Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Portugaliae Electrochimica Acta
versão impressa ISSN 0872-1904
Port. Electrochim. Acta vol.29 no.6 Coimbra nov. 2011
https://doi.org/10.4152/pea.201106429
Corrosion Inhibition by an Aqueous Extract of Phyllanthus Amarus
M. Sangeetha1,*, S. Rajendran1,2, J. Sathiyabama1, A. Krishnaveni3, P. Shanthy3, N. Manimaran3, B. Shyamaladevi4
1 PG and Research Department of Chemistry, GTN Arts College, Dindigul - 624005, Tamil Nadu, India
2 Servite College of Education for Women, Thogaimalai - 621313, Tamil Nadu, India
3 Department of Chemistry, Yadava College, Madurai - 625014, Tamil Nadu, India
4 Department of Chemistry, Vivekanandha Institute of Engineering and Technology for Women, Elayampalayam, Tiruchengode, Tamil Nadu, India
doi: 10.4152/pea.201106429
Abstract
The inhibition efficiency (IE) of phyllanthus amarus extract (PAE)-Zn2+ system, in controlling corrosion of carbon steel in an aqueous solution containing 60 ppm of Cl-, has been evaluated by weight loss method. Weight loss study reveals that the formulation consisting of 2 mL of PAE and 25 ppm of Zn2+ has 98% inhibition efficiency in controlling corrosion of carbon steel immersed in an aqueous solution containing 60 ppm of Cl-. Synergistic parameters suggest that a synergistic effect exists between PAE and Zn2+. Polarization study reveals that this system functions as mixed type of inhibitor controlling the cathodic reaction and anodic reaction to an equal extend. AC impedance spectra reveal that a protective film is formed on the metal surface. The FTIR spectra reveal that the protective film consists of Fe2+-phyllanthus complex.
Keywords: corrosion inhibition, carbon steel, green inhibitor, environmentally friendly inhibitor, phyllanthus amarus.
Introduction
A number of heterocyclic compounds [1-3] have been reported as corrosion inhibitors and the screening of synthetic heterocyclic compounds is still being continued. Several other inorganic inhibitors such as zinc, chromate, polyphosphate, and nitrite were used as corrosion inhibitors. Also, molybdate, phosphates, phosphonocarboxylic acids and polymers were used as inhibitors, along with metal ions such as Zn2+. Though many synthetic compounds showed good anticorrosive activity, most of them are highly toxic to both human beings and environment. These toxic effects and ecological problems associated with the discharge of such materials have resulted in the development of other efficient and environmentally acceptable inhibitors. Hence the recent trend is the search for environmental friendly inhibitors. Most of the natural products are non toxic, bio degradable and readily available in plenty. Various parts of the plants - seeds, fruits, leaves, flowers, etc., have been used as corrosion inhibitors. Several studies have been published on the use of natural products as corrosion inhibitors [4, 5]. Tannins are being used for protection of steel against corrosion in cooling water systems and in paints [6-8]. The biocidal and inhibitive effects of Azadirachta Indica have been studied on mild steel in fresh water [9]. The scale inhibition efficiencies of the aqueous extracts of plant materials namely Cordia latifolia, Eucalyptas and Jasmine auriculatum have been evaluated [10]. Corrosion inhibition by an aqueous extract of turmeric rhizome powder on carbon steel has been investigated by means of weight-loss, electrochemical polarization and impedance studies [11]. Corrosion inhibition has been studied with Eugenia jambolans [12], Androgaphis paniculata [13], Acacia Arabica [14]. Kliskic et al. have used the first neutral phenol sub fraction of the aqueous extract of rosemary leaves as corrosion inhibitor for the Al-Mg alloy in a 3% NaCl solution at 298 K [15]. Lawsonia extract has been used to inhibit corrosion of metals [16]. Corrosion inhibition of iron in hydrochloric acid solutions by naturally occurring henna has been investigated [17]. The corrosion inhibition efficiency of a caffeine-Zn2+ system on mild steel immersed in an aqueous solution containing 60 ppm of chloride was investigated by weight loss study [18]. Aqueous extract of kalmegh (Andrographis paniculata) leaves has been used as green inhibitor [19]. Eco-friendly corrosion inhibitor - garcinia kola - has been used to prevent corrosion of mild steel in H2SO4 solutions [20]. Inhibitory effects of ocinum tenuiplorum (Tulsi) have been investigated [21]. Anti-corrosive effect of raphia hookeri exudates gum-halide mixtures has been studied [22]. Ethanol extract of phyllanthus amarus green inhibitor has been used to prevent the corrosion of mild steel in H2SO4 [23]. Terminalia catappa extract has corrosion inhibitive properties [24].
There are several reviews on the use of plant extracts as corrosion inhibitors [25]. Recently, aqueous extract of cocos nucifera -coconut palm - petiole [26], fennel (foeniculum vulgare) essential oil [27], pericarp of the fruit of garcinia mangostana [28], natural fenugreek [29], ethanol extract of vernonia amygdalina[30] and ipomoea involcrata [31], have been used as corrosion inhibitors.
