Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Revista de Ciências Agrárias
Print version ISSN 0871-018X
Rev. de Ciências Agrárias vol.35 no.1 Lisboa June 2012
Screening of wheat Landraces from Madeira Island for iron tolerance
Avaliação de cultivares tradicionais de trigo da Ilha da Madeira quanto à tolerância ao excesso de Ferro
Ana Maria Domingues1* and Miguel Ângelo Pinheiro de Carvalho2
1*Instituto de Investigação Científica Tropical/ Tropical Research Institute - Apartado 3014 1301-901 Lisboa Codex, Portugal, dam.portugal@hotmail.com
2 Isoplexis Germplasm Bank - Campus da Penteada - University of Madeira 9000-390 Funchal, Portugal
ABSTRACT
Since the fifteen century, the rainfed-cultivation of wheat for grain is traditionally performed on the Island of Madeira. Under several microclimatic conditions and along very sloppy mountains, the landraces are grown on isolated terraces of Andosols with high amounts of iron. Iron oxides are the main inorganic binding agent contributing to the stability of aggregates and to soil fertility in long-term sustainable agriculture in acid and iron-rich soils.
After a two day period of seedling initial growth, a screening test of sixty traditional wheat (Triticum spp.) landraces from the ISOPlexis Genebank at the University of Madeira, Funchal, was performed using nutrient solutions containing 10 or 600 mM Fe, during five days, under controlled laboratory conditions. The elongation of the longest primary root was measured for each genotype and the mean root increment relative to control (as, % relative root increment or RRI; n=28) calculated. This parameter appeared to be a sensitive indicator of Fe tolerance in wheat. Over 85% of wheat germplasm showed the RRI higher than 50%, while the RRI of seven accessions exceeded 70%. This indicates that those landraces are Fe tolerant and might be of particular interest for cultivation under acid rich iron soils of tropical and subtropical areas.
Keywords: Acid soils,Iron tolerance, Triticum.
RESUMO
Desde o século quinze que é tradicionalmente cultivado trigo na Ilha da Madeira. Em sequeiro, sob condições microclimáticas diversificadas, em terraços isolados e dispostos em encostas íngremes, o trigo ocupa solos ácidos, sobretudo da classe dos Andossolos, com elevada concentração de ferro. Nestes solos, os óxidos de ferro constituem o principal agente inorgânico de ligação das partículas do solo, contribuindo para a estabilidade dos agregados.
Foi realizada uma experiência com sessenta cultivares tradicionais de trigo (landraces de Triticum spp.) do Banco de Germoplasma, da Universidade da Madeira, em soluções nutritivas com 10 (controlo) ou 600 mM Fe, durante 5 dias, após 2 dias de crescimento inicial das plântulas. O alongamento da principal raiz primária foi medido para cada planta sob stress e calculada a média do incremento relativo ao controlo (como percentagem de Incremento Radicular Relativo ou IRR; n = 28). O IRR evidenciou ser um indicador sensível da toxicidade do ferro em trigo. Mais de 85% do germoplasma estudado evidenciou IRR superior a 50%, enquanto o IRR correspondente a sete landraces ultrapassou o valor de 70%. Isto foi considerado como indicador da maior tolerância ao excesso de ferro destas landraces de trigo, as quais poderão ter especial interesse na ocupação de solos ricos em compostos inorgânicos de ferro, nas zonas tropicais e subtropicais.
Palavras-chave: Solos ácidos, tolerância ao ferro, Triticum.
