Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Portuguese Journal of Nephrology & Hypertension
Print version ISSN 0872-0169
Port J Nephrol Hypert vol.30 no.4 Lisboa Dec. 2016
ORIGINAL ARTICLE
Exit-site fungal infections – experience of a Peritoneal Dialysis Unit
Sofia Coelho1, Ana Beco2, Ana Oliveira2, Carla Santos2, Manuel Pestana2
1 Centro Hospitalar de Setúbal
2 Centro Hospitalar de São João
ABSTRACT
Introduction: Exit-site infections are an important complication of peritoneal dialysis; however, very little is known about fungi-related exit-site infections. The literature is very sparse and there are virtually no studies that report it. Objective: To evaluate in retrospect the risk factors associated with exit-site fungal infection and its development. Methods: The study included all diagnosed episodes of exit- site fungal infections in patients undergoing peritoneal dialysis in a hospital unit between 2011 and 2014, analyzed in relation to demographic, clinical and analytical variables. Results: The studied group included 26 patients, 70% of which were female, with a median age of 54 years old; average length in PD treatment three years. The majority of patients (70%) were undergoing manual peritoneal dialysis. About 30 episodes of exit-site fungal infections were diagnosed during the follow-up period, which corresponded to 5.6% of total exit-site infections diagnosed. About 23% of patients suffered from diabetes mellitus and 13% had undergone immunosuppressive therapy in the six months previous to the episode. Two thirds of patients (n=20) had undergone antibiotherapy in the previous three months, the majority (65%) for the treatment of peritoneal dialysis-related infection and 46% of these patients underwent concomitant antifungal prophylaxis with fluconazole. The exit-site fungal infections were predominantly caused by Candida parapsilosis (67%). Eighty per cent of patients (n=24) improved with antifungal therapy (fluconazole or itraconazole), and the remaining patients underwent peritoneal catheter replacement (n=3) or removal (n=3) due to persistent infection. Exit-site fungal infections were the cause of technique failure in 2 of these patients. Conclusion: Exit-site fungal infections are an unusual but important complication of peritoneal dialysis. In this study, the most relevant risk factor associated with these infections was previous antibiotherapy and the majority of patients evolving favourably under medical treatment.
Key Words: Antibiotherapy; exit-site infections; fungal infection; peritoneal dialysis.
BACKGROUND
The success of the peritoneal dialysis (PD) technique rests on the permanent and safe access to the peritoneal cavity. Despite the recent evolution, mainly concerning exit-site care, the establishing of infection prophylaxis protocols and the development of PD technique, infection is still an important cause of morbidity and PD failure1,2. Infections directly related to technique are, for the most part, caused by bacterial agents; infections caused by fungal agents are considerably less common2.
Although fungal peritonitis is well documented in the literature3-11, and there are international recommendations in place for its diagnosis and treatment1, this is not the case for exit-site fungal infections (ESFI). The literature is very sparse and there are virtually no studies that report its incidence, its impact on morbidity and death rate and the appropriate therapeutic regimen. On the whole, the agent responsible for the majority of fungal infections in humans is Candida spp12.
There are over 350 different species that colonize the skin, oropharynx, gastrointestinal tract and vagina. They may cause opportunist infection in the presence of normal flora disruption, in a breach of the skin and mucous membranes or in a defect of cellular immunity within the host13-15. At least 13 species of Candida are known to cause infection, the most common being albicans, glabrata, krusei, parapsilosis and tropicalis13,14. The more benign forms of infection are mucocutaneous infections14 (where exit-site infections (ESI) can be placed) and these occasionally recur, without the identification of an obvious predisposition element12. The main risk factors for fungal infection are widely known: immunosuppression; previous antibiotherapy, especially of long duration; malnutrition and the presence of other outbreaks of fungal infection, amongst others14,15.
