Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Portuguese Journal of Nephrology & Hypertension
Print version ISSN 0872-0169
Port J Nephrol Hypert vol.32 no.2 Lisboa June 2018
ORIGINAL ARTICLE
Infectious risk of elderly patients on peritoneal dialysis: Experience of a portuguese center
Carolina L Belino1, Patrícia S Barreto1, Maria C Santos1, Ana M Gomes1, Isabel Fonseca2‑4, João C Fernandes1
1 Department of Nephrology, Centro Hospitalar de Vila Nova de Gaia e Espinho, Vila Nova de Gaia, Portugal
2 Department of Nephrology, Hospital de Santo António, Centro Hospitalar do Porto – CHP, Porto, Portugal
3 Unit for Multidisciplinary Research in Biomedicine, Instituto de Ciências Biomédicas Abel Salazar (CBAS), Universidade do Porto, Porto, Portugal
4 ISPUP‑EPI Unit, Universidade do Porto, Porto, Portugal
ABSTRACT
Background: The goal of this study was to compare the risk of peritoneal dialysis‑related infections in younger and older patients and to identify risk factors for infection in elderly patients.
Methods: We performed a longitudinal retrospective study on a population of Portuguese peritoneal dialysis patients treated at the same center between January 2005 and December 2015. Clinical and laboratory data were collected from medical reports. Two groups of patients were compared: non elderly (< 65 years) and elderly (≥ 65 years).
Results: Among 100 patients, there were 73 non elderly (median age 52 years) and 27 elderly (median age 74 years). Elderly patients were not associated with higher PD‑infection rates or with less time to PD‑related infections. Cerebrovascular disease was the only significant adverse predictor of peritonitis in elderly (crude HR 3.8; 95% CI 1.10 to 13.34; P = 0.035) and those with higher levels of serum albumin were less likely to developperitonitis (crude HR 0.47 per each g/dl of increase; 95% CI 0.24 to 0.89; P = 0.023).
Conclusions: In our study, elderly patients did not present a greater risk for peritonitis or catheter‑related infections.
Key‑words: older patients, peritoneal dialysis, infection risk
INTRODUCTION
The incidence and prevalence of older patients (≥65 years) with end stage kidney disease (ESKD) is increasing worldwide as life expectancy is growing1‑8.
Data from the Australia and New Zealand Dialysis and Transplant (ANZDATA) registry revealed an increase of elderly with ESKD by 8% per year between 2004 and 2008 and patients over 60 years were more than 50% of ESRD patients2. Similar results were obtained from United States and Canada3‑5, Europe6 and China7.
The major advantages of PD in this age group are preservation of residual renal function and hemodynamic stability, avoidance of problems related with vascular access, postdialysis recovery time and falls8‑19.
However, infectious risk is a major concern and remains an obstacle to PD prescription in elderly20.
The goal of this study was to compare the risk of peritoneal dialysis‑related infections between younger and older patients and to identify risk factors for infection in elderly patients.
METHODS
Data from incident PD patients from a Portuguese center between January 2005 and December 2015 were collected from medical reports. The number of patients on assisted PD was residual so these patients were excluded. Patients were stratified into two groups according to their ages at admission on PD program: non‑elderly (< 65 years) and elderly (≥65 years) patients. Demographic and clinical data were recorded at entrance in PD program. Laboratory data were consequently recorded from routine analysis, considering all time on PD for each patient till the end of follow‑up period, which was from January 1, 2005 to December 31, 2015. Mean values of these laboratory data (for all the follow‑up period) were calculated for each patient and included posteriorly in the analysis. The retrieved data are expressed in table 1.
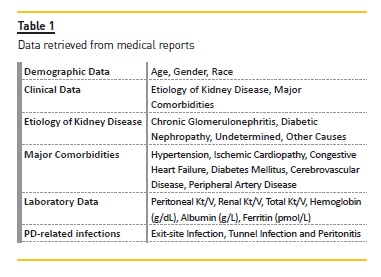
PD‑related infections that occurred during follow‑up period were categorized as exit‑site infection, tunnel infection and peritonitis. All patients were treated according to the same protocol.
Statistical Analysis
Patient characteristics were summarized using descriptive statistics. Continuous variables were expressed as the mean (standard deviation – SD) or median [interquartile range – IQR] and categorical variables were expressed as percentage. The Kolmogorov‑Smirnov test was used to determine the normality of the distribution of parameters. Patient characteristics were compared for elderly and non elderly patients using chi‑squared or Mann‑Whitney tests as appropriate.
