Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Portuguese Journal of Nephrology & Hypertension
Print version ISSN 0872-0169
Port J Nephrol Hypert vol.28 no.2 Lisboa June 2014
ORIGINAL ARTICLE
Renal Disease in Madeira Islands: twenty-seven years' experience of native kidney biopsies
Doença Renal na Região Autónoma da Madeira: experiência de vinte e sete anos de biópsias de rim nativo
Pedro Vieira1, Antonio M Goncalves1, Jose M Duraes1, Nuno G Rosa1, Luis Resende1, Gil Silva1, Alves Teixeira1, Fernando Rodrigues1, Maria J Galvão2, Ana Brinca2, Helena Viana2, Fernanda Carvalho2, Jose A Araujo1
1 Nephrology Department, Hospital Central do Funchal. Funchal, Portugal
2 Renal Morphology Laboratory, Hospital de Curry Cabral. Lisboa, Portugal
ABSTRACT
Introduction: Renal biopsy plays an essential role either in the diagnosis or in the prognosis of patients with renal disease. In order to assess its epidemiology and evolution in Madeira Islands, we analysed twenty-seven years of native kidney biopsies. Methods: We performed a retrospective analysis of clinical records, including histological revision from 1986 to 2012, totalling 315 native kidney biopsies. They were assessed regarding the temporal evolution both for the quality/indications for renal biopsy and for the patterns of kidney disease. Results: A total of 315 native kidney biopsies were analysed. The patients mean age was of 40.8 ± 18.4 years and 50.5%(n = 159) were males. The most common indications for renal biopsy were nephrotic syndrome (36.2%, n = 114) and acute kidney injury (20.0%, n = 63). Among primary glomerular diseases (41.5%, n = 115) the most common were IgA nephropathy (26.1%, n = 30) and focal-segmental glomerulosclerosis (17.4%, n = 20) and among secondary glomerular diseases (31.4%, n = 87), lupus nephritis (51.7%, n = 45) and amyloidosis (20.7%, n = 18). Statistical analysis revealed significant correlation between gender and major pathological diagnosis (Fishers exact test, p <.01) and between indications for renal biopsy and major pathological diagnosis (χ2, p <.01). Regarding the temporal evolution, no statistically significant differences were found in the number of renal biopsies (χ2, p =.193), number of glomeruli per sample (Fishers exact test, p =.669), age (Kruskal-Wallis, p =.216), indications for renal biopsy (χ2, p =.106) or major pathological diagnosis groups (χ2, p =.649). However, considering the specific clinico-pathological diagnoses and their temporal variation, a statistically significant difference (Fishers exact test, p <.05) was found for lupus nephritis and membranous nephropathy with an increasing incidence and for amyloidosis with an opposite tendency. Discussion: The review of the native kidney biopsies from a population with particular characteristics, geographically isolated, such as those from Madeira Islands, showed parallel between epidemiological numbers referring to other European subpopulations, allowing simultaneously a comprehensive approach to our renal biopsy policies.
Key-Words: Epidemiology; ; nephropathy; renal biopsy; renal disease.
