Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Revista de Ciências Agrárias
Print version ISSN 0871-018X
Rev. de Ciências Agrárias vol.42 no.1 Lisboa Mar. 2019
https://doi.org/10.19084/RCA18248
ARTIGO
Alterations in soil salinity with the use of different biochar doses
Alterações na salinidade do solo com o uso de diferentes doses de biocarvão
Josely D. Fernandes, Lúcia H.G. Chaves*, Jacqueline S. Mendes, Iêde B. Chaves and Gilvanise A. Tito
Departamento de Engenharia Agrícola – Universidade Federal de Campina Grande, Avenida Aprígio Veloso, 882, CEP 58429-190, Campina Grande, Paraíba, Brasil
(*E-mail: lhgarofalo@hotmail.com)
ABSTRACT
Biochar produced from a poultry litter has a pH around 10 and a high concentration of basic cations. The objective of this work was to evaluate the effect of this biochar in the salinity of a Yellow Red Latosol. The experiment was conducted in a completely randomized design with four replicates using columns of 20 cm height filled with soil, presenting in the first 5 cm of the column, doses corresponding to 0; 10; 15; 20; 25 and 30 t ha-1 of biochar mixed to the soil, totalizing 24 experimental units. For 30 days, 44 mL of deionized water was applied daily to each column. After this period, soil samples were collected at two depths (0-10; 10-20cm) and analyzed chemically concerning: ECse, pH, potassium, sodium, calcium and magnesium in each experimental plot. From the results SAR and ESP indicators were calculated. The biochar rates increased ECse, pH, Na and K, as well as SAR and ESP, being more pronounced in the first depth (0-10 cm) for most of the analyzed parameters. The soil was classified as non-saline, however, the increase in all the analyzed parameters points to a careful use of the biochar studied.
Keywords: Oxisol, Chicken bed, Soil chemical properties.
RESUMO.
Biocarvão produzido com cama de galinha apresenta um pH de cerca de 10 e uma elevada concentração de cátions básicos. O objetivo deste trabalho consistiu em avaliar o efeito da aplicação do biocarvão na salinidade de um Latossolo Vermelho Amarelo. O ensaio experimental foi conduzido em delineamento inteiramente casualizado com quatro repetições utilizando colunas de 20 cm de altura preenchidas com solo, apresentando nos primeiros 5 cm da coluna, doses correspondentes a 0; 10; 15; 20; 25 e 30 t ha-1 de biocarvão misturadas ao solo, totalizando 24 unidades experimentais. Durante 30 dias, aplicou-se diariamente 44 mL de água desionizada em cada coluna. Após este período, foram retiradas amostras de solo, em cada coluna, em duas profundidades (0-10; 10-20cm) que foram analisadas quimicamente no que respeita a: CEse, pH, potássio, sódio, cálcio e magnésio. A partir dos resultados foram calculados os indicadores RAS e PST. As doses de biocarvão promoveram aumento da CEes, pH, Na e K, bem como da RAS e PST, sendo mais acentuado na primeira profundidade de avaliação para a maioria dos parâmetros analisados. O solo foi classificado como não salino, no entanto, o aumento verificado em todos os parâmetros analisados aponta para uma utilização cuidadosa do biocarvão em estudo.
Palavras-chave: Oxissol, Cama de galinha, propriedades químicas do solo.
INTRODUCTION
From an agricultural point of view, sodic soils are those that present exchangeable sodium in excess, presenting a exchangeable sodium percentage (ESP) of more than 15%, an electrical conductivity (ECse) below 4 dS m-1 and a pH above 8.5. The adverse effects of soil sodicity are very serious and can lead to physical and chemical degradation of soil properties, which can cause the loss of one or more soil functions with consequent reduction of crop production (Singh and Singh, 2013). While soil sodization is the process by which the Na+ ion gains preponderance in the soil exchange complex, soil salinization is a process that leads to increase the concentration of the soil solution in soluble salts (Na+, Ca2+, Mg2+, K+) to levels detrimental to plants. Soil salinization could be due to natural causes, like the weathering of rocks, involving physical, chemical and biological processes, through the action of factors such as climate, relief, living organisms and time, or by human interventions such as inappropriate irrigation practices, use of salt-rich irrigation water and/or poor drainage control (Dias, 2004). Excess salts severely limit agricultural production mainly in the arid and semi-arid regions, where about 25% of the irrigated area is salinized (FAO, 2006).