Infectious rates were calculated by dividing the total number of PD related infections (peritonitis, exit‑site and tunnel infections) by total time at risk (calculated as number of days from dialysis initiation to the end of follow‑up for all patients divided by 365.25 days) and expressed as number of infection episodes per dialysis year. Poisson regression analysis was used to assess the impact of various factors on outcome of interest (peritonitis, exit‑site and tunnel infections), including demographics (age categorized into <65 yr and ≥ 65 yr), comorbidities (diabetes mellitus, hypertension, cerebrovascular disease, and peripheral artery disease) and albumin level from routine analysis.
Cox proportional hazard models were used to calculate crude hazard ratios (HRs) and 95% confidence intervals for the association between several demographic and clinical variables and PD‑related infections (peritonitis, exit‑site and tunnel infections). The following variables, obtained at the start of dialysis, were included in the analysis: age (categorized into <65 yr and ≥ 65 yr, PD modality, etiology of kidney disease, comorbidities and serum albumin level. All analyses were conducted using SPSS® software (version24.0).
RESULTS
Characteristics of patients
A total of 100 caucasian patients were included, 73 non elderly and 27 elderly. In non elderly group, median age was 52 [44‑58] years. In elderly group, median age was 73 [68‑77] years. The demographic and clinical data are described in Table 2.
PD‑related infections
Patients were followed for a median time of 2.6 [1.4 – 4.0] years. During the study period, we identified 294 PD‑related infections. There were 218 infections in non elderly group and 76 infections in elderly group. The number of patients who never had a PD‑related infection was 20 (27.4%) in non elderly group and 8 (29.6%) in elderly group.
Exit‑site infections were the most frequent event (0.48 per patient‑year), followed by peritonitis (0.41 per patient‑year). Rates of PD‑related infections are shown in Table 3. Table 4 expresses the patient status according to history of a PD‑related infection.
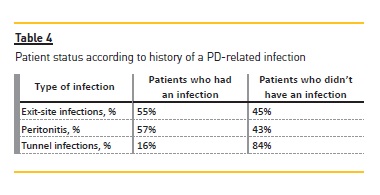
No significant rate differences were found between elderly and non elderly patients for exit‑site infection and peritonitis. No tunnel infections were identified in elderly patients; thus an infection episode was added in order to allow to study the effect of age on tunnel infection rate. In this context, the age under 65 years was marginally predictive of peritonitis (P = 0.063).
None of the studied comorbidities were significant predictors of any of PD‑related infections. The only significant univariate predictor of exit‑site infection was albumin level (rate ratio [RR], 0.58 per 1‑g/ dL increase; 95% CI, 0.39 to 0.85; P = 0.005).
Risk of PD‑related infectious complications
Exit‑site Infections
Considering the entire sample, the effect of age as a predictor of exit‑site infection was not statistically significant (HR 0.32; 95% CI 0.37 to 1.38; P = 0.72).
Factors associated with shorter time to exit‑site infection were cerebrovascular disease (crude HR 4.86; 95% CI 1.80 to 13.11; P = 0.002) and hypertension (crude HR 2.29; 95% CI 0.17 to 4.51; P = p=0.016). Patients with higher levels of serum albumin were less likely to develop exit‑site infection (crude HR 0.41 per each g/dl of increase; 95% CI 0.21 to 0.81; P = 0.01). Parameters of dialysis adequacy and other laboratory parameters did not have a significant effect.
In non elderly group, cerebrovascular disease (crude HR 5.29; 95% CI 1.76 to 15.89; P = 0.003), diabetes mellitus (crude HR 3.37; 95% CI 1.39 to 8.19; P = 0.007), hypertension (HR 3.11; 95% CI 1.39 to 6.99; P = 0.006) and peripheral artery disease (crude HR 3.31; 95% CI 1.39 to 7.88; P = 0.007) were associated with greater risk of infection. Non elderly patients with higher albumin levels were less likely to develop exit‑site infections (crude HR 0.31 per each g/dl of increase; 95% CI 0.13 to 0.73; P = 0.008).
In elderly group, none of the evaluated factors was significantly associated with shorter time to exit‑site infection.
Peritonitis
Older age (≥ 65 yr) was not associated with a shorter time to peritonitis (HR 0.43; 95% CI 0.42 to 1.44; P = 0.78).
Cerebrovascular disease was the only significant adverse predictor of peritonitis (crude HR 3.8; 95% CI 1.10 to 13.34; P = 0.035), and patients with higher levels of serum albumin were less likely to develop peritonitis (crude HR 0.47 per each g/dl of increase; 95% CI 0.24 to 0.89; P = 0.023).
