Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Portugaliae Electrochimica Acta
versão impressa ISSN 0872-1904
Port. Electrochim. Acta vol.30 no.2 Coimbra mar. 2012
https://doi.org/10.4152/pea.201202121
Kinetic Study of the Degradation of Crystal Violet by K2S2O8. Comparison with Malachite Green
L.M.A. Fayoumi, M.A. Ezzedine, H.H. Akel, M.M. El Jamal*
Chemistry Department, Faculty of Sciences (I), Lebanese University, El Hadeth, Lebanon
doi: 10.4152/pea.201202121
Abstract
The degradation of crystal violet (CV) and malachite green (MG) by potassium persulfate, was investigated by spectrophotometric methods. The behavior of degradation of crystal violet by persulfate was found to be similar to that of malachite green with only one important difference concerning the order with respect to the dye: the degradation is pseudo second order with respect to CV, but first order with respect to MG. The order with respect to persulfate is one in both cases. Degradation of CV by persulfate was effective at pH range of 2-8 and was found to increase with an increase in the initial concentrations of persulfate, temperature, and the presence of Ag+. The factors that were found to decrease the degradation rate of CV include: the initial concentration of CV, and the presence of halide salts and of the surfactant SDS. The rate of degradation remained the same after addition of Co(II), Ni(II) and Cu(II) salts. The activation parameters of the degradation reaction (Ea, ΔG‡, ΔH‡ and ΔS‡) were calculated. Finally, cytotoxic study revealed a decrease in the toxicity of the degradation products.
Keywords: crystal violet, persulfate, degradation, matrix effect, cytotoxic study.
Introduction
Textile industry produces large amounts of highly colored effluents, which are generally toxic and resistant to destruction by biological treatment methods. Basic dyes, such as crystal violet (CV) and malachite green (MG) are widely used in textile, paper, leather, cosmetic, and food industries [1-4]. Crystal violet is a triphenyltmethane dye, with one dimethylamino group on each phenyl ring.
It is antimicrobial, mutagenic, and used to prevent fungal growth in poultry feed. It is used as a bacteriostatic agent in medical solutions. MG has similar chemical structure to CV, but it has only two out of three phenyl rings substituted with dimethylamino groups, allowing a partial planar structure of MG+.
Various chemical and physical processes, such as electrodegradation [5-8], photodegradation [9-11], adsorption [12-14], are applied for color removal from textile effluents.
Chemical oxidation of contaminants by oxidants has been studied to develop novel remediation technologies. The oxidants, KBrO3 [15], KClO3 [16], O3 [17], Fenton's reagent [18-19], H2O2 [20] and persulfate (K2S2O8) [21-24], have been widely tested in laboratory work and used for the remediation of wastewater and groundwater contaminated by organic compounds. The use of persulfate has recently been the focus of attention for an alternative oxidant in the chemical oxidation of contaminants [21, 25, 26].
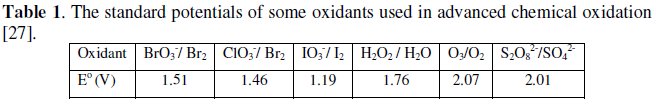
Persulfate (KSP) is one of the strongest oxidants known in aqueous solution and has a higher potential (E0 = 2.01 V) than H2O2 (E0 = 1.76 V) [27] (Table 1).
It offers some advantages over other oxidants as a solid chemical at ambient temperature with ease of storage and transport, high stability, high aqueous solubility and relatively low cost [24]. It has great capability for degrading numerous organic contaminants through free radicals (SO4-* and OH-*) generated in the persulfate system [21, 25, 28].