Phyllanthus amarus plant is bitter, febrifuge and antiseptic. It is useful in dropsy, jaundice, diarrhea, dysentery, intermittent fevers, diseases of urino-genital system, scabies ulcers and wounds.
The major ingredients of phyllanthus amarus are phyllanthin, hypophyllanthin, phyllanthusiin D, amarin, amarulone and amarinic acid [32,33]. Structure of phyllanthusiin D is shown in scheme 1.
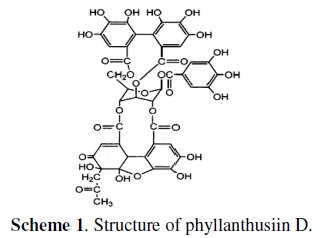
All of them have the following functional groups in common: -OCH3, C=O, OH, ring oxygen and conjugated double bonds.
The present work is under taken (i) to evaluate the inhibition efficiency (IE) of an aqueous extract of phyllanthus amarus in controlling the corrosion of carbon steel in an aqueous solution containing 60 ppm of Cl-in the presence and absence of Zn2+ ; (ii) to examine the influence of biocides such as N-cetyl-N,N,Ntrimethylammonium bromide (CTAB) and sodium dodecyl sulphate (SDS), and the influence of pH and duration of immersion on the IE of the phyllanthus amarus extract; (iii) to analyse the protective film formed on the carbon steel by FTIR and UV-visible spectra; (iv) to understand the mechanistic aspects of corrosion inhibition by potentiodynamic polarization studies and AC impedance analysis.
Experimental
Preparation of phyllanthus amarus extract
An aqueous extract of phyllanthus leaves was prepared by grinding 10 g of shade dried phyllanthus leaves, with distilled water, filtering the suspending impurities, and making up to 100 mL. The extract was used as corrosion inhibitor in the present study.
Preparation of the specimens
Carbon steel specimens (0.026% S, 0.06% P, 0.4% Mn, 0.1% C and rest iron) of the dimensions 1.0 × 4.0 × 0.2 cm were polished to a mirror finish, degreased with trichloroethylene, and used for the weight-loss method and surface examination studies.
Weight -loss method
Carbon steel specimens were immersed in 100 mL of a solution containing 60 ppm of Cl-and various concentrations of the inhibitor in the presence and absence of Zn2+ for a period of 1 day. The weights of the specimens before and after immersion were determined using a balance, Shimadzu AY62 model. The inhibition efficiency (IE) was then calculated using the equation
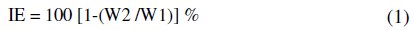
where W1 = corrosion rate in absence of the inhibitor; W2 = corrosion rate in presence of the inhibitor.
Surface examination study
The carbon steel specimens were immersed in various test solutions for a period of one day. After one day, the specimens were taken out and dried. The nature of the film formed on the surface of the metal specimen was analyzed by surface analysis techniques, such as FTIR and UV-visible reflectance spectroscopy, namely, fluorescence spectroscopy.
Synergism parameters
Synergism parameters are indications of the synergistic effect existing between two inhibitors.
Synergism parameters were calculated using the relation

where Θ1+2 = (Θ1+Θ2) -(Θ1 × Θ2), being Θ1 = surface coverage of substance 1 and Θ2 = surface coverage of substance 2. Θ'1+2 = combined surface coverage of substances 1 and 2. Θ = surface coverage = IE% / 100.
Analysis of variance (F-Test)
F-test was carried out to investigate whether the synergistic effect between CAE and Zn2+ was statistically significant.
Potentiodynamic polarization study
This study was carried out using a CHI 660A electrochemical impedance analyzer model. A three -electrode cell assembly was used. The working electrode used was carbon steel with 1 cm2 exposed area. A saturated calomel electrode (SCE) was used as reference electrode. A rectangular platinum foil was used as the counter electrode. Polarization curves were recorded after doing iR compensation. The parameters such as Tafel slopes, Icorr and Ecorr were calculated.
AC impedance measurements
A CHI 660A electrochemical impedance analyzer model was used to record AC impedance measurements. The cell set up was the same as that used for polarization measurements. The real part (Z') and imaginary part (Z'') of the cell impedance were measured in ohms for various frequencies. The Rt (charge transfer resistance) and Cdl (double layer capacitance) values were calculated.
UV-visible spectra
UV-visible spectra were recorded in an UV spectrod S-100 Analytic Jena spectrophotometer.
FTIR spectra
These spectra were recorded with a Perkin-Elmer 1600 spectrophotometer. The FTIR spectrum of the protective film was recorded by carefully removing the film mixing it with KBr and making the pellet.
Results and discussion
Analysis of results of weight-loss study
The inhibition efficiency (IE) of an aqueous extract of phyllanthus amarus, in controlling corrosion of carbon steel in an aqueous solution containing 60 ppm Cl-has been evaluated by weight loss method. The results are given in Table 1.