The recovery of saline/sodic soils generally involves two processes: the leaching of soluble salts (saline soils) and the replacement of exchangeable Na+ by exchangeable Ca2+ (sodic soils). A surplus of irrigation water and correctives such as gypsum, sulfuric acid, elemental sulfur and organic matter could be used. When used correctly, they improve the chemical and physical properties of the soil, facilitating the removal of excess salts and even replacing the exchangeable sodium (Araújo et al., 2017).
Recently, several researches were using biochar as a conditioner in salt affected soils (Wu et al., 2014; Elshaikh et al., 2017; Sappor et al., 2017). Biochar is a carbon residue resulting from the pyrolysis of plant or animal biomass (Lehmann et al., 2006; Verheijen et al., 2010; Kookana et al., 2011). Pyrolysis is a thermal treatment of decomposition of biomass in an environment with high temperatures (> 400°C) and low concentration of oxygen, resulting in gas (mainly hydrogen, methane and carbon monoxide), bio-oil , (alcohols, oils, tars and acids) and biochar (mainly C, O, H, N and ash) (Laird et al., 2009; Novak et al., 2010).
The type of feedstock used in the pyrolysis process significantly influences the properties of the biochar (Silva et al., 2017). In Brazil, a type of available residue is the poultry litter which has been used in the manufacture of biochar. However, their effects on the environment require specific studies but, unfortunately, the knowledge about the impact of this material on soil properties is still scarce.
The objective of this study was to evaluate the effect of a biochar, produced from a poultry litter, in the salinity/sodicity of a Yellow Red Latosol.
MATERIALS AND METHODS
The experiment was carried out in Irrigation and Salinity Laboratory of the Agricultural Engineering Department, UFCG, from May 2017 to July 2017 using soil columns described below. The experimental design was completely randomized, in subdivided plots, with 4 replications. The plots corresponded to the six biochar doses (0; 7.85; 11.77; 15.7; 19.62 and 23.55 g/0.00785m2, according to 0, 10, 15, 20, 25 and 30 t ha-1) and the subplots were the soil depths (0-10 and 10-20 cm).
The biochar used in this study was produced from poultry litter (PL), a solid waste resulting from chicken rearing, under slow pyrolysis, at a temperature of 400 oC, by SPPT Technological Research Ltda. The following attributes were found according to the methodology Brasil (2014): pH = 9.45, P2O5 = 7.78%, K2O = 4.90%, Ca = 6.83%, Mg = 1.34%, Na = 0.73%, S = 0.74%, Fe = 0.46%, Mn = 0.09%, Cu = 0.04%, Zn = 0.08%, B = 0.01%, organic matter = 39.77%..
Soil sample used was collected from the top layer (0-0.20 m) of a Red Yellow Latosol, located in the municipality Areia, State of Paraiba, Brazil. This sample was, then, air-dried for 3 days, sieved with 2 mm screen and characterized according to the methodology EMBRAPA (2011). It presents the following physical and chemical attributes: clay = 305.5 g kg-1, silt = 262.0 g kg-1, sand = 432.5 g kg-1, pH (H2O) = 5.30, Ca = 0.93 cmolc kg-1, Mg = 0.55 cmolc kg-1, Na = 0.02 cmolc kg-1, K = 0.04 cmolc kg-1, H = 1.83 cmolc kg-1, Al = 1.8 cmolc kg-1, P = 3.3 mg kg-1, organic matter = 13.6 g kg-1. .
Soil column was prepared using PVC pipe with diameter of 10 cm and length of 22 cm. In general it is in the first 20 cm of the soil that the chemical reactions between the soil and any corrective occur, and also where we can find the presence of a greater density of roots. Soil sample was put into the column and compacted by knocking the base of the column until the soil height achieved 20 cm; the biochars treatments were incorporated into the top 5 cm of the soil in the columns in order to simulate the disposition of this material on the surface as usually organic matter is applied to the soil. The remaining 2 cm of the upper part were not filled with soil in order to facilitate the addition of water during the percolations. A mesh was attached to the bottom of the columns to prevent soil from falling out and, to facilitate the drainage of the leachates which were collected in containers placed below the columns. The columns were placed in trays containing water to reach the field capacity through the capillary ascension. After 24h, a volume of 44.0 mL of deionized water was applied on each column, daily (during 30 days), simulating a monthly rainfall of 168.15 mm (average for the city of Campina Grande in April).