It is known that in patients undergoing PD, especially in relation to fungal peritonitis, an ascertained risk factor is the occurrence of previous episodes of bacterial peritonitis, in which there is also overgrowth of the fungal agent in the gastrointestinal tract (due to a flora imbalance subsequent to the use of antibiotics) and a decline in the activity of the local immune system caused by peritonitis3. Although not proven, Gram negative infections seem to carry a greater risk3. It remains to be seen if the same mechanisms related to ESFI apply, in what way they should be considered and if they carry a greater risk of technique failure that imply the immediate application of early antifungal therapy or even peritoneal catheter removal.
METHODS AND STUDIED POPULATION
A retrospective and observational study was undertaken to analyze all ESFI episodes occurring in a population of prevalent peritoneal dialysis patients at the Peritoneal Dialysis Unit of the São João Hospital (Hospital de São João – Porto, Portugal) between January 2011 and December 2014. All patients had a swan neck, double-cuffed peritoneal catheter with a curved edge implanted via laparoscopy, minilaparotomy or Y-TEC® system in the paraumbilical region. All patients were submitted to antibiotic prophylaxis with 1 gram of intravenous cephazolin. On average, patients initiated treatment at least two weeks after catheter insertion. The training included exit-site care, which was based on the use of a povidone iodine solution in the initial healing phase and, afterwards, on daily cleaning with 0.9% sodium chloride and the prophylactic topical application of mupirocin. The nasal search for Staphylococcus aureus colonization was undertaken in all patients, and those with a positive result were treated with nasal mupirocin, according to Unit protocol. All ESI episodes were obtained through a clinical archive where they are systematically registered. The data relative to ESFI were obtained from medical records for the following variables: demographic characteristics; comorbidities; aetiology of chronic kidney disease; previous episodes of ESI or peritonitis (in the preceding three months); isolated agents and whether or not prophylaxis of fungal peritonitis with fluconazole was undertaken; antibiotherapy in the preceding three months or immunosuppressive therapy in the preceding six months; characterization of PD technique (time, modality and adequacy parameters); analytical profile; characterization of ESFI episodes, mainly an isolated agent, the isolating of bacterial agents taking place within the episode, type and duration of medical treatment and clinical progression. The instances of ESFI where there was a previous ESI episode with a bacterial agent, in which the infection was resolved through the use of antibiotherapy, were considered as previous ESI; in the instances where the bacterial infection was not resolved, the ESFI was considered subsequent. ESI was diagnosed according to the recommendations of the International Society of Peritoneal Dialysis (ISPD)1.
In the cases of a clinically questionable orifice (in the absence of purulent exudates) observation was maintained and local treatment with topical antibiotic and/or silver nitrate was administered, and systemic antibiotherapy was initiated if there was no clinical improvement at the end of one week. In most cases, this was undertaken according to Unit protocol, with sulfamethoxazole-trimethoprim or ciprofloxacin. As soon as the microbial examination result was available, drug therapy was adjusted according to fungal susceptibility to antibiotics/antifungal agents. In the ESI episodes with no resolution under appropriate and prolonged medical treatment, the patient was submitted to a shaving of the external cuff (if tunnel infection was present), peritoneal catheter substitution under antifungal coverage and/or antibiotic in the cases of controlled infection, or peritoneal catheter removal in the cases of unsuccessfully controlled infection.
RESULTS
During the four years, 30 ESFI episodes were diagnosed occurring in 26 patients. In the same time period, there was a total register of 534 ESI episodes, resulting in a proportion of ESFI corresponding to 5.6% of the total ESI. Twelve percent (3 patients) of patients had more than one ESFI episode.