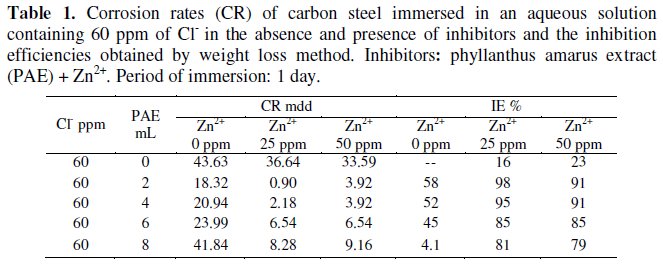
It is seen from Table 1 that when the carbon steel is immersed in aqueous solution containing 60 ppm of Cl-, the corrosion rate is 43.63 mdd. When 2 mL of phyllanthus amarus extract (PAE) are added, the corrosion rate is reduced to 18.32 mdd and the IE is found to be 58%. Upon addition of various concentrations (2, 4, 6, and 8 mL) of PAE, IE decreases. This is due to the fact that the complex (Fe2+-active principle in PAE) formed on the metal surface dissolves and goes into solution. Similar observation has been made in the case of corrosion inhibition by Henna extract [34]. It is seen from Table 1 that when zinc is added to the PAE, the IE increases to a great extent. For example, when 25 ppm of Zn2+ are added to 2 mL of PAE, the IE increases from 58% to 98%. This suggests that a synergistic effect exists between Zn2+ and the active principles present in PAE. However, it is observed that when the concentration of Zn2+ increases from 25 ppm to 50 ppm, the IE slightly decreases. This may be due to the fact that, when the concentration of Zn2+ increases, the Zn2+-PAE complex formed is precipitated in the bulk of the solution. Hence PAE is not transported towards the metal surface. So the IE decreases.
Influence of duration of immersion on the inhibition efficiency of PAE Zn2+ system
The influence of duration of immersion on the inhibition efficiency of the Zn2+ phyllanthus system is given in Table 2.
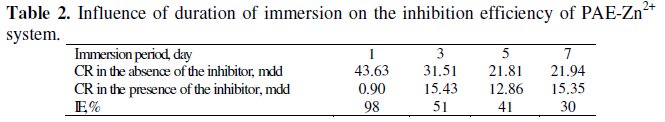
It is observed that as the duration of immersion increases, the inhibition efficiency decreases. This is due to the fact that as the duration of immersion increases, the protective film formed on the metal surface is not able to withstand the attack of Cl-. The film is broken and hence the IE decreases. Similar observation was made with Fe2+ curcumin system [11], Fe2+-Henna leaves system [34] and pentanesulphonic acid -Zn2+ system [35].
Influence of sodium dodecyl sulphate (SDS) on the IE of PAE-Zn2+ system
It is observed from Table 3 that as the concentration of SDS increases, the IE increases and then decreases and again increases. SDS is an anionic surfactant. It has biocidal activity also [36].
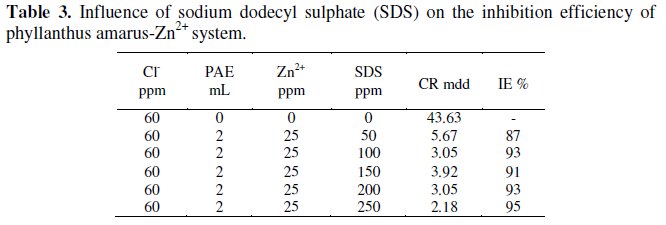
A micelle would have been formed at the minimum efficiency concentration. When more amount of SDS is added, SDS exists as monomer. These monomers are easily adsorbed on the metal surface. A protective film is formed. This prevents corrosion of metal [37]. It is observed that the formulation consisting of 2 mL of PAE 25 ppm of Zn2+ and 250 ppm of SDS has 95% corrosion IE. The biocidal nature of SDS is known [36]. Hence, if the biocidal study of the system (PAE-Zn2+-SDS) is established in future, this formulation may find application in cooling water system, where corrosion of the metal is caused by aggressive ions and also by micro organisms present in cooling water.
Influence of N-Cetyl N, N, N-trimethyl ammonium bromide (CTAB) on the inhibition efficiency of PAE -Zn2+ system
The influence of (CTAB) on the corrosion rates of carbon steel containing Zn2+ +PAE is tabulated in Table 4.
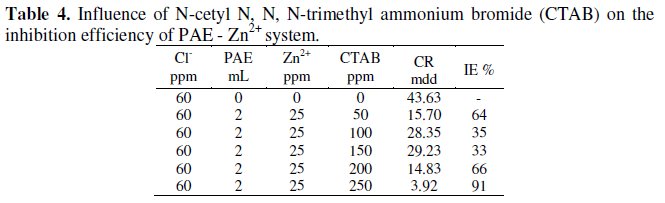
When various concentrations of N-cetyl-N,N,N-trimethylammonium bromide (CTAB) are added to the inhibitor system, the inhibition efficiency decreases and reaches a minimum, and then increases. A micelle would have been formed at the minimum efficiency concentration [37, 38]. Afterwards the micelles would have been converted into monomers, which improved the inhibition efficiency. When more amount of CTAB is added, CTAB exists as monomer; these monomers are easily adsorbed on the metal surface. A protective film is formed. This prevents corrosion of metal. This can be explained as stated in the case of SDS.
