulacion como alternativa para el tratamiento de aguas residuales del bano de ganado bovino en el departamento de Cordoba. Universidad de Cordoba, Colombia; 2015. [ Links ]
7. Holt P, Barton G, Mitchell C. Electrocoagulation as a Wastewater Treatment. The Third Annual Australian Environmental Engineering Research Event; 1999. [ Links ]
8. Daneshvar N, Sorkhabi HA, Kasiri MB. J Hazard Mater. 2004;112:55. [ Links ]
9. Chen G. J Environ Eng. 2000;126:858. [ Links ]
10. Chen G. Sep Purifi Technol. 2004;38:11. [ Links ]
11. Kumar P, Chaudhari S, Khilar K, et al. Chemosphere. 2004;55:1245. [ Links ]
12. Bayramoglu M, Kobya M, Can O, et al. Sep Purif Rev. 2004;37:117. [ Links ]
13. Heidmann I, Calmano W. J Hazard Mater. 2008;152:934. [ Links ]
14. Linares I, Martinez V, Barrera C, et al. Avances Ciencias Ingenieria. 2011;2:21. [ Links ]
15. Montgomery DC. Design and analysis of experiments. Nebraska: John Wiley & Sons; 2000. [ Links ]
16 Sarabia LA, Ortiz MC. Response surface methodology. In: Brown SD, Tauler R, Walczak B. editors. Comprehensive chemometrics. Oxford: Elsevier; 2009. pages 345-390. [ Links ]
17. Thirugnanasambandham K, Sivakumar V, Maran JJP. J Taiwan Inst Chem Eng. 2015;46:160. [ Links ]
18. Chen G. Sep Purif Technol. 2004;38:11. [ Links ]
19. Chen G, Chen X, Yue PL. J Environ Eng. 2000;858. [ Links ]
Acknowledgements
The authors wish to thank the Water, Applied and Environmental Chemistry Group, Laboratory of Toxicology and Environmental Management, University of Cordoba, Monteria-Colombia.
*Corresponding author. E-mail address: josejph@hotmail.com
Received May 17, 2016; accepted August 23, 2016