Introduction
In order to support the global increasing needs for food, fodder, fibre, and bioenergy there is a growing concern with long-term sustainable agriculture, which is based on low inputs and environment friendly cultivation methods. Soil is the foundation of the majority of the agricultural production and therefore its health affects profitability and sustainability of the agronomic production. The quality of soils is often evaluated by soil aggregation among other characteristics. The major factors controlling macro-aggregates turnover and micro-aggregates formation involve multiple interactions of environmental variables, such as soil micro-organisms, fauna, plant roots, and inorganic binding agents, mainly the iron oxides and the calcium compounds (Six et al., 2004). The lithosphere contains about 5% of iron. In spite of its abundance in arable layers, in well drained soils, most of the element is present in insoluble mineral forms with ferric compounds – with Fe (III) - that are unavailable to plant roots (Eco-SSL, 2003). However, in acid soils, the iron oxides and hydroxides might be the principal component of mineral fraction and crop production might be decreased due to iron toxicity.
Frequently, the economic constrains of farmers and the edaphoclimatic context confine soil neutralization practices or even may require the maintenance of acidic soil conditions. The "adapt-a-plant concept" (Foy, 1993) emphasized the need for the plant genetic approach in solving the limiting soil conditions, particularly the acid soil infertility affecting large areas all over the world. In acidic pedogenesis, aluminous and ferric organic complexes have a protective effect against biodegradation of soil organic matter components (Boudot et al., 1989). The occupation of forest acidic and iron rich soils (with pH ranging from 3.6 to 5.8) by agriculture, combined with the use of local rocks for road veneers critically advised for the exemption of fields liming (Craw, 2005). Because of the acid soil pH neutralization, in the limed fields there was an important reduction of mineralised As and Hg in situ, sub-superficially, principally in the C horizon enriched with iron oxyhydroxides, but there was a very significant raise in the concentration of the soluble forms of these toxic elements in the percolation waters with increased ecological problems down stream (Craw, 2005).
The micronutrient is essential to all living organisms, as iron is necessary for many enzymatic systems and its absence can affect cellular respiration, photosynthesis, carbon and nitrogen metabolism (Chatterjee et al., 2006). But the excess of soluble iron might induce plant stress and affect agronomic yields (Chatterjee et al., 2006). In early studies, Hoffer and Carr (1923) reported the development of seedling-blight and root rots in corn plants grown in acidic soils rich in organic matter, but containing low levels of calcium and phosphorus. The observed brownish-purple discolorations and disintegrations of nodal tissues of stalks were associated with aluminium and iron accumulation in the tissues. The use of limestone alone proved to be harmful, but the application of available phosphates produced stronger and more resistant plants to the described physiological injuries (Hoffer and Carr, 1923).
Often crop plants suffer from iron deficiency or toxicity, because soil soluble iron levels generally change from values near 200 mg kg-1 to 300 g kg-1 or more. Normally, the concentration of iron in plant tissues must vary between 50 to 250 mg kg-1 dry weight (De Varennes, 2003). Monocotyledons show higher Fe2+ tolerance than dicotyledons, as measured by the relative growth rates (RGR), under iron excess in nutrient solutions. These observations were consistent with the higher iron concentrations on sites where the Fe-tolerant ecotypes occurred (Snowden and Wheeler 1993). The Graminaceous plants (species from the family Poaceae) have a strategy to the efficient uptake of Fe2+ with phytosiderophores (Guerinot and Yi, 1994). However, the soil ferric compounds submitted to low potential redox (ranging between 110 to 80 mV or lower) are reduced to ferrous chemical forms, which are much more soluble, increasing the ferrous ion (Fe2+) concentration in soil solution to phytotoxic levels. To face these problems plants developed special mechanisms to iron import (Guerinot and Yi, 1994) or exclusion (Wu et al., 1998), which are genetically expressed. Wu et al. (1998) greatly contributed to the clarification of the genetic basis of Fe tolerance in rice (Oryza sativa). They examined lines derived from the cross between Azucena (a Fe-tolerant japonica variety) and IR-64 (an indica variety moderately sensitive to Fe), using the leaf bronzing index and the relative decrease in shoot dry weight, after 28 days growing in hydroponics with 4.47 mM Fe (at pH 4.5) to evaluate their response to iron presence. Physiological strategies of rice plants adaptation and the agronomic management practices to cope with Fe excess were reviewed by Becker and Asch (2005). Also, several symptoms and nutritional disorders associated with high Fe2+ concentration in leaf tissues under categorized agro-ecological environments of rice were described (Becker and Asch, 2005).