Influence of pH on IE of phyllanthus amarus-Zn2+ system
It is seen from Table 5 that at pH 7, the phyllanthus amarus (PAE) (2 mL)-Zn2+ (25 ppm) (60 ppm) Cl-system has 98% IE.
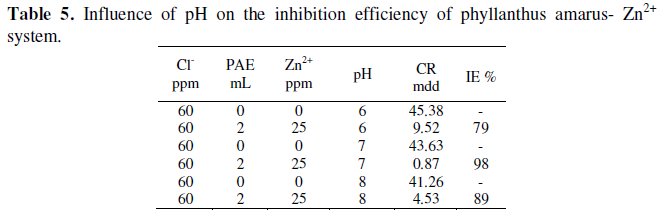
When pH is lowered to 6 by addition of dilute sulphuric acid, the IE decreased to 79%. This is due to the fact that when the acid is added the protective film is broken by the aggressive H+ ion present in the acid. When the pH is increased to 8 by addition of diluted sodium hydroxide solution, the IE increased to 89% (when compared to an acidic medium). This is due to the fact that the phenolic -OH groups would have been ionized to phenolate anion, -O- Na+. This helped anchoring of phenolic -O- on the anodic sites of the metal surface effectively and hence IE increased at higher pH values. Similar observation has been observed in the case of corrosion inhibition by curcumin extract and by henna extract: as the value of pH is increased the corrosion inhibition efficiency also increased [34,35]. However this 89% IE in basic medium (pH 8) is lower than the IE of 95% in neutral medium. This is due to the fact when NaOH is added, a portion of Zn2+ is precipitated as Zn(OH)2 in the bulk of the solution. Hence PAE is not effectively transported to the metal surface and hence the IE slightly decreases.
Synergism parameters (SI )
Synergism parameters have been used to confirm the synergistic effect existing between two inhibitor systems [34,39]. Synergism parameters have been calculated for PAE-Zn2+ (25 ppm and 50 ppm) systems. The results are given in Tables 6 and 7.
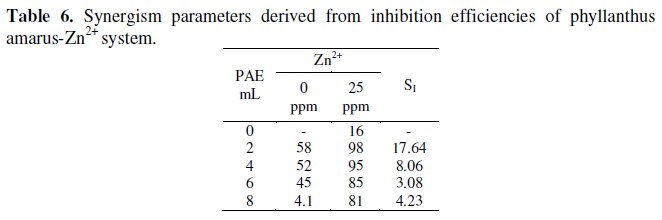
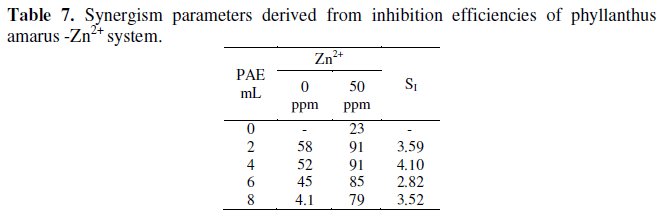
It is observed that the synergism parameters are greater than 1.
This confirms the synergistic effect existing between PAE-Zn2+ . It is also interesting to note that the values of SI are slightly smaller in the case of 50 ppm of ZnZn2+ when compared with 25 ppm of Zn-Zn2+. This is in agreement with the inhibition efficiencies obtained by weight loss method. Thus the values of synergism parameters give a quantitative value of synergism existing between the two inhibitors.
Analysis of F-values (analysis of variance ANOVA)
Analysis of variance has been used to established if the synergistic effect existing the between two inhibitor systems is statistically significant [40,41]. The F-value calculated for PAE-Zn2+ (25 ppm) system is 60.68 (Table 8).
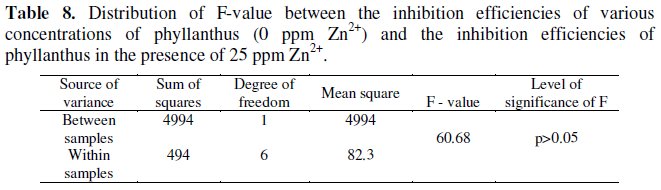
This is greater than the critical F-value (5.99) for 1,6 degrees of freedom at 0.05 level of significance. Hence it is concluded that the synergistic effect existing between 25 ppm of Zn2+ and various concentrations of PAE is statistically significant.
The F-value calculated for PAE-Zn2+ (50 ppm) system is 13.93 (Table 9).