After that period (30 days), each column was sectioned in two parts, 10 cm each (0-10 cm, denominated top and 10-20 cm, denominated base), dried at room temperature, sieved in a 2 mm mesh. Then, soil samples were analyzed for soil salinity, measuring in the saturation extract the electrical conductivity (ECse) and the soluble cations: sodium, potassium (by flame photometer), calcium and magnesium (by titration).
The indicator used to determine sodicity was the percentage of exchangeable sodium (ESP) based on the ratio between exchangeable sodium, extracted from the soil with Mehlich 1 method and quantified in the extract according to flame photometry, and cation exchange capacity (CEC). The exchangeable sodium percentage was determined from equation 1.
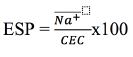 (1)
(1)
Where: ESP is the exchangeable sodium percentage, %.
Na+ is the exchangeable sodium content, cmolc dm-3. CEC is the cation exchange capacity,
cmolc dm-3. It
was also determined the sodium adsorption ratio (SAR), defined by the relationship
between soluble Na+ concentration (mmol L-1) and soluble Ca2+
+ Mg2+ concentration (mmol L-1), according to equation 2. The
data of the soil were submitted to analysis of variance (ANOVA) applying the SISVAR
software (Ferreira, 2011). With the significance of the doses the average of the
parameters evaluated in the soil (plots) were submitted to regression analysis.
These parameters were also analyzed in the soil according to the two depths (subplots)
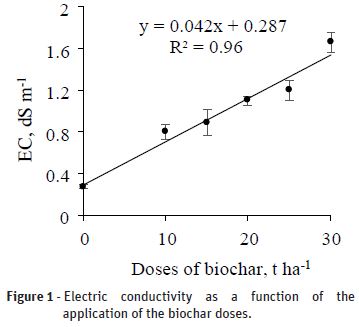
comparing their means to each other within the same dose by the t test. RESULTS AND DISCUSSION Soil electrical conductivity The mean electric conductivity of the saturation
extract (ECse) was adjusted to the linear positive model (Table 1), with an approximate
increase of 0.042 dS m-1 per unit increase (t ha-1) of applied
biochar (Figure 1). Also with respect to Figure 1, with the application of the highest dose (30
t ha-1) the ECse was estimated at 1.55 dS m-1, corresponding
to an increase of 439.9% when compared to ECse of the absolute control. Similar results were observed by Rombolà et al. (2015),
who evaluated changes in the chemical characteristics of soils with the application
of biochar and found that their use promoted an increase in soil salinity. Although the salinity response differs considerably among plant species, seed
germination and plant growth in saline conditions may be impaired, especially in
the seedling phase (Mengel and Kirkby, 1987). In general, salinity
effects are practically insignificant in soil extracts with readings less than or
equal to 2 dS m-1 (Hoekstra et al., 2002). Although this critical
level was not exceeded with the application of biochar, the increase of EC from
0.287 dS m-1 (control) to 1.55 dS m-1 (30 t ha-1)
is worrying given the short period in which the test was conducted. The electrical conductivity did not change significantly as a function of the
analyzed soil layers. This may be associated with the concentration and solubility
of the nutrients present in the biochar that promoted, as well as increased salinity,
an increased flow of solutes and leaching of salts to deeper soil layers. These results differ from those found in the literature (Yue et al.,
2016), who verified a higher concentration of salts in the superficial layer and
decreasing with depth. It is known that materials with high electrical conductivity,
that is, with high salinity, must be handled with caution, because if applied to
the soil in large quantities can cause toxicity in plants (Song and Guo, 2012). Soil pH Biochar promoted
an increase in soil pH mainly in the first soil layer, whose highest estimated value
corresponded to 7.87 with the application of 26.17 t ha-1 (Figure 2A).