Thermal activation [26, 28, 29], activation by transition metal catalysis [26, 3032], base activation [34] and UV-irradiation [22, 24, 35, 36] have been used to generate a sulfate radical, a stronger oxidant (E0= 2.5-3.1 0 V, [29, 37]) than persulfate:
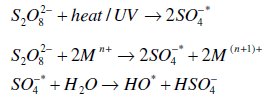
Usually, a great amount of salts are employed in various dyeing processes and the strength of dissolved inorganic ions in dyeing wastewater may affect the efficiency of dye degradation reaction. The presence of some metallic ions such as Co2+, Ni2+, Fe2+ and Ag+ may affect the rate constant through formation of metallic ion of higher oxidation degree, able to react faster with CV or MG [38, 39]. Catalytic kinetic methods for determination of heavy metal ions are based on the degradation of CV or MG [40, 41]. The addition in low concentration of 1,8-dimethyl- 1,3,6,8,10,13-hexaazacyclotetradecane-Ni2+ increased strongly the degradation rate of MG by persulfate. The order of degradation of MG with persulfate under mentioned experimental conditions is as follows: Fe2+ < Ni2+ < persulfate alone < Ni2+ complex [38]. Also the presence of halide salt may affect negatively the rate constant by reaction with persulfate directly or indirectly through reaction with SO4-* or by increasing the ionic strength [36, 42]. The present work is focused on the kinetic study of the oxidation of CV with persulfate by UV-Visible spectrophotometry. The effect of various parameters such as pH, initial persulfate concentration, initial CV concentration, transition metals concentration, and halide salts concentration was studied. A comparison between the degradation of CV and MG with persulfate is undertaken to interpret better the results.
Experimental
Crystal violet is used as purchased from BDH (C25H30N3Cl, MW: 407 g mol-1). Malachite green oxalate (C46H50N4 - 3 C2H2O4, MW: 929.02 g mol-1) was supplied by Sigma-Aldrich. The other chemical reagents used were of analytical grade. Diluted solution of CV (12 mg L-1) was prepared from a mother solution (120 mg L-1) in order to have an initial absorbance at maximum wavelength (590 nm) close to 1. The kinetic study of the discoloration of CV by persulfate was carried on a double beam spectrophotometer, Specord 200 (Analytical Jena). In some cases, Na2SO4 solution was added to the reactional mixture in order to keep the ionic force constant. Salt solutions of halide (1 M KCl, 1 M KBr, and 0.01 M KI), and transition metal solutions (10 g L-1) of Co(NO3)2, Ni(NO3)2, CuSO4, FeII(NH4)2(SO4)2.6 H2O, and AgNO3 were also prepared to study the matrix effect on the degradation rate.
Kinetic study
The order with respect to CV was carried out in the presence of a large excess of persulfate. The order with respect to persulfate was determined in the presence of different excess amounts, while other parameters were kept constant. For the determination of the pseudo order with respect to CV and the effect of the initial CV concentration, the reactional mixture was obtained as follows: a specific amount (x mL) of 12 mg L-1 of CV was added to a mixture of 5 mL of 0.05 M persulfate and (5-x) mL of H2O. For the determination of the pseudo order with respect to persulfate: 5 mL of 12 mg L-1 CV were added to a mixture of (5 - x) mL of 0.05 M persulfate and x mL of 0.05 M of Na2SO4. Quickly after the addition of the dye, the absorbance of the solution was recorded every 20 s for 10 minutes at 590 nm (λmax of CV). The rate expression for the discoloration reaction is:
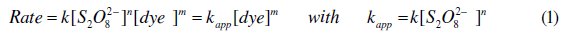
where k is the rate constant of the reaction, m and n are the pseudo order of the reaction with respect to CV and K2S2O8, respectively. The effect of pH on the degradation rate was studied by replacing water with 0.1 M of the solutions H3PO4, KH2PO4 and Na2HPO4 separately. The effect of salt, transition metal and surfactant (SDS) on the rate constant was carried out at several concentrations of the mentioned salts, while keeping the other reactants constant. The same procedure was used for malachite green.
Cytotoxic study
In order to study the cytotoxic effect of crystal violet on human cells, and if the pretreatment with K2S2O8 (KSP) protects against its cytotoxicity, two Blymphocytic cell lines were used: SU-DHL-4 and OCI-LY-1 [43-45]. Both cell lines were cultured in suspension in RPMI-1640 media which was supplemented with 10 % heat inactivated fetal bovine serum (FBS), L-glutamine (100x GlutaMaX, Gibco/Invitogen 35050), penicillin, streptomycin (100xPen/strep, Gibco/ Invitrogen 15070-063) at 37 °C and 5 % CO2. The SU-DHL-4 and OCI-LY-1 cells were seeded 24 h prior to treatment in 24 well plates allowing them to reach a density of 5x104 cells per mL upon treatment. Cells were exposed to 10 mg L-1 of CV, 5×10-3 M of KSP, and a mixture of CV and KSP of equal volume (prepared 90 min before). Counting of cells using trypan blue was performed at 4, 8, and 24 h intervals. The experiments were done in quadruplicate.