C haracterization of the population studied (tables I and II)
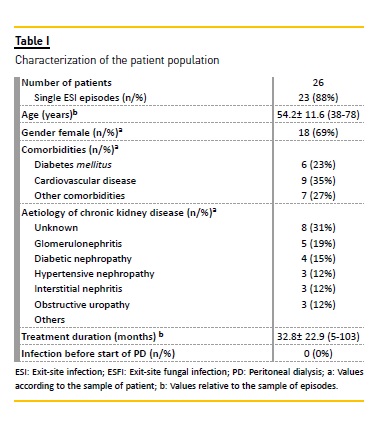
The median age at time of diagnosis was 54.2±11.6 years of age and 69% of the population was female.
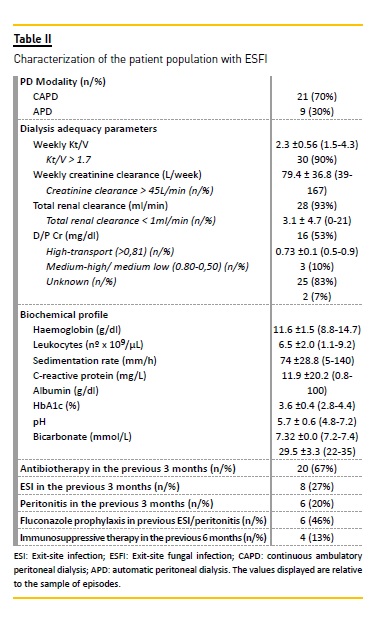
About 23% had diabetes mellitus. The average span of time undergoing PD was 32.8±22.9 months, with 80% of patients receiving treatment for over a year and 33% for over five years. No episode occurred before the start of PD; in other words, during the healing or training period. Of the studied population, 70% was undergoing continuous ambulatory peritoneal dialysis (CAPD) and 30% received automatic peritoneal dialysis (APD), where 90% showed a total Kt/V over 1.7, as recommended by international guidelines29. In regards to the analytical profile, the average albumin was of 3.6g/dl (lower than 3.5g/dl in 70% of cases) and the average HbA1c was 5.7% (6.5% in patients with known cases of diabetes mellitus). The remaining ascertained parameters are presented in table 2. Two thirds of the patients (n=20) had undergone antibiotherapy in the previous three months; of these, over half (n=14) owing to PDrelated infection: eight cases of ESI (all Gram positive) and six cases of peritonitis (four Gram negative). Of this patient group, 46% had undergone concomitant fungal peritonitis prophylaxis with fluconazole. There was no recent history of fungal infection in any of the cases (<3 months). Two patients corresponding to four ESFI episodes) had undergone immunosuppressive therapy in the six months prior to the ESFI episode. Of the nine cases not associated with any of the aforementioned conditions, diabetes mellitus was identified among six other risk factors of fungal infection.
C haracterization and evolution of ESFI episodes (tables III and IV)
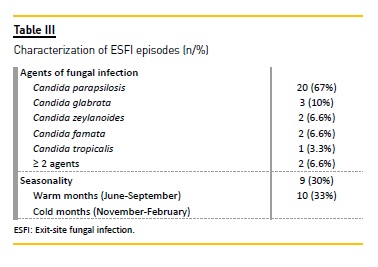
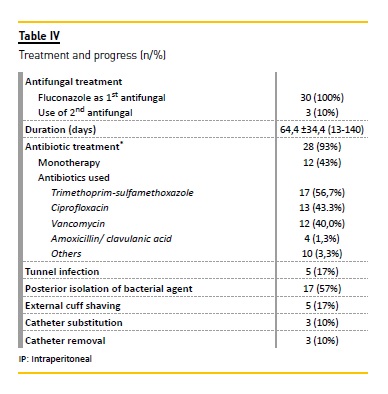
All ESFI episodes were caused by Candida spp. The most frequently isolated species was Candida parapsilosis (67%), followed by Candida glabrata (10%), Candida famata (7%) and Candida zeylanoides (7%). In two cases there was concomitant isolation of two fungal agents (Candida parapsilosis/guilliermondii and Candida parapsilosis/albicans). In 23% of the cases the isolation of the fungal agent was concomitant (n=2) or posterior (n=5) to the isolation of bacterial agents, more commonly Staphylococcus epidermidis (44%), and an average of 47 days after bacterial isolation in these. No difference was ascertained in ESFI incidence according to season. Concomitant tunnel infection was observed in 17% of the episodes. All cases were initially treated with fluconazole (a dosage of 200mg/day), and in 10% of cases, due to poor clinical response, a second antifungal was associated (itraconazole). The average length of treatment was 64.4 (±33.9) days. In 93% (n=28) of episodes there was concomitant use of antibiotics, overall applied empirically at the moment of diagnosis. Eighty per cent of cases(n=24) progressed favourably with medical treatment.