RESUMO
Introdução: A biopsia renal e fundamental na abordagem diagnostica e no prognostico de doentes com patologias nefrológicas. No sentido de avaliar a epidemiologia das doenças renais na Região Autónoma da Madeira e a sua evolução, analisou-se as biopsias de rim nativo nos ultimos vinte e sete anos. Métodos: Procedeu-se a análise retrospectiva dos registos e laminas histológicas de 1986-2012, com avaliação da evolução temporal na qualidade/indicações para biopsia renal e padrões de patologias nefrologicas. Resultados: A amostra compreende 315 biopsias de rim nativo, sendo a idade media dos doentes de 40.8 ± 18.4 anos, sendo 50.5% (n = 159) do sexo masculino. As síndromes nefrologicas mais frequentes foram a sindrome nefrotica (36.2%, n = 114) e lesão renal aguda (20.0%, n = 63). Das patologias nefrologicas mais frequentes, destacam-se no grupo das glomerulopatias primárias (41.5%, n = 115), a nefropatia de IgA (26.1%, n = 30) e a glomeruloesclerose segmentar e focal (17.4%, n = 20) e nas glomerulopatias secundarias (31.4%, n = 87), a nefrite lupica (51.7%, n = 45) e amiloidose (20.7%, n = 18). Na analise estatistica, realca-se a correlação significativa entre sexo e grupos de sindromes nefrologicas (Fishers exact test, p<.01) e entre grupos de sindromes nefrologicas e diagnostico patologicos (χ2, p <.01). Avaliando a evolução temporal, não se objectivaram diferencas estatisticamente significativas em relação ao numero de biopsias renais (χ2, p =.193), numero de glomerulos na amostra (Fishers exact test, p =.669), idade (Kruskal-Wallis, p =.216), sindromes nefrologicas (χ2, p =.106) ou grandes grupos de diagnostico patologicos (χ2, p =.649).
No entanto, considerando os dignósticos patologicos especificos e a sua variação temporal foram encontradas diferencas significativas (Fishers exact test, p <.05) para nefrite lupica e nefropatia membranosa com uma incidencia crescente e para a amiloidose com uma tendência oposta. Discussão: A analise do registo de biopsias renais de uma população com características particulares, isolada geograficamente, permitiu mostrar concordância com outros números epidemiológicos referentes a outras subpopulações europeias, permitindo simultaneamente uma compreensão das directrizes locais sobre biopsias renais.
Palavras-chave: Biopsia renal; doença renal; epidemiologia; glomerulonefrite; nefropatia.
INTRODUCTION
Renal biopsy was introduced into regular clinical practice in the early 1950s and, since then, it has become an essential tool in assessing the diagnosis, providing information of prognostic value and guiding the treatment of patients with renal disease1.
However, given its invasive nature and despite the advances in the renal biopsy technique with the development and utilization of real-time ultrasound guidance and automated biopsy needles2 there are still inherent risks and one must balance the pros and cons considering each patient. In a broader perspective, reviewing the epidemiology of renal diseases besides assessing its incidence, prevalence and trends may help to identify environmental or genetic factors contributing towards disease development and other relevant demographical, clinical and histological features, as well as the influence from single-centre biopsy policies.
Given the particular characteristics of the Madeira Islands, an archipelago geographically isolated in the North Atlantic Ocean comprising about 250,000 inhabitants, a virtually self-contained community with only one major medical provider facility makes it exceptionally suited for these observational studies. Therefore, we reviewed twentyseven years experience of native kidney biopsies in order to assess its renal disease epidemiology and evolution.
SUBJECTS AND METHODS
This retrospective study identified all patients for whom a native kidney biopsy was performed, between January 1986 and December 2012, in the Nephrology Department of Hospital Central do Funchal, the single referral nephrology centre for Madeira Islands. During this time period, a total of 315 native kidney biopsies were performed, including children (< 16 years) and adult patients (≥ 16 years).
The renal biopsy technique in our centre has evolved during the time period the study refers to.
In the beginning, the technique used was by computerized tomography marking and then the biopsy itself was accomplished using a 14G Vim Silverman needle. Since 1995, we started using a standard real-time ultrasound-guided technique with an automated spring-fired biopsy gun and a 14G disposable needle (Bard Biopsy SystemsR, Bard, UK). All renal biopsy specimens were then dispatched along with the clinical information (medical history, physical examination and laboratory data) and analysed at a Renal Pathology Laboratory in Curry Cabral Hospital, Lisbon. All biopsy specimens were evaluated by optical microscopy and immunofluorescence (IF) whenever possible, using standard procedures. Electron microscopy was available only for selected cases.
The renal biopsies were studied by means of routine staining: haematoxylin-eosin, periodic acid-Schiff, Massons trichrome and methenamine silver and, in selected cases, with other histochemical stains. For cases without adequate frozen tissue for IF, the formalin-fixed, paraffin-embedded renal tissue was used for IF.