In the literature were found results that corroborate those obtained in the present
research, like in the study of Rombolà et al. (2015) who observed an increase
in soil pH in response to application of chicken bed biochar. The alkalinity of biochar favored this increase. Moreover,
the presence of calcium (Ca2+) and bicarbonate (HCO3-)
may have promoted the formation of calcium carbonate (CaCO3) in the soil,
increasing the concentration of hydroxyls (OH-) (Sappor et al.,
2017). In relation to Figure 2A, in the depth of 10-20 cm, the soil pH increased
with the addition of 0.0075 units (t ha-1) of applied biochar. The lower influence of the biochar doses on the
pH variation observed in the second soil layer shows that the chemical reactions
occur preferentially in the superficial layer, where the biochar was applied. This
caused, with the exception of the control, a statistical difference of this attribute
of the soil between the depths analyzed within each biochar dose applied. Calcium and magnesium content The lowest concentration of calcium (0.48 mmolc
L-1) was found in the topsoil (0-10 cm) with the use of 10.16 t ha-1,
but when higher doses were applied, there was an increase in the content of this
nutrient, which had a mean of 4.02 mmolc L-1 at a dose of
30 t ha-1 (Figure 2B). In the second soil layer, the highest concentration
of Ca was estimated at 2.4 mmolc L-1, with 11.63 t ha-1
of biochar. Also, in relation to Figure 2B, it was verified that the depth promoted
significant difference, except in the absence of biochar. In the surface layer,
the highest concentrations of Ca occurred only with the use of the two largest biochar. The mean values of magnesium content, regardless
of the analyzed depth, presented the opposite behavior to that observed for calcium
(Figure 2C), that is, the increase in calcium concentration in the saturation extract
decreased to magnesium, and vice versa. This behavior may be the result of antagonism
between these nutrients, so that calcium had preference to be precipitated. The addition of bivalent cations, such as Ca2+ and Mg2+,
is essential in the recovery of soils affected by salts, compensating for excess
exchangeable Na+. The biochar used in this research is shown to be able
to play a positive role in this respect. These results corroborate Major et al.
(2010) who observed higher availability of Ca2+and Mg2+ after
addition of biochar at a rate of 20 t ha-1 to a Latosol. Potassium content Potassium
showed similar behavior at both depths, ie, at 4.20 and 4.17 mmolc L-1,
for the first and second soil layers, respectively, with 30 t ha-1 of
biochar (Figure 2D). Also with respect to this figure, K concentrations differed
between the depths at the 10 and 15 t ha-1 dosages. The exchangeable
cations present a retention energy to the soil colloids following a lyotropic series
(Al3+ > Ca2+ > Mg2+ > NH4+
> K+ > H+ > Na+ ) that considers the charge and the size of the
ion hydrated; in this series, potassium, being a monovalent cation, occupies the
fifth place (Duarte et al., 2013). As biochar increased soil salinity due
to the availability of nutrients, potassium competed with calcium and magnesium
at the exchange sites, thus, increases in the concentration of K in the saturation
extract at the two depths as a function of the applied doses evidenced the mobility
of the same in the leach column. According
to Walker and Bernal (2008), the use of chicken litter improves the chemical properties
of a soil affected by salts, increasing both CEC and soluble and exchangeable K+,
so potassium competes with Na+ in terms of adsorption limiting their
entry into exchange places. Chloride and bicarbonate content Biochar also promoted increases in chloride and
bicarbonate content. The maximum chloride concentration in the first soil layer
corresponded to 5.02 mmolc L-1, with 24.15 t ha-1,
and in the subsequent layer, its content increased proportionally with the application
of the doses, whose unit increase corresponded to 0.1224 mmolc L-1,
per t ha-1 of applied biochar (Figure 2E). Chloride accumulation can
promote a competitive effect with nitrate and sulfate by exchange sites in the soil-plant
system, which may affect crop productivity (Santos et al., 1984). As for
bicarbonate (Figure 2F), their means were adjusted independently of depth to the
second-order polynomial regression model, whose maximum values corresponded to 1.83
and 1.45 mmolc L-1 in the first and second layers respectively.