In the cases with evidence of tunnel infection and with inferior clinical response after prolonged therapy, a shaving of the external cuff was performed in 17% (n=5), in an attempt to control the infection, succeeding in only two cases. In total, 20% (n=6) did not respond to appropriate and prolonged antifungal therapy, and were submitted to peritoneal catheter removal (n=3) or substitution (n=3), resolving the infectious process. There was one case in which two consecutive episodes of peritonitis showed the same agent (Candida glabrata), and the second episode occurred 52 days after the termination of antifungal therapy and resulted in peritoneal catheter removal. ESFI were the cause of technique failure in 23 of 6.7% patients that required the removal of the peritoneal catheter.
DISCUSSION
ESFI are an uncommon complication of PD. Studies that discuss it are sparse and even international guidelines from the ISPD1 say very little beyond suggesting treatment with fluconazole. In our experience, ESFI represent a small but not insignificant percentage of total ESI. Luzar et al16 report a ESFI incidence between 1 and 3% in patients undergoing CAPD, which is considerably lower than that observed in our population. Another recent study17 with the aim of identifying, in a low occurrence context, the targets in ESI and tunnel prevention, reports an even lower incidence of ESFI (0.9%). ESFI seem to mostly share the general characteristics of fungal infections, mainly the association to variable degrees of immunosuppression and prior prolonged antibiotherapy.
In this study, the most important risk factor in ESFI seems to have been previous antibiotherapy. In regards to the prophylactic use of antifungals, some studies5-7,18 have shown a reduced occurrence of fungal peritonitis with the implementation of this therapy during prolonged treatment of bacterial peritonitis. However, we ascertained that of the ESFI patients with previous PD-related infection, almost half had undergone antifungal prophylaxis.
It may signify that antifungal prophylaxis use is not effective in preventing ESFI or, alternatively, that in previously treated ESI there was already a non-identified fungus involved (for which the prophylactic dose of fluconazole was ineffective). A factor that was not addressed and which might have contributed to the development of ESFI is the prophylactic use of antibiotics in the ES, which may be linked to an increased risk of fungal infection19. Bernardini et al20 showed that the prophylactic use of gentamicin in the ES, compared to mupirocin, is also associated to an important percentage (20%) of ESFI, even though it leads to a decrease in the number of ESI and peritonitis. On the other hand, Shan Shan Chen et al21 did not observe an increase in the occurrence of ESFI with the use of gentamicin. In regards to infections previous to ESFI, we observed that, similarly to what occurs in the development of fungal peritonitis3, in the instances where bacterial peritonitis preceded ESFI, it was more commonly caused by Gram negatives, probably related to the fact that Gram-negative infections tend to be more serious and in need of prolonged antibiotic treatment. The same was not observed in cases where ESFI was preceded by bacterial ESI (probably because Gram-negative ESI is significantly less frequent).