For each case, final diagnosis was then established concerning the correlation between both clinical and histological investigations. The reports were then forwarded back to the referring nephrologist and the renal biopsy specimens were stored in case future revision was needed. From the clinical information provided, an indication for biopsy was determined, according to the following clinical syndromes3:
1. Nephrotic syndrome (NS): defined as proteinuria > 3.5 g/d with hypoalbuminemia (serum albumin < 2.5 g/dl) with or without peripheral oedema;
2. Nephritic syndrome (NiS): defined as haematuria, hypertension, oedema and reduced glomerular filtration rate (GFR);
3. Asymptomatic urinary abnormalities (AUA): proteinuria < 3.5 g/d and/or haematuria with more than 3 red blood cells per field without clinical manifestations;
4. Acute kidney injury (AKI): rapid deterioration of GFR, including acute on chronic, with or without oligoanuria or rapidly progressive renal failure;
5. Chronic kidney disease (CKD): decline in renal function less severe and/or less abrupt than defined for AKI, with or without proteinuria and/or haematuria. (Patients with NS and chronic-progressive kidney injury were categorized under NS)
The histological findings were classified as follows, using a modified classification scheme of World Health Organization recommendations4-6:
1. Primary glomerulonephritis (GN): minimal change disease (MCD), focal segmental glomerulosclerosis (FSGS), membranous nephropathy (MN), IgA nephropathy (IgAN), mesangioproliferative GN other than IgAN (MesGN), crescentic GN (CrGN)(not fulfilling the criteria for systemic disease), membranoproliferative GN (MPGN) and proliferative endocapillary GN (PEGN);
2. Secondary GN: lupus nephritis (LN), systemic vasculitis (VAS), anti-glomerular basement membrane disease (antiGBM), Henoch-Schonlein purpura (HSP), renal amyloidosis and GN caused by dysgammaglobulinaemia or paraproteinaemia, such as light-chain deposit disease (LCDD), myeloma kidney (MM), essential cryoglobulinaemia and Waldenstroms macroglobulinaemia, diabetic nephropathy (DN), GN associated with infectious diseases (non-streptoccocal GN, endocarditis, shunt GN and others) and hereditary GN;
3. Tubulointerstitial diseases, including acute and chronic tubulointerstitial nephritis and acute tubular necrosis;
4. Vascular diseases, including benign and malignant nephroangiosclerosis (NAS), thrombotic microangiopathy and cortical necrosis;
5. Other findings including end-stage renal disease (ESRD) of undetermined cause, unclassified nephropathies, others such as pregnancy related nephropathies, rare nephropathies and normal renal tissue samples.
STATISTICAL ANALYSIS
Statistical analysis was performed using SPPS, version 17.0 (SPSS, Chicago, IL), considering three inclusive 9-year time frames: 1986–1994, 1995–2003 and 2004–2012. Chi-square test and Fishers exact test were used for categorical variables, and Kruskal-Wallis test for ordinal variables. P-values <.05 were considered statistically significant.
RESULTS
Over the 27 year study, a total of 315 renal biopsies were performed, totalling 295 patients given that 20 records were identified as re-biopsy, with 10 of these carried out due to inadequate sampling during the first biopsy, and 10 for therapeutic/diagnostic reasons at different time points of follow-up (range 6–118 months). The incidence of renal biopsies, considering the 9-year time frames varied between 53.3 per million population (p.m.p.)/year in 1986–1994 to 42.6 p.m.p./year in 1995–2003 and to 44.0 p.m.p./year in 2004–2012, though there were no statistically significant differences (χ2, p =.193).
In what concerns sampling adequacy, renal tissue was obtained in 277 renal biopsies (87,9%) and in spite of the changing renal biopsy technique, no statistical differences were found concerning the number of glomeruli per sample (Fishers exact test, p =.669). Regarding the demographic variables, the patients mean age was of 40.8 ± 18.4 years and 50.5% (n = 159) were male, including children < 16 years (n = 17, 5.4%). The patients mean age was compared according to the previously established time-frames, but no significant variability was found (Kruskal-Wallis, p =.216).