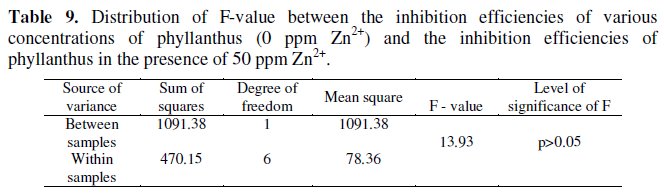
This is greater than the critical F-value (5.32) for 1,6 degrees of freedom at 0.05 level of significance. Hence it is concluded that the synergistic effect existing between 50 ppm of Zn2+ and various concentrations of PAE is statistically significant.
Analysis of polarization curves
Polarization study has been used to detect the formation of protective film on the metal surface [42-49]. When a protective film is formed on the metal surface, the linear polarization resistance (LPR) increases and the corrosion current (Icorr) decreases. The potentiodynamic polarization curves of carbon steel immersed in various test solutions are shown in Fig.1(a) and (b).
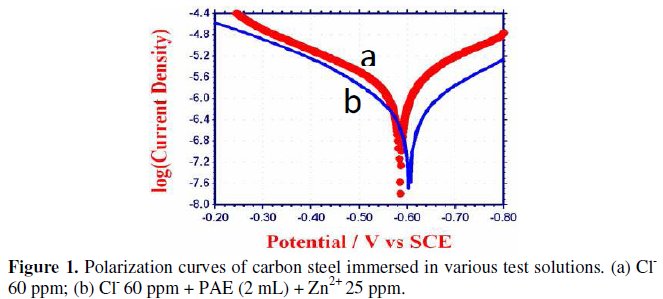
The corrosion parameters, namely corrosion potential (Ecorr), Tafel slopes (bc = cathodic; ba = anodic), linear polarization resistance (LPR) and corrosion current (Icorr), are given in Table 10.

When carbon steel is immersed in an aqueous solution containing 60 ppm Cl-, the corrosion potential is -586 mV vs. SCE. The formulation consisting of 2 mL of phyllanthus amarus extract (PAE) and 25 ppm of Zn2+ shifts the corrosion potential to -604 mV vs. SCE. This suggests that the reaction is predominantly cathodically controlled.The LPR value increases from 23589 ohm cm2 to 67322 ohm cm2. This suggests that a protective film is formed on the metal surface.
Further the corrosion current decreases from 1.748 × 10-6 A cm-2 to 0.5071 × 10-6 A cm-2. The IE calculated from corrosion current is 71%. This value is lower than the IE obtained by weight loss method (98%). The discrepancy may be explained by the fact that in electrochemical process, the instantaneous corrosion current is measured. However, in the case of the weight loss method, IE is calculated after a long time. The protective film formed is strengthened as the duration of immersion increases.
In presence of inhibitors, the cathodic and anodic Tafel slopes are more or less equal. This indicates that the formulation consisting of PAE and Zn2+ functions as a mixed type inhibitor controlled by both the cathodic and anodic reactions to an equal extend. However the cathodic Tafel slope is slightly higher. This is reflected in the shift of corrosion potential to the cathodic side.
Analysis of AC impedance spectra
AC impedance spectra have been used to detect the formation of a film on the metal surface. If a protective film is formed, the charge transfer resistance increases and the double layer capacitance value decreases [42]. The AC impedance spectra of carbon steel immersed in various solutions are shown in Fig. 2 (Nyquist plot).
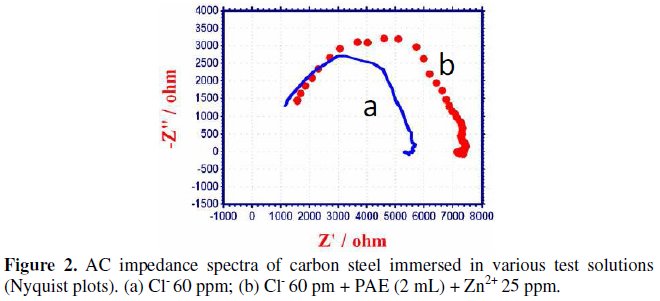
The AC impedance parameters, namely, charge transfer resistance (Rt) and double layer capacitance (Cdl) are given in Table 11.