Results and discussion
Order with respect to crystal violet
Crystal violet shows a strong absorption band at 590 nm and follows Beer's law for low concentrations (2-8 mg/L). The relation found between the absorbance A590 and CV concentration in mg L-1 is A590 = 0.18×[CV] (R2: 0.99). The discoloration is faster with the increase in persulfate concentration. In the first step of degradation (until 700 s), the decrease in absorbance (A) at 590 nm is accompanied with an increase of absorbance at 468 nm. The overlay of spectra showed an isobestic point at 496 nm (as shown in Fig. 1a).
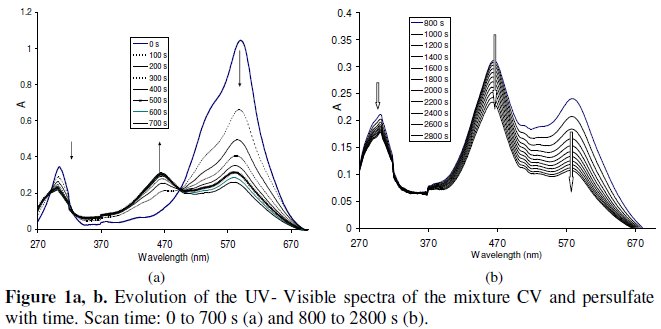
The solution turns green - yellow. After a time, the absorbance at 468 nm begins to decrease and that at 307 and 590 nm continues its decrease (Fig.1b). After 110 min the solution became transparent.
The plot of A, Ln A and 1/A vs. time, let us concluded that the best pseudo order with respect to crystal violet is 2 (according to the R2 value) (as shown in Fig. 2a).

kobs (mg-1 L s-1) is the slope of the plot LnA or 1/A vs. time (kobs â kapp). For all the experiments concerning the determination of order with respect to CV, the correlation coefficient (R2) for second order is closer to one (in the range 0.998 - 0.999) than that for first order (in the range 0.957-0.980). Order two is confirmed by differential method and by modeling the function A590 vs. time. The rate constant Kobs was evaluated by linear and non linear regression using Excel and Origin 7.0 programs. The correlation coefficients for first and second order in the case of MG are very close to each other, but the slope obtained from the differential method allows us to select the first order.
The discoloration order mentioned in literature for CV and MG is one [26, 38]. The difference in order with respect to CV - between the published result and that obtained in the present work - may be due to the condition of the experiment such as [persulfate]/[CV], activation by UV light.
The degradation of malachite green with persulfate is accompanied by a decrease in the three bands at 620, 432, and 318 nm and an increase in the region between 450 and 500 nm. Also the decrease in absorbance at 432 nm is accompanied by a shift to 468 nm. The solution became clear with time. The degradation products are already determined by LC-ESI-MS analysis [38] and the degradation mechanism is not the object of this project, but according to the evolution of UV - visible spectra, we can say that in CV and MG, the first intermediate degradation product is triphenyl-carbocation of yellow color (l : 445 nm) (total deamination) [46] followed by degradation of triphenyl-carbocation. The degradation of high concentration of CV (> 500 mg/L) gave tiny black particles with very low solubility in many organic solvents (polar or nonpolar) and aqueous yellow color with maximum at 460 nm (insoluble in CH2Cl2). Probably the precipitate corresponds to a salt of triphenyl-carbocation.
Effect of CV concentration
The rate constant increases linearly with the decrease in CV (or MG) concentration (as illustrated in Fig. 2b). The rate of discoloration increases with the decrease in the number of species to be oxidized. The same results are obtained with the systems MG/O3 [17], MG/persulfate [38] and CV/UV/H2O2 [36], but different from those obtained in ref. 36.