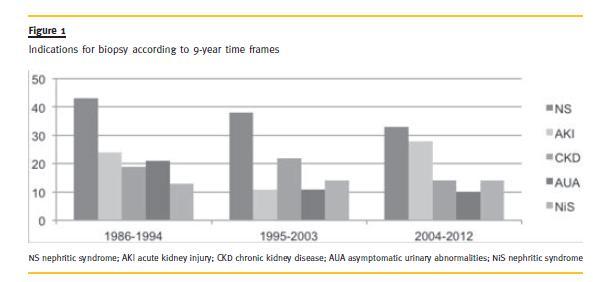
The most common indications for a biopsy were NS (n = 114, 36.2%) and AKI (n = 63, 20.0%) followed by CKD (n = 55, 17.5%), AUA (n = 42, 13.3%) and NiS (n = 41, 13.0%) and once more the timeframed analysis showed no statistically significant differences (χ2, p =.106) (Fig. 1).
The frequency of the clinico-histological diagnoses is shown in Table I. Overall, among the major pathological groups, primary GN was the most common diagnosis (41.5%), followed by secondary GN (31.4%), tubulointerstitial diseases (7.6%), vascular diseases (3.6%) and other findings (15.9%). Considering the specific diagnosis, the most common type of primary GN were IgAN (n = 30, 26.1%), FSGS (n = 20, 17.4%) and MCD (n = 19, 16.5%) and amid the secondary GN the most common were LN (n = 45, 51.7%), amyloidosis (n = 18, 20.7%) and DN (n = 11, 12.6%).
In view of the temporal evolution, according to the time-frames previously established, no statistically significant differences were found regarding the major pathological groups (χ2, p =.649). Considering the specific clinico-pathological diagnoses, its distribution according to temporal evolution is shown in Fig. 2 and taking into account the statistical analysis of the most common types of specific diagnoses, the temporal evolution shows a statistically significant difference (Fishers exact test, p <.05) only for LN and MN with an increasing incidence and for amyloidosis with an opposite tendency. For informational purposes we have subdivided LN and amyloidosis according to classes and subtypes, respectively. Among LN, the histopathological classes most found were class IV (51.1%), class II (26.7%) and class III (17.8%) and as amyloidosis is referred AA subtype (66.7%) and AL (22.2%) were the most frequent. Once we had established a decreasing incidence of amyloidosis, we analysed the subset of AA, AL and other amyloidosis for temporal variation having found no statistically significant differences (p =.14, p =.74 and p =.34, respectively). Correlations between gender, indications for renal biopsy and clinico-histological diagnoses are shown in Table II and, as one can observe, statistically significant differences were found between gender and major pathological diagnoses (Fishers exact test, p <.01) and between indications for renal biopsy and major pathological diagnosis (χ2, p <.01).
DISCUSSION
This retrospective study enables a comprehensive approach to our centre biopsy policies and their adjustments in the past twenty-seven years. According to the demographical and clinical analysis, we found no statistically significant differences in what concerns number of biopsies performed, sampling adequacy and indications for biopsy. This may suggest, on one hand, a stable approach and workup for nephropathies and, acknowledged the indispensable evolution in the kidney biopsy technique, that the sampling adequacy kept the same standards. On the other hand, given the small sized sample the detection of any underlying statistically significant differences may be underpowered.