Concerning other risk factors, malnutrition may also have significantly contributed. Moreover, the presence of disease/immunosuppressive therapy was identified in a comparatively smaller percentage. In relation to diabetes mellitus, specifically, Rodríguez-Carmona et al22 showed a causal link between poor glycaemic control in diabetic patients in the early stages of PD and the subsequent development of ESI. In our study, this was not a contributing factor to the development of ESFI. There were four episodes (13%) in which no risk factor was identified and that progressed favourably, requiring a shorter span of antifungal therapy, raising the pertinent question of whether treatment is sometimes initiated in cases of local colonization, when clinically irrelevant changes at the orifice of the dialysis catheter adapter precipitated a diagnosis of infection when there was no infection. Furthermore, in 23% of total episodes and in 50% of those which required surgical treatment, the isolation of the fungus was concomitant or posterior to bacterial isolation, which casts doubt on the effective contribution of the fungal agent to the infection. Contrary to what is observed in most Candida infections, including peritonitis3, the most prevalent species was not the albicans but the parapsilosis. Although in relation to peritonitis, this agent is already predominant in certain series9-11. The recommended treatment for mucocutaneous candidiasis rests fundamentally on antifungal therapy with topical or systemic azoles, according to guidelines by the Infectious Diseases Society of America12,13.
Candida parapsilosis is generally highly susceptible to most antifungal agents13, which might explain the favourable clinical progress of most ESFI in this study.
Hsu WD et al28 report the effective use of topical sulconazole for four weeks in the treatment of four cases of ESI to Candida parapsilosis. On the other hand, it is known that some species of Candida, especially glabrata, are resistant to azoles12,24 (the only therapy used in this study), which might have been an important factor in the instances of medical therapy failure. However, that was not analyzed. The sensitivity tests are especially useful in these cases of infection to non-albicans Candida species12. The length of antifungal therapy was generally considerable, although it was substantially greater in the patient group submitted to surgical therapy. Yamaguchi et al25 compared the use of prolonged antibiotherapy with peritoneal catheter substitution in resistant ESI and concluded that early peritoneal catheter substitution (in the absence of internal cuff infection) seems to constitute a simple and effective method in the treatment of intractable ESI, allowing the patient to continue PD and avoid being transferred to haemodialysis, suggesting that the substitution is evaluated after two weeks of antibiotherapy with no infection resolution. In our study, the four cases of ESFI submitted to catheter substitution under antifungal therapy progressed favourably, effectively resolving the infection. External cuff shaving, however, was not an effective measure in the treatment of these infections. Just as with other aspects of ESFI, the most effective therapeutic approach is still to be ascertained. There are studies with divergent results26,27 in regards to the establishment of a consistent causal link between ESI and subsequent peritonitis, but it is believed that peritonitis occurred due to the persistence in colonization of the peritoneal catheter by the fungal agent.
Nevertheless, in our experience, the majority of ESFI showed a favourable clinical evolution under medical treatment, although it is wise to consider removal or replacement of the catheter in the absence of response to prolonged antifungal therapy.
CONCLUSION
Although relatively uncommon, fungal infections represent a complication of PD with relevant prognostic implications. Concomitant infection with bacterial agents is still frequent; therefore the correct diagnosis and distinction between infection or colonization by fungi might be a challenge. The most relevant factor for the development of ESFI was previous systemic antibiotherapy. Shaving of the external cuff does not seem to be an effective therapeutic approach; therefore, peritoneal catheter removal or substitution must always be considered to control the infection. Additional studies that contribute to a more precise ESFI characterization are required, mainly in terms of clinical repercussions, progress and effective therapy and, above all, regarding patient morbidity and technique survival.