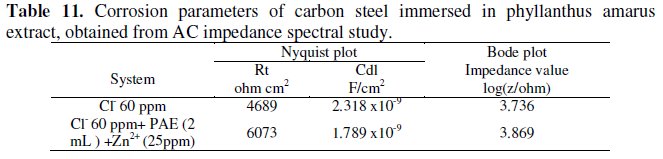
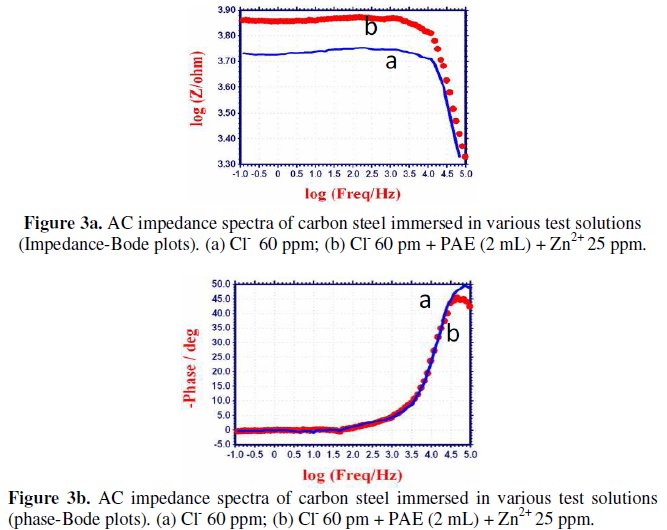
When carbon steel is immersed in aqueous solution containing 60 ppm Cl-, the Rt value is 4689 ohm cm2 and the Cdl value is 2.318 × 10-9 F cm-2. When PAE and Zn2+ are added, the Rt value increases from 4689 ohm cm2 to 6073 ohm cm2 and Cdl decreases from 2.318 × 10-9 F cm-2 to 1.789 × 10-9 F cm-2. This suggests that a protective film is formed on the surface of the metal. This accounts for the very high IE of PAE-Zn2+ system. Further there is increase in impedance, log(Z/ohm), value from 3.736 to 3.869 (derived from Bode plot Fig.3a, Fig.3b).
Analysis of UV-visible spectra
The UV-visible absorption spectrum of an aqueous solution of PAE and Fe2+ (freshly prepared FeSO4.7H2O solution) is shown in (Fig. 4).
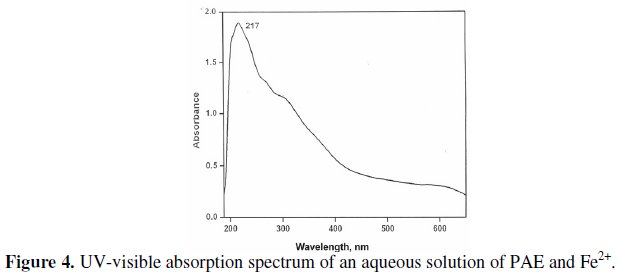
Peaks appear at 217, 260 and 310 nm. This is due to Fe2+-PAE complex formed in solution. The UV-visible reflectance spectrum of the film formed on the metal surface after immersion in a solution containing 60 ppm Cl-, 2 mL of PAE and 25 ppm of Zn2+, is shown in (Fig. 5).
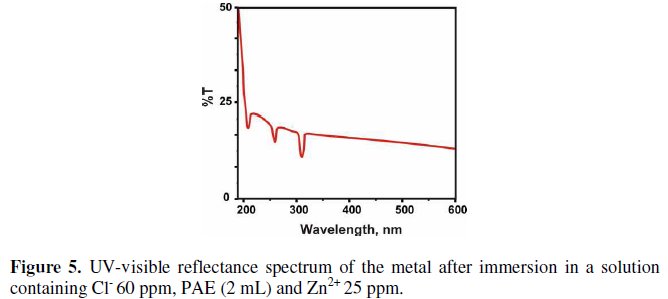
Peaks appear at 217, 260 and 310 nm. This matches with the Fe2+-PAE complex in solution. Hence it is confirmed that the protective film consists of Fe2+-PAE complex [50,51].
Analysis of FTIR spectra
The active principle in an aqueous extract of phyllanthus amarus is phyllanthusiin D (Scheme 1).
A few drops of an aqueous extract of phyllanthus were dried on a glass plate. A solid mass was obtained. Its FTIR spectrum is shown in Fig.6.
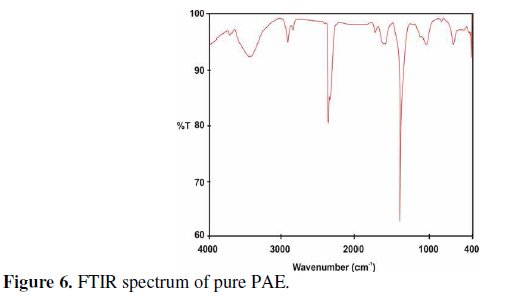
The C=O stretching frequency appears at 1606 cm-1. The OH stretching frequency appears at 3446 cm-1. The band due to conjugated double bonds appears at 3727 cm-1. The peak at 1112 cm -1 is due to ring oxygen atom. Thus the structure of phyllanthusiin-D is confirmed by FTIR spectrum [52].
The FTIR spectrum of the protective film formed on the surface of the metal after immersion in the solution containing 60 ppm of Cl-25 ppm of Zn2+, and 2 mL of phyllanthus extract is shown in Fig.7.