Regarding the renal biopsy rate in our study, the numbers are quite similar to those described in almost all European registries (48 p.m.p./year in Spain7, 34 p.m.p./ year in Italy6, 16.3–20.1 p.m.p./year in France8, 44.1–69.3 p.m.p./year in the Czech Republic9, 40 p.m.p./year in Denmark10 and to those of our own national registry, 61.4 p.m.p./year in 2011-2012) apart from some registries whose numbers are somewhat uneven due to particular reasons (10.8 p.m.p./year en Serbia11, 10.7-12.8 p.m.p./ year in Romania12, 126.3 p.m.p./year in Scotland13 and 176 p.m.p./year in Finland14. Whereas indications for biopsy are concerned, NS was our most frequent clinical presentation as in some other registries (7, 9, 11, 12), however we found some divergences with other registries as the AUA came responsible only for 13.3% while in many other studies it imposed itself as a more frequent indication for biopsy (24.3-38.7%)(6, 7, 9, 11, 14). This may foretell a somewhat conservative approach at our centre in terms of kidney biopsy as the workup for these clinical presentations or, as Okpechi suggests, it may reflect healthcare investment15. Our report shows that, among the major pathological groups, primary GN stands as the most common (41.5%), followed by secondary GN (31.4%) what mirrors other registries results where primary GN varies between 51.5-64.1%(6-9, 11, 12) and secondary GN between 20.4-26.4%(6, 7, 9, 11, 12). Addressing the specific diagnosis analysis we recognized IgAN (26.1%) as the most common primary GN, as in other European subpopulations (6-9, 14), and LN (51.7%) as the most frequent among the secondary GN going in line not only with European subpopulations, but also with worldwide biopsy-provenglomerular diseases16, both of which go according to our own national registry.
Deliberately, we have revised the Canary Islands Registry of renal biopsies (17, 18) separately, since it somehow presents with similar characteristics to those of our own. As a result, we found parallel epidemiological data among pathological groups, with primary GN attaining top place with 39.8%, and its subset of specific diagnosis showing also similar distribution with IgAN being the most frequent (36.6%), followed by MCD (18.1%) and FSGS (17.1%), with the latter two assuming an inverse order when compared to our data. However, we would like to draw attention to the differences found in the prevalence of certain glomerulopathies among different provinces in the Canary Islands, mainly as FSGS is concerned, what seems to be related to a different genetic load on each province and set the authors in search of genetic mutations17.
Examining the results of the epidemiological analysis in relation to temporal variation we found no significant differences in the major pathological diagnoses, nonetheless, considering the most common specific diagnoses and assessing their variation along the time periods we found, on the one hand, statistically significant increasing incidence in biopsy-proven LN and MN and, on the other, decreasing incidence among amyloidosis. Focusing on the absence of temporal variation of IgANs incidence, as shown in other series19, this may reflect once more the conservative biopsy policies. Then again, LNs raise is probably due to both low nephrology referral in the early period the study refers to and re-biopsying relapses for therapeutic purposes, while the increasing incidence in MN is somewhat similar to that described in other registries (6, 7). Taking into consideration the decreasing incidence of amyloidosis, the subset analysis for amyloidosis (AA, AL and others) despite not having found statistical significance among them, demonstrated a higher tendency (=.14) for amyloidosis AA as being the reason for the decline for amyloidosis. This decline in the incidence of amyloidosis contrasts with the results of the Italian Registry20 but goes along with those from Spain21. In our study, the decrease is mainly justified by the AA form, what previous studies(21, 22) suggest being due to advances in the treatment of chronic infections and autoimmune inflammatory diseases, thus reflecting an increasing healthcare investment and leaving the aforementioned AUAs low prevalence as indication for kidney biopsy as probably resulting from a conservative approach at our centre. In the course of the subset analysis of amyloidosis, the recognition of four cases of AL amyloidosis caught our eye as none of myeloma kidney or cast nephropathy was diagnosed.
Such result discloses that the true prevalences of different kidney diseases are not always represented by biopsy rates, once cast nephropathy is considered the most common renal pathology in myeloma patients23. However, one must bear in mind that in the case of myeloma patients with cast nephropathy their common clinical contraindications and sometimes presumptive diagnosis avert the definitive diagnosis made by renal biopsy, justifying the discrepancy by one side with necropsy results (11% cases of AL amyloidosis vs. 32% cases of cast nephropathy)24 that reflect more truthful prevalences in opposition to renal biopsy databases revision results (68% cases of AL amyloidosis vs. 17% cases of cast nephropathy)25.