References
1. Li PK, Szeto CC, Piraino B, Bernardini J, Figueiredo AE, Gupta A, et al. on behalf of the International Society for Peritoneal Dialysis. Peritoneal dialysis-related infections recommendations: 2010 update. Perit Dial Int 2010; 30:393–423 [Erratum in: Perit Dial Int 2011; 31:512]. [ Links ]
2. Burkart JM, Bleyer A. Tunnel and peritoneal catheter exit site infections in continuous peritoneal dialysis. UpToDate; Mar 31, 2016. [ Links ]
3. Matuszkiewicz-Rowinska J. Update on fungal peritonitis and its treatment. Perit Dial Int 2009; 29(2):161–165. [ Links ]
4. Cox GM, Kauffman CA, Schwab SJ. Fungal peritonitis in continuous peritoneal dialysis. UpToDate; May 18, 2015. [ Links ]
5. Restrepo C, Chacon J, Manjarres G. Fungal peritonitis in peritoneal dialysis patients: successful prophylaxis with fluconazole, as demonstrated by prospective randomized control trial. Perit Dial Int 2010; 30(6):619-625. [ Links ]
6. Záruba K, Peters J, Jungbluth H. Successful prophylaxis for fungal peritonitis in patients on continuous ambulatory peritoneal dialysis: six years experience. Am J Kidney Dis 1991; 17(6):726. [ Links ]
7. Wadhwa NK, Suh H, Cabralda T. Antifungal prophylaxis for secondary fungal peritonitis in peritoneal dialysis patients. Adv Perit Dial 1996; 12:189–91. [ Links ]
8. Kleinpeter MA. Successful treatment of Candida infections in peritoneal dialysis patients: case reports and review of the literature. Adv Perit Dial 2004; 20:58–61. [ Links ]
9. Prasad KN, Prasad N, Gupta A, Sharma RK, Verma AK, Ayyagari A. Fungal peritonitis in patients on continuous ambulatory peritoneal dialysis: a single centre Indian experience. J Infect 2004; 48:96–101. [ Links ]
10. Wang AY, Yu AW, Li PK, Lam PK, Leung CB, Lai KN, Lui SF. Factors predicting outcome of fungal peritonitis in peritoneal dialysis: analysis of a 9-year experience of fungal peritonitis in a single center. Am J Kidney Dis 2000; 36:1183–1192. [ Links ]
11. Kaitwatcharachai C. Candida parapsilosis peritonitis in patients on CAPD. Mycopathologia 2002; 154(4):181–184. [ Links ]
12. Rex JH, Walsh TJ, Sobel JD, Filler SG, Pappas PG, Dismukes WE, Edwards JE. Practice Guidelines for the Treatment of Candidiasis. Clin Infect Dis 2000; 30:662-678. [ Links ]
13. Schell WA. Biology of Candida infections. UpToDate; Set 21, 2015. [ Links ]
14. Kauffman CA. Overview of Candida infections. UpToDate; Jan 5, 2016. [ Links ]
15. Kauffman CA. Epidemiology and pathogenesis of candidemia in adults. UpToDate; Nov 11, 2016. [ Links ]
16. Luzar MA. Exit-Site Infection in Continuous Ambulatory Peritoneal Dialysis: a Review. Perit Dial Int 1991; 11:333-340. [ Links ]
17. Santos C, Pérez-Fontán M, Calvo-Rodríguez M, López-Muñiz A, López-Claviño B, García-Falcón T. Identification of targets for prevention of peritoneal catheter tunnel and exit-site infections in low incidence settings. Perit Dial Int 2016; 36(1):43-51. [ Links ]
18. Moreiras-Plaza M, Vello-Román A, Sampróm-Rodríguez M, Feijóo-Piñeiro D. Ten years without fungal peritonitis: a single centers experience. Perit Dial Int 2007; 27:460–462. [ Links ]
19. Piraino B, Bernardini J, Brown E, Figueiredo A, Johnson DW, Lye WC, Price V, Ramalakshmi S, Szeto CC. ISPD position statement on reducing the risks of peritoneal dialysis–related infections. Perit Dial Int 2011; 31(6):614–630. [ Links ]