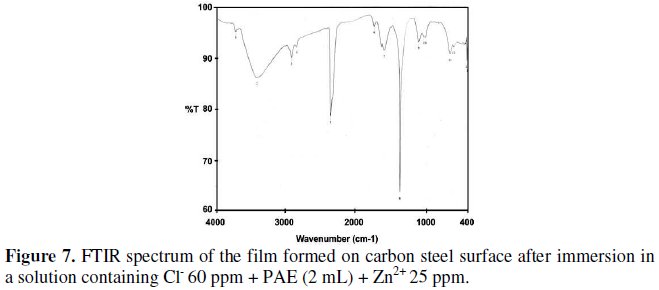
It is found that the phenolic -OH stretch has shifted from 3446 cm-1 to 3418 cm-1. The band due to conjugated double bonds shifts from 3727 cm-1 to 3726 cm-1. Ring oxygen atom stretching frequency has shifted from 1112 cm-1 to 1118 cm-1. The C=O stretching frequency has shifted from 1606 cm-1 to 1612 cm-1.
This confirms the presence of Fe2+-phyllanthus extract complex on the metal surface. Fe2+ has coordinated with the O-atom of the -OH group, C=O group and the ring oxygen atom. These shifts confirm the formation of Fe2+ -phyllanthus complex on the anodic sites of the metal surface. The formation of Fe3+ phyllanthus amarus complex cannot be ruled out. The peak at 1384 cm-1 is due to Zn(OH)2 formed on the cathodic sites of the metal surface [52,53].
Conclusions
The inhibition efficiency (IE) of phyllanthus amarus extract (PAE)-Zn2+ system in controlling corrosion of carbon steel in an aqueous solution containing 60 ppm of Cl-has been evaluated by weight loss method. The present study leads to the following conclusions.
Weight loss study reveals that the formulation consisting of 2 mL of PAE and 25 ppm of Zn2+ has 98% inhibition efficiency in controlling corrosion of carbon steel immersed in an aqueous solution containing 60 ppm of Cl-. Synergistic parameters suggest that a synergistic effect exists between PAE and Zn2+.
Polarization study reveals that this system functions as a mixed type inhibitor, controlling the cathodic reaction and anodic reaction to an equal extent. AC impedance spectra reveal that a protective film is formed on the metal surface.
The FTIR spectra reveal that the protecting film consists of Fe2+-phyllanthus amarus (active ingredient) complex.
References
1. F. Bentiss, M. Traisnel, M. Lagrenee, Corros. Sci. 42 (2000) 127. [ Links ]
2. S.T. Arab, E.A. Noor, Corrosion. 49 (1993) 122. [ Links ]
3. H. Luo, Y.C. Guan, K.N. Han, Corrosion 54 (1988) 721. [ Links ]
4. K. Srivastava, P. Srivastava, Br. Corros. J. 16 (1981) 221. [ Links ]
5. R.M. Saleh, A.A. Ismail, A.A. El Hosary, Br. Corros. J. 16 (1982) 131. [ Links ]
6. A.J. Saevell, J. Oil. Col. Chem. Assn. 61 (1978) 439. [ Links ]
7. E. Knowles, T. White, J. Oil. Col. Chem. Assn. 41 (1958) 10. [ Links ]
8. T.K. Ross, R.A. Francis, Corros. Sci. 18 (1978) 351. [ Links ]
9. M. Manimegalai, P. Rajeswari, S. Mohanam et all, ''Biocidal and Inhibition effect of Naturally occurring Substance-Azadiracta Indica in Fresh Water'', Proc. of 10th National Congress on Corrosion Control, held Sept. 6-8 (Karaikudi, India: National Corrosion Council of India (2000)) 153. [ Links ]
10. I.H. Farooqui, M.A. Quraishi, ''Breakthroughs in scale and deposit Control'', Proc. of Industrial Corrosion Causes and Mitigation CORCON 2000, vol.2, held Nov.20-23 (Mumbai, India: Quest publication (2000)) 103. [ Links ]