Order with respect to persulfate
The pseudo order with respect to persulfate is determined by keeping the concentration of CV constant and varying the concentration of persulfate in the medium. On increasing the concentration of persulfate, more reactive radicals could be generated and the rate of CV or MG degradation could become faster. Similar trend was observed in the degradation of MG with persulfate [38, 39] and CV /UV/ persulfate (H2O2) [36]. The pseudo order with respect to persulfate is one for both dyes since there is linear relation between the observed rate constant (kobs) and the concentration of persulfate (R2: 0.97) (as shown in Fig. 3).
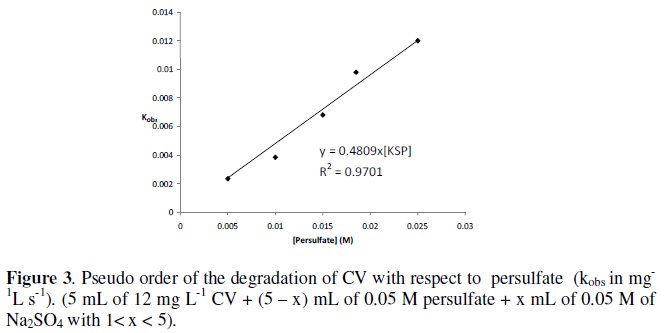
For the degradation of MG with persulfate, the order with respect to persulfate obtained by Jonnalagada is half [39]. This order is different from that obtained in the present work.
Effect of pH
Crystal violet has acid-base property. The pKa's for the loss of the two protons are approximately 1.15 and 1.8 [47]. At pH 1.0, the dye is green with absorption maxima at 420 nm and 620 nm, while in neutral pH, the dye is violet with an absorption maximum at 590 nm. The different colors are a result of the different charged states of the dye molecule. At neutral pH both extra protons are lost to the solution leaving only one of the nitrogen atoms positive charged.
In alkaline solution and in very acidic solution, discoloration of CV and MG occurred due to the nucleophilic attack of the central carbon [48], so the zone of pH undertaken for this study is limited to 2 < pH < 9. The oxidation of CV with persulfate at several pH does not affect only the rate constant, but affects also the intermediate products of the oxidation. In H3PO4 the evolution of CV spectra is approximately similar to those in distilled water (as shown in Fig. 4a).
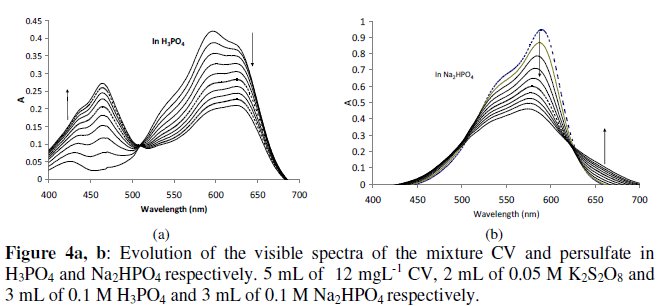
The intermediate and the final products are probably similar in both cases, but the reaction is faster in H3PO4 (kobs in H3PO4 ~ 2 kobs in H2O).
The increase of the rate constant in acidic medium was also reported in ref. 34 and 36. In H3PO4, the isobestic point shifted little to right (510 nm instead of 496 nm in water), but the band at 590 nm became wider than in distilled water.
In presence of NaH2PO4 or K2HPO4, the isobestic point is located at 626 nm and the increase in absorbance is observed in another zone (> 630 nm) (as shown in Fig. 4b). The intermediate products of the oxidation of CV in K2HPO4 are different from those in acidic medium or in distilled water. The rate constants in basic medium (NaH2PO4 or K2HPO4) are little smaller than those in water.
Matrix effect
Salt effect
The rate constant kobs decreases exponentially with the increase in KCl or KBr concentrations (R2: 0.99). The decrease is more pronounced with KBr than with KCl. The results are shown in Fig. 5a.