In the non elderly group, the predictors of peritonitis included diabetes mellitus (crude HR 2.72; 95% CI 0.99 to 7.46; P = 0.05), hypertension (crude HR 2.25; 95% CI 1.07 to 4.72; P = 0.03) and peripheral artery disease (crude HR 3.9; 95% CI 1.65 to 9.42; P = 0.002). Patients with higher levels of serum albumin were less likely to develop peritonitis (crude HR 0.40 per each g/dl of increase; 95% CI 0.17 to 0.98; P = 0.045). Parameters of dialysis adequacy and other laboratory parameters did not have a significant impact.
Considering only patients with 65 years old or higher, no significant predictors of peritonitis infection were found.
Tunnel Infections
All the cases of tunnel infections occurred in non elderly group. Better dialysis adequacy, as measured as total Kt/V (HR 0.15 per each unit of increase; 95% CI 0.03 to 0.88; P = 0.036) and preserved residual renal function, represented by renal Kt/V (HR 0.11 per each unit of increase; 95% CI 0.02 to 0.59; P = 0.01) were associated with lower probability of infection occurrence.
DISCUSSION
In this study, patient characteristics are comparable to other European cohorts1,21. Higher prevalence of cardiovascular disease and diabetes mellitus stand out in both groups. Older age seems not to be associated with greater risk of peritonitis or catheter‑related infections, despite the higher comorbidity burden.
In the ANZDATA registry, between 1991 and 2007, elderly had similar peritonitis‑free survival rate, despite higher peritonitis‑related and all‑cause mortality, probably related with severity of episodes in a more fragile patient2. Other international studies from this era showed similar results, as reported in France21, Canada22 and Brazil23. A more recent study from China, which analyzed peritonitis rates in 1252 incident patients between 1998 and 2012, also confirmed that age was not a risk predictor for peritonitis24.
Better results were achieved with introduction of training programs and education, technical advances (new machinery, use of the flush‑before‑fill technique and novel types of catheter), newer PD solutions and decontamination protocols with topical antibiotics20,25‑27.
Practical advice on managing older patients on PD has recently been published and showed that, with appropriate training and adequate support, it ́s possible to keep a respectable peritonitis rate in those patients30.
Impact of comorbidities and primary kidney disease in peritonitis risk has been addressed in some studies.
Diabetes mellitus is related with a higher severity of infection and is not yet clear whether it is a risk factor for peritonitis or not22,26. It has been suggested that chronic lung disease, congestive heart failure, ischemic cardiopathy and hypertension also increase the risk26,30,31. In this study, cerebrovascular disease was the only significant adverse predictor of peritonitis found in elderly, probably because of the small sample of this age group.
Glomerulonephritis as primary kidney disease was marginally associated with a decreased risk for peritonitis in a large Canada study22 but Huang et al.32 found that lupus nephritis had significant increased risk, although was not clear if was secondary to steroid use or by nephritis itself. However, a recent analysis from the BRAZPD II cohort did not confirm this association33. Chronic glomerulonephritis was not the major cause of ESRD in our patient groups, which, allied with the small samples, may have prevented the identification of a significant impact.
Hypoalbuminemia is also a well‑known risk factor for peritonitis, so it is expected that higher albumin levels to be associated with a reduced infectious risk, as showed in our results26. The relation between infection and Kt/V are not totally clarified35. A recent 10‑year single‑center analysis from Italy compared two groups of PD patients: a group who started incremental PD with 1 to 2 dwells per day versus a standard group (> 3 dwells per day). Despite lower total Kt/V values in the incremental group, a trend towards lower peritonitis incidence was observed, with similar survival rates and less hospitalization. A good PD program with professionals with expertise, the reduced number of connections and the dry period are some reasons appointed by the authors to explain these results35.
With respect to tunnel infections, none occurred in elderly group. Some authors agree that catheter‑related infections are less frequent in elderly. Aging is associated with reduced response of sweat glands to thermal alterations, with less local humidity. Lower functional status in older age may reduce mechanical stress on catheter and there for contribute to maintenance of skin and tunnel integrity, reducing bacteria colonization and catheter biofilm creation26,27.
This study has several limitations. This is an observational and retrospective study, so it cannot eliminate the presence of additional variables that might confound results. The small sample mad had prevented the identification of other factors with significant impact in outcomes. Missing data, particularly in older clinical processes, limited the retrieval of additional information (for example. the number of patients who started incremental PD). Additional investigations are necessary to better analyze and validate these results.
CONCLUSIONS
Peritoneal dialysis is underused in the elderly population.
However, multiple studies have proved that this is a suitable option for this age group, with respectable results and many advantages over hemodialysis. There is no solid evidence of more PD‑related complications, especially infection. With adequate planning, education and support, many of the barriers for PD prescription in older patients can be successfully overcome.