11. S. Rajendran, S. Shanmugapriya, T. Rajalakshmi, A.J. Amalraj, Corrosion 61 (2005) 685. [ Links ]
12. A.S. Verma, G.N. Mehta, Trans. SAEST. 32 (1988) 4. [ Links ]
13. G.O. Avwiri, F.O. Igho, Mater. Lett. 57 (2003) 3705. [ Links ]
14. A.S. Verma, G.N. Mehta, Bull. Electrochem. 15 (1999) 67. [ Links ]
15. M. Kliskic, J. Radosevic, S. Gudic, V. Katalinic, J. Appl. Electrochem. 30 (2000) 823. [ Links ]
16. A.Y.E. Etre, M. Abdullah, Z.E.E. Tantawy, Corros. Sci. 47 (2005) 385. [ Links ]
17. A. Chetouani, B. Hammouti, Bull. Electrochem. 19 (2003) 23. [ Links ]
18. S. Rajendran, S. Vaibhavi, N. Anthony, D.C. Trivedi, Corrosion 59 (2003) 529. [ Links ]
19. A. Singh, V.K. Singh and M.A. Quraishi, Int. J. Corros. (2010) 10.
20. P.C. Okafor, V.I. Osabar, E.E. Ebenso, Pigment Resin Tech. 36 (2007) 299. [ Links ]
21. S.K. Sharma, A. Mudhoo, G. Jain, J. Sharma, Rasayan J. Chem. 2 (2009) 332. [ Links ]
22. S.A. Umoren, E.E. Ebenso, Pigment Resin Tech. 37 (2008) 173. [ Links ]
23. N.O. Eddy, Port. Electrochim. Acta 27 (2009) 579. [ Links ]
24. N.O. Eddy, A. Patricia, Ekwelmemgbo, P.A.P.Mamza, Green Chem. Lett. Rev. 2 (2009) 223.
25. M. Sangeetha, S. Rajendran, T.S. Muthumegala, A. Krishnaveni, Zastita Materijala 52 (2011) 3. [ Links ]
26. P.R. Vijayalakshmi, R. Rajalakshmi, S. Subhashini, Port. Electrochim. Acta 29 (2011) 9. [ Links ]
27. N. Lahhit, A. Bouyanzer, J.-M. Desjobert et all, Port. Electrochim. Acta 29 (2011) 127. [ Links ]
28. K.P.V. Kumar, M.S.N. Pillai, G.R. Thusnavis, Port. Electrochim. Acta, 28 (2010) 373. [ Links ]
29. A. Bouyanzer, B. Hammouti, L. Majidi, B. Haloui, Port. Electrochim. Acta 28 (2010) 165. [ Links ]
30. A.O. Odiongenyi, S.A. Odoemelam, N.O. Eddy, Port. Electrochim. Acta 27 (2009) 33. [ Links ]
31. I.B. Obot, N.O. Obi-Egbedi, Port. Electrochim. Acta 27 (2009) 517. [ Links ]
32. Indian Medicinal Plants, A Compendium of 500 Species, Orient Longman Ltd., Madras, 4 (1995) 252. [ Links ]
33. L.Y. Foo, Phytochemistry 33 (1993) 487. [ Links ]
34. S. Rajendran, M. Agasta, R. Bama Devi et all, Zastita Materijala 50 (2009). [ Links ]
35. C. Mary Anbarasi, Susai Rajendran, J. Electrochem. Soc. India 60 (2011) 115.
36. N. Antony, H.B. Sherine, S. Rajendran, Port. Electrochim. Acta 28. (2010) 1. [ Links ]
37. P. Shanthy, P. Rengan, A.T. Chelvan, K. Rathika, S. Rajendran, Indian J. Chem. Tech. 16 (2009) 328. [ Links ]
38. S. Rajendran, B.V. Apparao, N. Palanisamy, Bull. Electrochem. 13 (1997) 441. [ Links ]
39. H.B. Sherine, A.J.A. Nasser, S.Rajendran, J. Electrochem. Soc. India 58 (2009) 30. [ Links ]
40. S.A. Kanimozhi, S. Rajendran, Arabian J. Sci. Eng. 34 (2009) 37. [ Links ]
41. B. Sherine, A.J.A. Nasser, S. Rajendran, Int. J. Eng. Sci. Tech. 2 (2010) 341. [ Links ]
42. Y. Yesu Thangam, M. Kalanith, C. Mary Anbarasi, S. Rajendran, The Arabian J. Sci. Eng. 34 (2009) 49.
43. J. Sathiyabama, S. Rajendran, J.J. Sundari, B. Shyamaladevi, J. Eng. Sci. Tech. Rev. 3 (2010) 27. [ Links ]
44. B. Narayanasamy, S. Rajendran, Prog. Org. Coat. 67 (2010) 246. [ Links ]
45. S. Rajendran, P. Sumithra, B.S. Devi, J. Jeyasundari, Zastita Materijala 50 (2009) 223. [ Links ]
46. H. Benita Sherine, R.K Mani, S. Rajendran, J. Electrochem. Soc. India 57 (2008) 67.
47. S. Rajendran, V. Uma, A. Krishnaveni et all, Arabian J. Sci. Eng. 34 (2010) 147. [ Links ]
48. S.A. Kanimozhi, S. Rajendran, Arabian J. Sci. Eng. 35 (2009) 41. [ Links ]
49. R.J. Rathish, S. Rajendran, J.L. Christy et all, Open Corros. J. 3 (2010) 38. [ Links ]
50. S. Rajendran, B.V. Apparao, N. Palanisswamy, Electrochim. Acta 44 (1998) 513. [ Links ]
51. S. Rajendran, B.V. Apparao, N. Palaniswamy, Anti-corrosion Methods 45 (1998) 338. [ Links ]
52. R.M.S. Francis, G.C. Bassler, T.C. Morrill, Spectrometric identification for organic compounds (New York , N.Y: John Wiley & Sons, 1986. [ Links ]
53. I.Sekine, Y.Hirakawa, Corrosion 42 (1986) 272. [ Links ]
* Corresponding author. E-mail address: sangeethamanirevathi@gmail.com
Received 10 May 2011; accepted 10 December 2011