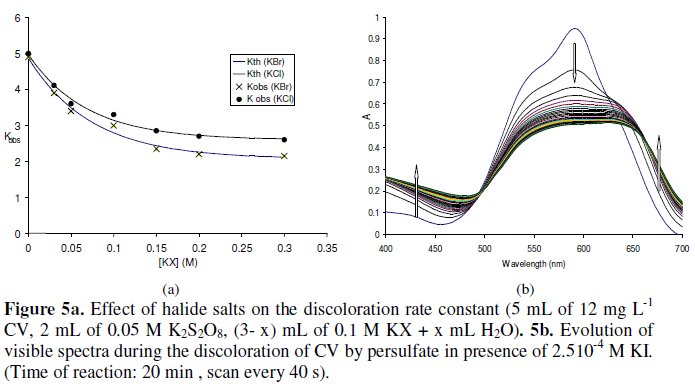
The decrease in rate constant is in accordance with that obtained during the degradation of Orange G with the persulfate/Fe2+ reagent [37]. The higher ionic concentration in the solutions, the lower reaction rate of CV and MG degradation occurred. It might result from a decreased amount of sulfate radicals due to the high ionic concentration. There is linear relation between kobs and I1/2 or I1/2/(1+I1/2) with R2 equal to 0.94 and 0.97 respectively, supporting the idea of primary kinetic salt effect. No reaction occurred between persulfate and KCl or KBr as in the case with KI. The presence of KCl or KBr did not affect the evolution of the visible spectrum with time (isobestic point remained at ~500 nm and appearance of new band at 468 nm).
Iodine can inhibit or catalyst some redox reactions. For example, it catalyses the reaction between Rhodamine B and KIO4 [49] but inhibits it with KBrO3 [50]. In presence of KI, persulfate reacts with iodide to produce iodine slowly (increase of absorbance between 400 nm and 450 nm (as indicated in Fig. 5b). The reaction between persulfate and iodide is faster than with persulfate and CV, preventing the direct reaction between CV and persulfate. The presence of iodine even in low concentration (2.5x10-4 M) changes the spectrum of CV significantly (λmax became 564 nm instead of 590 nm). As the absorbance of iodine solution is negligible at wavelength higher than 600 nm, the increase of absorbance above this wavelength is probably due to an interaction between CV and I3- as was the case with Rhodamine 6 G [51]. The addition of iodine to CV solution directly (in the absence of persulfate) gave the same spectral behavior. Iodine inhibits the degradation of CV by persulfate dramatically, even in low concentrations. The pseudo order with respect to CV in the presence of iodine is not a simple order (no linearity in LnA and 1/A plots vs. time). The decrease in A564 vs. time (t) followed the relation A = A0 + e-ct.
Metal effect
The addition of Cu2+, Ni2+, and Co2+ to the reactional mixture did not affect the rate constant, but the addition of Ag+ increases strongly the rate constant. Fig. 6a presents the variation of the rate constant as a function of Ag+.
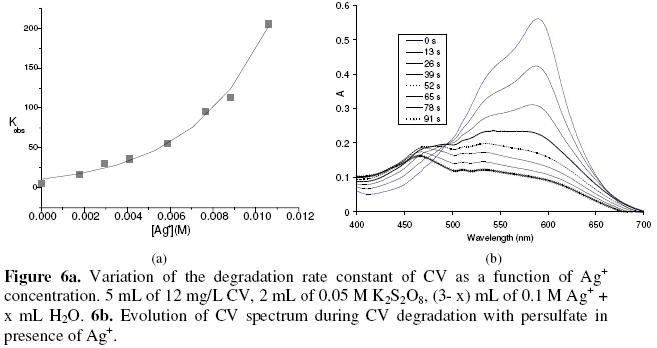
There is an exponential relation between Kobs and Ag+ concentration (R2: 0.99). The spectra did not show an isobestic point as in absence of silver, but the behavior in the region between 400 and 500 nm remained the same: at the beginning the absorbance increases, then decreases with time (as shown in Fig. 6b). The rate constant of the degradation of MG with persulfate increases linearly with Ag+ concentration. The variation of the rate constant in function of Ag+ concentration is more pronounced with CV than with MG.
Concerning, the addition of ferrous salt: at first, the rate constant increases little to reaches a maximum, for a concentration of Fe2+ equal to 0.003 M then decreases linearly with the increase of Fe2+ concentration (as shown in Fig. 7).