20. Bernardini J, Bender F, Florio T, Sloand J, PalmMontalbano L, Fried L, Piraino B. Randomized, double-blind trial of antibiotic exit site cream for prevention of exit site infection in peritoneal dialysis patients. J Am Soc Nephrol 2005; 16(2):539-545. [ Links ]
21. Chen SS, Sheth H, Piraino B, Bender F. Long-term exit-site gentamicin prophylaxis and gentamicin resistance in a peritoneal dialysis program. Perit Dial Int 2016; 36(4):387-389. [ Links ]
22. Rodríguez-Carmona A, Pérez-Fontán M, López-Muñiz A, Ferreiro-Hermida T, García-Fálcon T. Correlation between glycemic control and the incidence of peritoneal and catheter tunnel and exit-site infections in diabetic patients undergoing peritoneal dialysis. Perit Dial Int 2014; 34(6):618–626. [ Links ]
23. Pappas PG, Kauffman CA, Andes D, Benjamin DK, Calandra TF, Edwards JE, Filler SG, Fisher JF, Kullberg BJ, Ostrosky-Zeichner L, Reboli AC, Rex JH, Walsh TJ, Sobel JD. Clinical Practice Guidelines for the Management of Candidiasis: 2009 Update by the Infectious Diseases Society of America. Clin Infect Dis 2009; 48(5):503-535. [ Links ]
24. Kauffman CA. Treatment of candidemia and invasive candidiasis in adults. UpToDate; Nov 11, 2016. [ Links ]
25. Muraoka K, Ishibashi Y, Yamaguchi J, Kawarazaki H, Kume H, Fujita T. Early partial reimplantation of Tenckhoff catheters to treat intractable exit-site or tunnel infection. Perit Dial Int 2011; 31(3):350–353. [ Links ]
26. van Diepen AT, Jassal SV. A qualitative systematic review of the literature supporting a causal relationship between exit-site infection and subsequent peritonitis in patients with end-stage renal disease treated with peritoneal dialysis. Perit Dial Int 2013; 33(6):604-610. [ Links ]
27. Lloyd A, Tangri N, Shafer LA, Rigatto C, Perl J, Komenda P, Sood MM. The risk of peritonitis after an exit site infection: a time-matched, case–control study. Nephrol Dial Transplant 2013; 28:1915–1921. [ Links ]
28. Hsu WD, Lin SL, Wu FL, Chiang CK, Wu KD. Topical antifungal treatment cures exit-site fungal infection. Am J Kidney Dis 2002; 40(4):15. [ Links ]
29. Lo WK, Bargman JM, Burkart J, Krediet RT, Pollock C, Kawanishi H, Blake PG. Guideline on Targets for Solute and Fluid Removal in Adult Patients on Chronic Peritoneal Dialysis. Perit Dial Int 2006; 26(5):520–522. [ Links ]
30. Yoshino A, Honda M, Ikeda M, Tsuchida S, Hataya H, Sakazume S, Tanaka Y, Shishido S, Nakai H. Merit of the cuff-shaving procedure in children with chronic infection. Pediatr Nephrol 2004; 19(11):1267–1272. [ Links ]
31. Luzar MA, Brown CB, Balf D, Hill L, Issad B, Monnier B, Moulart J, Sabatier JC, Wauquier JP, Peluso F. Exit-site care and exit-site infection in continuous ambulatory peritoneal dialysis (CAPD): results of a randomized multicenter trial. Perit Dial Int 1990; 10(1):25-29. [ Links ]
32. Mitra A, Teitelbaum I. Is it safe to simultaneously remove and replace infected peritoneal dialysis catheters? Review of the literature and suggested guidelines. Adv Perit Dial 2003; 19:255-259. [ Links ]
Sofia Semedo Coelho
Rua da Esperança, lote 547, Pinhal de Frades, 2840-310 Seixal
E-mail: sofiasc17@gmail.com
Disclosure of potential conflicts of interest: none declared.
All authors declare that they have no competing interests and have had no involvements that might raise the question of bias in the work reported or in the conclusions, implications, or opinions stated.
No funding was obtained for this study.
Received for publication: May 7, 2016
Accepted in revised form: May 16, 2016