Another finding that drew our attention was that of two cases of TI nephritis presenting with NS (Table II). Although the histological analysis revealed no more than tubular and interstitial changes, the NS suggests the presence of glomerular pathology, thus we thought further individual consideration on these two cases was imperative. Considering the first of the two, an acute interstitial nephritis was clinically interpreted as a case of NSAIDs nephropathy, thus justifying the association with NS as in patients who develop both AIN and minimal change disease(26-28). These cases usually conceal extensive foot process effacement only revealed by electron microscopy that was not performed in this particular situation justifying, therefore, the non-description of glomerular changes by optical microscopy26. The latter was histologically characterized as a chronic interstitial nephritis but no aetiology was clinically plain. In this case, we can only consider a possible sampling bias as only 4 glomerulus were present, what might withhold the finding of glomerular sclerosis in the context of CIN.
Although a broader analysis could enhance our epidemiology knowledge of this geographical region, in view of such a small sample an in depth analysis of temporal evolution, covering all clinico-histological diagnoses rendered itself unattainable.
CONCLUSION
This study allowed us, on the one hand, to acknowledge comprehensively our centre biopsy policies and on other to render information that, despite the geographical isolation of Madeira Islands, showed that the demographical and clinical data of renal diseases is in many aspects similar to that from other European subpopulations. However, as in other similar regions it may conceal some endemic mutations.
References
1 . Iversen P, Brun C. Aspiration biopsy of the kidney. Am J Med 1951;11(3):324-330. [ Links ]
2 . Donovan KL, Thomas DM, Wheeler DC, Macdougall IC, Williams JD. Experience with a new method for percutaneous renal biopsy. Nephrol Dial Transplant 1991;6(10):731-733. [ Links ]
3 . National Kidney Foundation. K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Am J Kidney Dis 2002;39(2 Suppl 1):S1-266. [ Links ]
4 . J C, LH S. Renal disease. Classification and atlas of glomerular diseases. Tokyo: Igaku Shoin,1982. [ Links ]
5 . Polito MG, de Moura LA, Kirsztajn GM. An overview on frequency of renal biopsy diagnosis in Brazil: clinical and pathological patterns based on 9,617 native kidney biopsies. Nephrol Dial Transplant 2010;25(2):490-496. [ Links ]
6 . Schena FP. Survey of the Italian Registry of Renal Biopsies. Frequency of the renal diseases for 7 consecutive years. The Italian Group of Renal Immunopathology. Nephrol Dial Transplant 1997;12(3):418-426. [ Links ]
7 . Rivera F, Lopez-Gomez JM, Perez-Garcia R; Spanish Registry of Glomerulonephritis. Frequency of renal pathology in Spain 1994-1999. Nephrol Dial Transplant 2002;17(9):1594-1602. [ Links ]
8 . Simon P, Ramee MP, Boulahrouz R, et al. Epidemiologic data of primary glomerular diseases in western France. Kidney Int 2004;66(3):905-908. [ Links ]
9 . RychlikI, Jancova E, Tesar V, et al. The Czech registry of renal biopsies. Occurrence of renal diseases in the years 1994-2000. Nephrol Dial Transplant 2004;19(12):3040-3049. [ Links ]
10. Heaf J, Lokkegaard H, Larsen S. The epidemiology and prognosis of glomerulonephritis in Denmark 1985-1997. Nephrol Dial Transplant 1999;14(8):1889-1897. [ Links ]