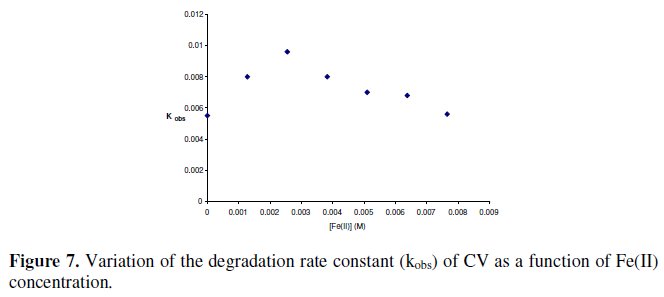
The main reason for the decrease of kobs at higher Fe2+ concentration is due to the fact that sulfate radical is scavenged by the excessive Fe2+ according to Fe2+ + SO4-* → Fe3+ + SO42-. A similar result is observed during the oxidation of other compounds with persulfate [25, 26, 52]. The matrix effect (salt and metal) on MG degradation is similar to that observed with CV.
Surfactant effect
The addition of surfactants to the reactional mixture may change the pathway reaction and may vary (positively or negatively) the rate constant of some reaction [15, 40, 53]. The results obtained in the present case show that the addition of anionic surfactant such as sodium dodecyl sulfate (SDS) decreases the rate constant even at low concentrations of SDS. The discoloration is inhibited with the increase of SDS concentration in the medium.
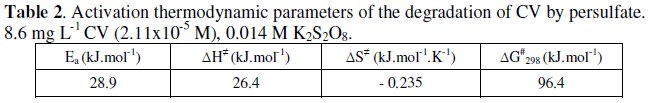
Effect of temperature
The increase in temperature increases the discoloration rate of CV. The activation parameters associated with the discoloration are calculated as follow: plot of ln Kobs vs. 1/T gives the value of the activation energy (Ea), according to Arrhenius equation:
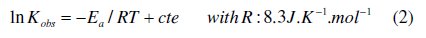
The ΔH‡ and ΔS‡ values can be calculated from Eyring plot:
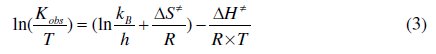
where kB = Boltzmann's constant (1.381x10-23 J K-1), h = Plank's constant (6.626×10-34 J s) and ln (kB/h) = 23.76.
The free activation enthalpy ΔG‡ is equal to:

The linear equation of ln (kobs) vs. 1/T is -3480/T + 6.47 (as shown in Fig. 8).
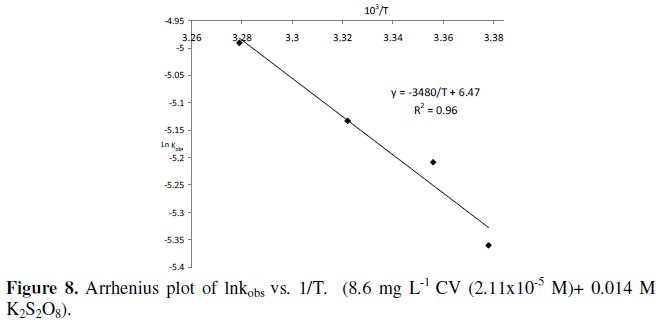
The activation energy and the other kinetics parameters in the range of temperature studied (23 °C -32 °C ) are listed in Table 2.
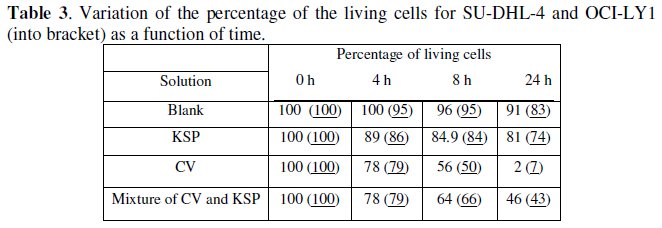
Cytotoxic Study
Cell counting of SU-DHL-4 revealed that CV being highly cytotoxic induced almost 100 % of cell death after 24 h. Altough KSP had slight cytotoxicity killing 10 % of the SU-DHL-4 cells after 24 h. Interestingly, the mixture of CV and KSP had intermediate cytotoxicity, killing 40 % of the cells after 24 h. The results are given in Table 3. The pretreatment of CV with KSP protected cells by about 60 % from the cytotoxic CV effect. These results showed that the degradation products of CV by KSP were less toxic than CV. Similar results were observed with OCI-LY-1 cells.