According to Garg and Garg (1980) the carbonate / bicarbonate can lead to sodification
of the soil, besides decreasing the concentration of calcium and magnesium in the
soil due to the precipitation of these. In addition, it can affect nutrient uptake
by increasing pH, decreasing the availability of soil nutrients (Paliwal et al.,
1978). Exchangeable sodium percentage (ESP) In relation to the percentage of exchangeable
sodium (ESP), the application of biochar significantly increased this parameter
in both layers of soil analyzed, reaching maximum values of 7.74% and 5.23% in the
first and second depth, respectively, representing an increase of 203.53% and 224.18%
when compared to the absolute control (Figure 2G). Still in
relation to Figure 2G, the ESP averages were higher in the first depth differing
significantly from those recorded in the second layer regardless of the applied
dose. The result obtained in this research differs from Chaganti et al. (2015),
who found a significant reduction of soil ESP with the use of biochar, when evaluating
the remediation of saline-sodic soils with organic inputs. Sodium content and sodium adsorption ratio (SAR) Sodium and sodium adsorption ratio (SAR) presented similar behavior, that
is, their contents were higher in the treatments that received the biochar, presenting
as higher averages 0.40 and 0.27 mmolc L-1, respectively,
with use of 30 t ha-1 (Figures 3A and 3C). When the influence of the
layers was analyzed, the Na+ and SAR concentrations were higher in the
depth of 10-20 cm (Figures 3B and 3D). This result suggests that sodium showed good
mobility within the leaching column. This is because the enrichment of exchangeable
sites of the soil profile with Ca2+ and Mg2+ may decrease
the exchangeable Na+ concentration at these sites, making it soluble
in the soil solution and therefore more susceptible to leaching. Due
to the increase in sodium, the indicators exchangeable sodium (ESP) and sodium adsorption
ratio (SAR) presented higher averages in the treatments that received the biochar,
which provides greater possibility of soil sodicity problems, which suggests caution
in its use. These results corroborate Subhan et al. (2015) who reported increase
in SAR due to high Na+ concentrations in the biochar produced from cotton
stems. Salt affected soils are generally classified based on the electrical conductivity
of the saturated extract (CEse), the exchangeable sodium percentage (ESP) and the
pH (Richard, 1954). Based on these properties, the soil used in this research was
classified as normal, at the end of the experiment, because it presented an ECse
<4 dS m-1, an ESP <15 and a pH less than 8.5. However, even if
there are no salt problems, the increase in all parameters analyzed in a short period
(30 days) points to caution regarding the use of biochar studied. The results found in the literature are contradictory. Zhang et al. (2016),
after evaluating the influence of different types of biochar in the chemical properties
of two soils, a sandy and a clay one, verified increase of salinity for both soils.
However, Lashari et al. (2013), showed a significant decrease in soil pH
and salt and sodium contents with the application of a biochar. Therefore, the raw
material used to produce the biochar is the key factor that determines its effectiveness
as an organic soil conditioner. Current data on salinization or recovery of salt/sodic
affected soils with biochar are inconsistent and it is difficult to compare the
existing studies in the literature with each other. This is probably due to the
large variation between the biochar composition and the diversity of soils used
in the different studies. In addition, there is a lack of long-term field experiments
to verify the mechanisms observed in controlled conditions like experiments in pots
or incubation studies. CONCLUSION Biochar increased all the salinity parameters analyzed, however,
regardless of the applied dose and depth analyzed, the soil was still classified
as a non-saline. However, the increase in all the analyzed parameters points to
a careful use of the biochar studied. Further research is needed to evaluate the effect of different doses of chicken
litter biochar for a longer incubation period, using soils with different salinity
levels, in order to verify possible changes in soil physicochemical properties. The composition of the raw material used to produce
the biochar is a key factor that determines its effectiveness as an organic soil
conditioner. REFERENCES Araújo, M.M.; Sousa, G.M. & Rodrigues, L.R.C.C. (2017) -
Técnicas utilizadas na recuperação de solos afetados por sais. Revista de Agroecologia
no Semiárido, vol. 1, n. 2, p. 1-11. [ Links ] Brasil (2014) - Manual de métodos analíticos oficiais para fertilizantes
minerais, orgânicos, organominerais e corretivos. Ministério da Agricultura,
Pecuária e Abastecimento. Secretaria de Defesa Agropecuária. Coordenação-Geral de
Apoio Laboratorial, Murilo Carlos Muniz Veras (Org.) – Brasília: MAPA/ SDA/CGAL,
220 p. Chaganti, V.N.; Crohn, D.M.
& Simunek, J. (2015) - Leaching and reclamation of a biochar and compost amended
saline–sodic soil with moderate SAR reclaimed water. Agricultural Water Management,
vol.158, p. 255–265. https://doi.org/10.1016/j.agwat.2015.05.016 [ Links ] Dias, N.S. (2004) - Manejo da fertirrigação e controle
da salinidade em solo cultivado com melão rendilhado sob ambiente protegido.
110 f. Tese (Doutorado em irrigação e Drenagem) - Escola Superior de Agricultura
Luiz de Queiroz - Universidade de São Paulo. Piracicaba. [ Links ] Duarte, I.N.; Pereira, H.S. & Korndoifer, G.H.