Contributions: All authors intervene in the drafting of the manuscript, search strategy, data extraction and result interpretation and discussion. All except IF contributed with medical expertise on Peritoneal Dialysis. IF performed the statistical analysis.
Support:No financial or non‑financial support of any kind was provided.
References
1. Castrale C, Evans D, Verger C, Fabre E, Aguilera D, Ryckelynck J et al. Peritoneal dialysis in elderly patients: report from the French Peritoneal Dialysis Registry (RDPLF). Nephrol Dial Transplant 2009; 25(1):255‑62. [ Links ]
2. McDonald S, Excell L, Livingston B. The thirty‑second report: Australia and New Zeland Dialysis and Transplant Registry, 2009. ANZDATA Registry; 2009:1‑12. [ Links ]
3. United States National Kidney Foundation. United States Renal Data System 2012 Annual Data Report: atlas of chronic kidney disease and end stage renal disease in the United States. Am J Kidney Dis 2013; 61(1):e1‑e480. [ Links ]
4. Fenron SS, Desmeules M, Jeffery JR, Corman JL. Dialysis therapy among elderly patients; data form the Canadian Organ Replacement Register, 1981‑1991. Adv Perit Dial 1993; 9:124‑9. [ Links ]
5. Kurella M, Covinsky KE, Collins AJ, Chertow GM. Octagenarians and nonagenarians starting dialysis in the United States. Ann Intern Med 2007; 146:177‑83. [ Links ]
6. Rapport annuel 2013 du Réseau Epidémiologie at information en Néphrologie (REIN). Available at https://www.agence biomedecine.fr/IMG/pdf/rapport_rein2013.pdf. Accessed November 2, 2016 [ Links ]
7. Ho YW, Chau KF, Choy BY, Fung KS, Cheng YL, Kwan TH, et al. Hong Kong renal registry report 2012. Hong Kong J Nephrol 2013; 15:28‑43. [ Links ]
8. Campos A, Malheiro J, Teixeira L, Santos S, Carvalho MJ, Cabrita A, et al. Peritoneal dialysis dropouts in different age and era cohorts: focus on the elderly. Port J Nephrol Hypert 2015; 29(4):41‑8. [ Links ]
9. Brown E, Finkelstein FO, Iyasere O, Kliger AS. Peritoneal or hemodialysis for the frail elderly patient, the choice of 2 evils? Kidney Int. 2016 Oct 20. pii: S0085‑2538(16)30474‑4. [ Links ]
10. Shea YF, Lam MF, Lee MS, Mok MY, Lui SL, Yip TP, et al. Prevalence of cognitive impairment among peritoneal dialysis patients, impact on peritonitis and role of assisted dialysis. Perit Dial Int 2016;36(3):284‑90. [ Links ]
11. Brown EA, Johansson L, Farrington K, Gallagher H, Sensky T, Gordon F, et al. Broadening Options for Long‑term Dialysis in the Elderly (BOLDE): differences in quality of life on peritoneal dialysis compared to haemodialysis for older patients. Nephrol Dial Transplant 2010; 25:3755‑63. [ Links ]
12. Van de Luijtgaarden MW, Noordzij M, Stel VS, Ravani P, Jarraya F, Collart F, et al. Effects of comorbid and demographic factors on dialysis modality choice and related patient survival in Europe. Nephrol Dial Transplant 2011;26(9):2940‑7. [ Links ]
13. Lim WH, Dogra GK, McDonald S et al. Compared with younger peritoneal dialysis patients, elderly patients have similar peritonitis‑free survival and lower risk of technique failure, but higher risk of peritonitis‑related mortality. Perit Dial Int 2011; 31:663‑71. [ Links ]
14. Baek MY, Kwon TH, Kim YL, Cho DK. CAPD, an acceptable form of therapy in elderly ESRD patients: a comparative study. Adv Perit Dial 1997; 13:158‑61. [ Links ]
15. Okayama M, Inoue T, Nodaira Y, Kimura Y, Nobe K, Sueyoshi K, et al. Aging is an important risk factor for peritoneal dialysis‑associated peritonitis. Adv Perit Dial 2012; 28:50‑4. [ Links ]
16. Fernandez MA, Ortiz AM, Valenzuela M, Morales RA. Peritoneal dialysis in chronic renal failure patients over 65 years of age. Adv Perit Dial 2004; 20:129‑31. [ Links ]
17. Dimkovic N, Oreopoulos DG. Chronic peritoneal dialysis in the elderly. Semin Dial 2002; 15:94‑7 [ Links ]