11. Naumovic R, Pavlovic S, Stojkovic D, Basta-Jovanovic G, Nesic V. Renal biopsy registry from a single centre in Serbia: 20 years of experience. Nephrol Dial Transplant 2009;24(3):877-885. [ Links ]
12. Volovat C, Caruntu I, Costin C, et al. Changes in the histological spectrum of glomerular diseases in the past 16 years in the North-Eastern region of Romania. BMC Nephrol 2013;14:148. [ Links ]
13. McQuarrie EP, Mackinnon B, et al. Centre variation in incidence, indication and diagnosis of adult native renal biopsy in Scotland. Nephrol Dial Transplant 2009;24(5):1524-1528. [ Links ]
14. Wirta O, Mustonen J, Helin H, Pasternack A. Incidence of biopsy-proven glomerulonephritis. Nephrol Dial Transplant 2008;23(1):193-200. [ Links ]
15. Okpechi I, Duffield M, Swanepoel C. Primary glomerular diseases: variations in disease types seen in Africa and Europe. Port J Nephrol Hypert 2012;26(1):25-31. [ Links ]
16. Pesce F, Schena FP. Worldwide distribution of glomerular diseases: the role of renal biopsy registries. Nephrol Dial Transplant 2010;25(2):334-336. [ Links ]
17. Gonzalez Cabrera F, Rodriguez A, Checa MD, Valenciano B, Plaza-Toledano C, Garcia Nieto V. Diferencias regionales en la prevalencia de algunas glomerulopatias en las Islas Canarias. Nefrologia 2009;29(1):83-84. [ Links ]
18. SCN. Registro de Biopsias Renales de la Sociedad Canaria de Nefrologia (1987-1993). In: Nefrologia SCd, editor. XI Reunion de la Sociedad Canaria de Nefrologia; Las Palmas de Gran Canaria1994.
19. Hanko JB, Mullan RN, ORourke DM, McNamee PT, Maxwell AP, Courtney AE. The changing pattern of adult primary glomerular disease. Nephrol Dial Transplant 2009;24(10):3050-3054. [ Links ]
20. Bergesio F, Ciciani AM, Manganaro M, et al. Renal involvement in systemic amyloidosis: an Italian collaborative study on survival and renal outcome. Nephrol Dial Transplant 2008;23(3):941-951. [ Links ]
21. Panizo N, Rivera F, Lopez-Gomez JM; Spanish Registry of Glomerulonephritis. Decreasing incidence of AA amyloidosis in Spain. Eur J Clin Invest 2013;43(12):1371. [ Links ]
22. Hazenberg BP, Van Rijswijk MH. Where has secondary amyloid gone? Ann Rheum Dis 2000;59(8):577-579. [ Links ]
23. Korbet SM, Schwartz MM. Multiple myeloma. J Am Soc Nephrol 2006;17(9):2533-2545. [ Links ]
24. Ivanyi B. Frequency of light chain deposition nephropathy relative to renal amyloidosis and Bence Jones cast nephropathy in a necropsy study of patients with myeloma. Arch Pathol Lab Med 1990;114(9):986-987. [ Links ]
25. Leung N, Gertz M, Kyle RA, et al. Urinary albumin excretion patterns of patients with cast nephropathy and other monoclonal gammopathy-related kidney diseases. Clin J Am Soc Nephrol 2012;7(12):1964-1968. [ Links ]
26. Praga M, Gonzalez E. Acute interstitial nephritis. Kidney Int 2010;77(11):956-961. [ Links ]
27. Warren GV, Korbet SM, Schwartz MM, Lewis EJ. Minimal change glomerulopathy associated with nonsteroidal antiinflammatory drugs. Am J Kidney Dis 1989;13(2):127-130. PubMed PMID: 2629709. [ Links ]
28. Kleinknecht D. Interstitial nephritis, the nephrotic syndrome, and chronic renal failure secondary to nonsteroidal anti-inflammatory drugs. Semin Nephrol 1995;15(3):228-235. [ Links ]
Dr. Pedro Vieira
Department of Nephrology, Hospital Central do Funchal
Avenida Luis de Camões no 57,
9004-514 Funchal, Portugal
E-mail: pedro.mds.vieira@gmail.com
Conflict of interest statement: None declared
Acknowledgments We want to express an immense appreciation to all the staff at the Renal Pathology Laboratory in Curry Cabral Hospital, headed by Fernanda Carvalho.
Received for publication: 28/12/2013
Accepted in revised form: 09/05/2014