(2013) - Lixiviação de potássio proveniente do termopotássio. Pesquisa Agropecuária
Tropical, vol. 43, n. 2, p. 195-200. [ Links ] Elshaikh, N.A.; Zhipeng, L.; Dongli, S. & Timm, L.C. (2017) - Increasing
the okra salt threshold value with biochar amendments. Journal of Plant Interactions,
vol. 13, n. 1, p. 51–63. https://doi.org/10.1080/17429145.2017.1418914 [ Links ] EMBRAPA (2011) - Manual e métodos de análise
de solo. Rio de Janeiro: Empresa Brasileira de Pesquisa Agropecuária, Embrapa
Solos. [ Links ] FAO (2006) - Water in agriculture:
opportunity untapped. Rome: Food and Agriculture Organization of the United
Nations. [ Links ] Ferreira, D.F. (2011)
- Sisvar: a computer statistical analysis system. Ciência e Agrotecnologia, vol.
35, n. 6, p. 1039-1042. http://dx.doi.org/10.1590/S1413-70542011000600001 [ Links ] Garg, B.B. & Garg, P.P. (1980) - Salinity
and plant nutrition. Effect of sodium carbonate and sodium bicarbonate on the growth
and absorption of essential macronutrients and sodium in peas (Pisum sativum).
Proceedings of the Indian National Science Academy, vol. 46, p. 694-698. [ Links ] Hoekstra, N.J.; Bosker, T. &
Lantinga, E.A. (2002) - Effects of cattle dung from farms with different feeding
strategies on germination and initial root growth of cress (Lepidium sativum
L.). Agriculture, Ecosystems & Environment, vol. 93, n. 1-3, p.189–196.
https://doi.org/10.1016/S0167-8809(01)00348. [ Links ] Kookana, R.S.; Sarmah, A.K.; Van Zwieten, L.; Krull, E. &
Singh, B. (2011) - Biochar application to soil: agronomic and environmental benefits
and unintended consequences. Advances in Agronomy, vol. 112, p. 103-143.
https://doi.org/10.1016/B978-0-12-385538-1.00003. [ Links ] Laird, D.A.; Brown, R.C.; Amonette, J.E. & Lehmann, J. (2009)
- Review of the pyrolysis platform for coproducing bio-oil and biochar. Biofuels,
Bioproducts and Biorefining, vol. 3, n. 5, p. 547–562.
https://doi.org/10.1002/bbb.16. [ Links ] Lashari, M.S.; Liu, Y.; Li, L.;
Pan, W.; Fu, J.; Pan, G.; Zheng, J.; Zheng, J.; Zhang, X. & Yu, X. (2013) -
Effects of amendment of biochar-manure compost in conjunction with pyroligneous
solution on soil quality and wheat yield of a salt-stressed cropland from Central
China Great Plain. Field Crops Research, vol. 144, p. 113–118.
https://doi.org/10.1016/j.fcr.2012.11.015 [ Links ] Lehmann, J. & Rondon, M. (2006) - Bio-char
soil management in highly weathered soils in the humid tropics. In: Uphoff, N.;
Ball, A.S.; Fernandes, E.; Herren, H.; Husson, O.; Laing, M.; Palm, C.; Pretty,
J.; Sanchez, P.; Sanginga, N. and Thies, J. (Eds.) - Biological approaches to
sustainable soil systems. p. 517-530. CRC Press Taylor & Francis Group,
Boca Raton. [ Links ] Major, J.; Rondon, M.;
Molina, D.; Riha, S.J. & Lehmann, J. (2010) - Maize yield and nutrition during
4 years after biochar application to a Colombian savanna oxisol. Plant and Soil,
vol. 333, n. 1-2, p. 117–128. https://doi.org/10.1007/s11104-010-0327-0 [ Links ] Mengel, K. & Kirkby, E.A. (1987) - Principles of Plant Nutrition.
4th Edition, Bern, International Potash Institute, 745p. [ Links ] Novak, J.M.; Busscher, W.J.; Watts, D.W.; Laird, D.A.; Ahmedna,
M.A. & Niandou, M.A.S. (2010) - Short-term CO2 mineralization after
additions of biochar and switchgrass to a Typic Kaniudult. Geoderma, vol.154,
n. 3-4, p. 281–288. https://doi.org/10.1016/j.geoderma.2009.10.014 [ Links ] Paliwal, K.W.; Maliwal, G.L. & Navawas, C.