18. Dimkovic NB, Prakash S, Roscoe J, Brissenden J, Tam P, Bargman J, et al. Chronic peritoneal dialysis in octagenerians. Nephrol Dial Transplant 2011; 16:2034‑40. [ Links ]
19. Kumar VA, Sidell MA, Jones JP, Vonesh EF. Survival of propensity matched incident peritoneal and hemodialysis patients in a United States health care system. Kidney Int 2014; 86:1016‑22. [ Links ]
20. Cheuk‑Chun Szeto. Peritoneal dialysis‑related infection in the older population. Perit Dial Int 2011; 35:659‑62. [ Links ]
21. Duqennoy S, Béchade C, Verger C, Ficheux M, Ryckelynck JP, Lobbedez T. Is peritonitis risk increased in elderly patients on peritoneal dialysis? Report from the French Language Peritoneal Dialysis Registry (RDPLF). Perit Dial Int 2015; 154:1‑6. [ Links ]
22. Nessim S, Bargman J, Austin P, Nisenbaum R, Jassal SV. Predictors of peritonitis in patients on peritoneal dialysis: results of a large, prospective Canadian database. Clin J Am Soc 2009; 7:1195‑200. [ Links ]
23. De Moraes TP, Olandoski M, Caramori JC, Martin LC, Fernandes N, Divino‑ Filho JC, et al. Novel predictors of peritonitis‑related outcomes in the BRAZPD cohort. Perit Dial Int 2014; 34:179‑87. [ Links ]
24. Ma TK, Chow KM, Kwan BC, Pang WK, Leung CB, Li PK, et al. Peritonitis before peritoneal dialysis training: analysis of causative organisms, clinical outcomes, risk factors and long‑term consequences. Clin J Am Soc Nephrol 2016; 11(7):1219‑26. [ Links ]
25. United States, Department of Health and Human Services, Public Health Service, National Institute of Diabetes and Digestive and Kidney Diseases, U.S. Renal Data System (USRDS). USRDS 2011 annual data report: atlas of chronic kidney disease in the United States. Bethesda, MD: USRDS; 2011. [ Links ]
26. Kerschbaum J, König P, Rudnicki M. Risk factors associated with peritoneal‑ dialysis‑related peritonitis. Int J Nephrol 2012; 2012:483250. [ Links ]
27. De vecchi AF, Maccario M, Braga M, Scalamogna A, Castelnovo C, Ponticelli C. Peritoneal dialysis in nondiabetic patients older than 70 years: comparison with patients aged 40 to 60 years. Am J Kidney Dis 1998; 31:479‑90. [ Links ]
28. SU YJ, Lee CT, Chuang FR, Lee CH, Wang YC, Cheng BC, et al. Comparison of clinical characteristics between automated peritoneal dialysis and continuous ambulatory peritoneal dialysis: a 2‑year single‑center observational study. Ren Fail 2010; 32(7):817‑24. [ Links ]
29. Beduschi Gde C, Figueiredo AE, Olandoski M, Pecoits‑Filho R, Barreti P, Moraes TP. Automated peritoneal dialysis is associated with better survial rates compared to continuous ambulatory peritoneal dialysis: a propensity score matching analysis. PLoS One 2015; 10(7):e0134047. [ Links ]
30. Cho Y, Johnson DW. Peritoneal dialysis‑related peritonitis: towards improving evidence practices, and outcomes. Am J Kidney Dis 2014; 64(2):278‑89. [ Links ]
31. Oo TN, Roberts TL, Collins AJ. A comparison of peritonitis rates from the United States Renal Data System database: CAPD versus continuous cycling peritoneal dialysis patients. Am J Kidney Dis 2005; 45(2):372‑80. [ Links ]
32. Huang JW, Hung KY, Yen CJ, Wu KD, Tsai TJ. Systemic lupus erythematosus and peritoneal dialysis: outcomes and infectious complications. Perit Dial Int 2001; 21(2):143‑7. [ Links ]
33. Proença de Moraes T, Massignan B, Figueiredo AE, Barretti P, Olandoski M, Kirk A, et al. Systemic lupus erythematous and clinical outcomes in peritoneal dialysis. Lupus 2015; 24(3):290‑8. [ Links ] [ Links ] [ Links ]
Carolina Lã Belino, MD
Department of Nephrology, CHVNG/E, Conceição Fernandes Street
4434‑502, Vila Nova de Gaia, Portugal.
E‑mail: carolinabelino@hotmail.com
Disclosure of potential conflicts of interest: The authors disclose no conflicts of interest
Received for publication: Nov 11, 2017
Accepted in revised form: Jun 2, 2018