Conclusions
The degradation of CV is pseudo second order and pseudo first order with respect to CV and persulfate. The discoloration increases with temperature but decreases exponentially with the addition of halide salts: the order of inhibition is as follow: KCl < KBr << KI. There was no effect on the rate constant upon addition of Cu2+, Ni2+, or Co2+ , but an important increase is observed after addition of Ag+ . Anionic surfactant (SDS) also inhibits the degradation rate of CV. The general behavior of CV is similar to that of MG with only one difference concerning the order of degradation of MG (one instead of two). The toxicity of CV is reduced after degradation with K2S2O8.
References
1. T.C. Chen, E.J Day, Poultry Sci. 53 (1974)1791.
2. C.L. Hall, P.B. Hamilton, Poultry Sci. 6 (1982) 62. [ Links ]
3. D.J. Alderman, J. Fish Disc. 8 (1985) 289. [ Links ]
4. S.J. Culp, F.A. Beland, J. Am. College Toxicol. 15 (1996) 219. [ Links ]
5. M. Panizza, A. Barbucci, R. Ricotti, G. Cerisola, Sep. Purif. Technol. 54 (2007) 382. [ Links ]
6. I. Alameddine, M.M. El Jamal, J. Univ. Technol. Metall. (Sofia) 44 (2009) 127. [ Links ]
7. R.E. Palma-Goyes, F.L. Guzman-Duque, G. Penuela et all, Chemosphere 81 (2010) 26. [ Links ]
8. J.M. Peralta-Hernandez, M. Mendez-Tovar, R. Guerra-Sanchez et all, Hindawi, Publishing Corporation International J. Electrochem. Vol. 2012, Article ID 154316. [ Links ]
9. S. Randorn, Wongnawa and P. Boonsin, Science Asia 30 (2004) 149.
10. L.A. Perez-Estrada, A. Aguera, M.D. Hernando et all, Chemosphere 70 (2008) 2068. [ Links ]
11. J.C. Edwards, Master of Sciences, in Env. Sci. and Eng., Virginia Polytechnic Institute and State Univ. 2000, Master Investigation of Color Removal by Chemical Oxidation (H2O2) for Three Reactive Textile Dyes and Spent Textile Dye Wastewater. [ Links ]