(1978) - Effect of bicarbonate-rich irrigation waters on the yield and nutrient
uptake in cotton (Gossypium sp.) and linseed (Linum usitatissimum).
Annals of Arid Zones, vol. 17, p .164-174. [ Links ] Richard, L.A. (1954) - Diagnosis and improvement of saline and
alkali soils. Washington D.C: Agricultural Handbook, 60, U.S.Salinity Laboratory. [ Links ] Rombolà, A.G.; Marisi, G.; Torri, C.; Fabbri,
D.; Buscaroli, A.; Ghidotti, M. & Hornung, A. (2015) - Relationships between
chemical characteristics and phytotoxicity of biochar from poultry litter pyrolysis.
Journal of Agricultural and Food Chemistry, vol. 63, n. 30, p. 6660-6667.
https://doi.org/10.1021/acs.jafc.5b0154. [ Links ] Santos, J.P.; Azevedo, S.G. & Mistretta, G. (1984) - Novos aspectos
da salinização das águas subterrâneas do cristalino do Rio Grande do Norte.
São Paulo, SP: IPT. 27p. (Comunicação técnica, 314). [ Links ] Sappor, D.K.; Osei, B.A. & Ahmed, M.R. (2017) - Reclaiming
sodium affected soil: The potential of organic amendments. International Journal
of Plant & Soil Science, vol. 16, n. 2, p. 1-11.
https://doi.org/10.9734/IJPSS/2017/33410 [ Links ] Silva, I. C. B.; Basílio, J.J.N.; Fernandes, L.A.;
Colen, F.; Sampaio, R.A. & Frazão, L.A. (2017) - Biochar from different residues
on soil properties and common bean production. Scientia Agricola, vol. 74,
n. 5, p. 378-382. http://dx.doi.org/10.1590/1678-992x-2016-0242 [ Links ] Singh, A.P. & Singh, A.R. (2013) - Seasonal changes
in the physic-chemical attributes of salt affected habitat. India Journal of
Science Research, vol. 4, n. 1, p.105-115. [ Links ] Song, W. & Guo, M. (2012) - Quality variations of poultry litter biochar
generated at different pyrolysis temperatures. Journal of Analytical and Applied
Pyrolysis, vol. 94, p. 138–145. https://doi.org/10.1016/j.jaap.2011.11.018 [ Links ] Subhan, D.; Uzma, Y.; Saira, N.; Noureen, A.;
Muhammad, E. & Iqbal, M.T. (2015) - Biochar consequences on cations and anions
of sandy soil. Journal of Biodiversity and Environmental Sciences, vol. 6,
p. 121-131. [ Links ] Verheijen, F.; Jeffery,
S.; Bastos, A.C.; Velde, M.V.D. & Diafas, I. (2010) - Biochar application
to soils: a critical scientific review of effects on soil properties, processes
and functions. Luxembourg, European Commission, 149 p. [ Links ] Walker, D.J. & Bernal, P.M. (2008) - The effects of
olive mill waste compost and poultry manure on the availability and plant uptake
of nutrients in a highly saline soil. Bioresource Technology, vol. 99, n.
2, p. 396–403. https://doi.org/10.1016/j.biortech.2006.12.006 [ Links ] Wu, Y.; Xu, G. & Shao, H.B. (2014) - Furfural and its biochar
improve the general properties of a saline soil. Solid Earth, vol. 5, p.
665–671. https://doi.org/10.5194/se-5-665-2014 [ Links ] Yue, Y.; Guo, W.N.; Lin, Q.M.; Li, G.T. & Zhao, X.R. (2016) - Improving
salt leaching in a simulated saline soil column by three biochars derived from rice
straw (Oryza sativa L.), sunflower straw (Helianthus annuus), and
cow manure. Journal of Soil and Water Conservation, vol. 71, n. 6, p. 467-475.
https://doi.org/10.2489/jswc.71.6.467 [ Links ] Zhang, Y.; Idowu, O.J. & Brewer, C.E. (2016) - Using Agricultural Residue
Biochar to Improve Soil Quality of Desert Soils. Agriculture, vol. 6, n.
1, art. 10.
https://doi.org/10.3390/agriculture6010010 [ Links ] Received/recebido: 2018.08.14 Received in revised form/recebido em versão revista: 2018.10.15 Accepted/aceite: 2018.10.15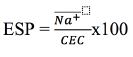 (2)
(2)