12. M. Rafatullah, O. Sulaiman, R. Hashima, A. Ahmad, J. Hazard. Mat. 177 (2010) 70. [ Links ]
13. B.R. Babu, A.K. Parande, S. Raghu, T.P. Kumar, J. Cotton Sc.11 (2007) 141. [ Links ]
14. S.J. Allen, B. Koumanova, J. Univ. Technol. Metall. (Sofia) 4 (2005) 175. [ Links ]
15. A.H. Gemeay, G.R. El-Ghrabawy, A.B. Zaki, Dyes Pigments 73 (2007) 90. [ Links ]
16. Y. Mohammed, A.C. Etonihu, V.A. Tsaku, Trakia J. Sci. 9 (2011) 1. [ Links ]
17. E. Kusvuran, O. Gulnaz, A. Samil, O. Yildirim, J. Hazard. Mat. 186 (2011) 133.
18. H.-J. Fan, S.-T. Huang, W.-H. Chung et all, J. of Haz. Mat. 171 (2009) 1032. [ Links ]
19. B.H. Hameed, T.W. Lee, J. Haz. Mat. 164 (2009) 468. [ Links ]
20. M.A. El-Ries, E. Khaled, F.I. Zidane et all, Drug Testing and Analysis 4 (2012) 129. [ Links ]
21. K. Huang, Z. Zhao, G.E. Hoag et all, Chemosphere 61 (2005) 551. [ Links ]
22. T.K. Lau, W. Chu, N.J.D. Graham, Environ. Sci. Technol. 41 (2007) 613. [ Links ]
23. N. Daneshvar, D. Salari, A.R. Khataee, J. Photochem. Photobiol. A 162 (2004) 317. [ Links ]
24. Y.-T. Lin, C. Liang, J.-H. Chen, Chemosphere 82 (2011) 1168. [ Links ]
25. S.Y. Oh, H.W. Kim, J.M. Park et all, J. Haz. Mat. 168 (2009) 346. [ Links ]
26. Y. Ding, L. Zhu, J. Yan et all, J. Environ. Monit. 13 (2011) 3057. [ Links ]
27. D.C. Harris, Quantitative Chemical Analysis, 4th Edition, W.H. Freeman and Company, New York, 1995. [ Links ]
28. R.A. Couttenye, K.C. Huang, G.E. Hoag, and S.L. Suib, Prevention, Conference and Exposition, Atlanta, GA, USA, (2002) 345. [ Links ]
29. C. Liang, C.J. Bruell, Ind. Eng. Chem. Res. 47 (2008) 2912. [ Links ]
30. G.P. Anipsitakis, D.D. Dionysiou, Environ. Sci. Technol. 38 (2004) 3705. [ Links ]
31. M.H. Rasoulifard, S.M.M.D-Mohammadi, A. Heidari, E. Farhangni, TUBITAK, Turkish J. Eng. Env. Sci. 36 (2012) 73. [ Links ]
32. W.E Fristad, J.A. Klang, Tetrahedron Letters 24 (1983) 2219. [ Links ]
33. O.S. Furman, A.L. Teel, R.J. Watts, Environ. Sci. Technol. 44 (2010) 6423. [ Links ]
34. M.A. Miraglio, Master of Sci. in Env. Eng., Washington State Univ., Dept. of Civil and Env. Eng., 2009, Base-Activated Persulfate Treatment of Contaminated Soils With pH Drift From Alkaline to Circumneutral. [ Links ]
35. K. Djebbar, S. Aliouche, H. Chenini, T. Sehili, Desalination 250 (2009) 76. [ Links ]
36. S. Aliouche, S. Belaidi, K. Djebbar, T. Sehili, Sci. & Technol. A (27-B) (2008) 31. [ Links ]
37. X.-R. Xu, X.-Z. Li, Sep. Purific. Technol. 72 (2010) 105. [ Links ]
38. S. Gokulakrishnan, P. Parakh, H. Prakash, J. Haz. Mat. 213-214 (2012) 19. [ Links ]v
39. T. Mushinga, S.B. Jonnalagadda, Int. J. Chem. Kinetics. 24 (1992) 41. [ Links ]
40. Z. Chen, N. Zhang, L. Zhuo, B. Tang, Microchim. Acta 164 (2009) 311. [ Links ]
41. E. Kjanghel, Y. Pervez, J. Sci. Ind. Res. 68 (2009) 940. [ Links ]
42. L. Chen, W. Jin-Hua, L. Ping et all, Water Sci. Technol. 64 (2011) 754. [ Links ]v
43. J. Deng, N. Carlson, K. Takeyama et all, Cancer Cell 12 (2007) 171. [ Links ]
44. H.F. Dietzfelbinger, A. Lang, D. Oberberg et all, Exp. Hematol. 20 (1992) 178. [ Links ]
45. S. Kiesel, R. Haas, G. Moldenhauer et all, Leuk. Res. 11 (1987) 1119. [ Links ]
46. http://www.demochem.de/chembox_trph-e.htm, Computer-Interfaced Exp., P. Keusch, Microscale Proj. Exp. Light Absorbtion of Triphenylmethylium Salts. [ Links ]
47. E.Q. Adams, L. Rosenstein, J. Am. Chem. Soc. 36 (1914) 1452. [ Links ]
48. M.A. El Haj Hasan, L. Fayoumi, M.M. El Jamal, J. Univ. Chem. Technol. & Metall. (Sofia) 46 (2011) 221. [ Links ]
49. J.P. Tu, Food Sci. (Chin.) 23 (2002) 114. [ Links ]
50. X. Zhu, Y. Zhang, Spectrochimica Acta Part A 70 (2008) 510. [ Links ]
51. L.H. Chen, Chin. J. Anal. Chem. 25 (1997) 937. [ Links ]
52. C. Liang, C.J. Bruell, M.C. Marley, K.L. Sperry, Chemosphere 55 (2004) 1213. [ Links ]
53. S. Pande, S. K. Ghosh, S. Nath et all, J. Colloid Interface Sci. 299 (2006) 421. [ Links ]
Acknowledgements
I would like to thank Dr. Iman Haidar for her help.
*Corresponding author. E-mail address: mjamal@ul.edu.lb
Received 12 June 2012; accepted 17 °C tober 2012