Transient water-logging conditions in acidic soils prone to root hypoxia and microelements toxicities induced reduction of shoot weight and elevated tissues content of Al, Mn and Fe of 21 days old wheat plant tops (Khabaz-Saberi et al., 2006). Camargo et al. (1995) evaluated Triticum durum inbreeds for Fe toxicitiy in hydroponic culture. The young plants were screened after twelve days in nutrient solutions containing 10, 90, 180 and 360 mM Fe, using as evaluation criteria the longer primary root growth (Camar-go et al., 1995).
A landrace is an autochthonous variety with a high capacity to tolerate biotic and abiotic stress, resulting in a high yield stability and an intermediate yield level under a low input agricultural system (Zeven, 1998). Wheat landraces from Madeira are traditionally rainfed cultivated in small and isolated terraces along steep hills. Five major soil types of volcanic origin compose the agricultural land on the main island of the Archipelago, including Andosols, Vertisols, Cambisols, Phaeozems, and Leptosols. But Andosols comprise 60% of the total land area (Madeira et al., 1994). They have acid or very acid conditions and often are aluminium and iron rich soils. The continuous cultivation of wheat ancient cultivars (Triticum spp) in acid Al- and Fe-rich soils for the last six hundred years, induced the adaptation of plant germplasm to the edaphic conditions present on the Island (Ganança et al., 2007). On the other hand, these Madeiran landraces are physiologically adapted to moderately pluvious winter and warm conditions therefore they do not demand seed vernalization to germinate, neither long photoperiod to flowering.
The aim of the present work was to characterize iron tolerance of wheat landraces from the Archipelago of Madeira. The identification of new sources of iron tolerance among these wheat genotypes can be particularly interesting for plant breeding, once the iron toxicity is very frequent in acid soils in tropical and subtropical regions.
Material and methods
Sixty wheat landraces (Triticum spp.), accessions designated by ISOPs from the Isoplexis Germplasm Bank, University of Madeira, at Funchal, Portugal (Table 1), have been screened for their iron tolerance. Four Portuguese traditional wheat cultivars, from the Unidade de Recursos Genéticos, Ecofisiologia e Melhoramento de Plantas (Instituto Nacional de Recursos Biológicos/ I.N.R.B., Elvas, Portugal), and one Brazilian wheat, the inbreed line IAS-58/4/KAL/BB// 3/ALD"S"/5/BOW "S", were used as standards.
Table 1 - Mean relative root increment (RRI, as percentage of control) of wheat seedlings exposed to 600 mM of soluble iron for five days. Sixty wheat landraces from the Madeira Archipelago (designated by ISOP’s number and cultivar common name), four cultivars from the region of Alentejo (Portugal), and one inbreed line from Brazil are ranked according to the increasing order of RRI values and assigned to statistical homogenous groups (n= 28; P= 0.95).

The wheat landraces were collected from several locations across the Archipelago of Madeira located between latitude 32º 39´ N and 32º 51´ N, and longitude 16º 47´ W and 17º 14´W.
Some preliminary experiments were performed with eight traditional wheat cultivars, relevant phytotoxic levels of Fe in solution (0 to 2000 mM of Fe) and different duration of exposure to toxic Fe, to optimize the criteria for the screening. In one of these experiments, after the initial growth for two days in the complete nutrient solution, the young plants were transferred to treatment solutions during the following eight days (Figure 1 and 2). Wheat seedlings were exposed to 10 (control), 400, 600 and 1000 mM of Fe in nutrient solution. Root length was measured in the beginning and three, five and eight days after the start of the Fe treatment (Figure 1). In this previous trial, fresh biomass produced by the plants was weighted after ten days (Figure 2). The young plants are more prone to metal stress than the old ones; so the earlier plant stages are usually used than later stages to evaluate the phytotoxicities.
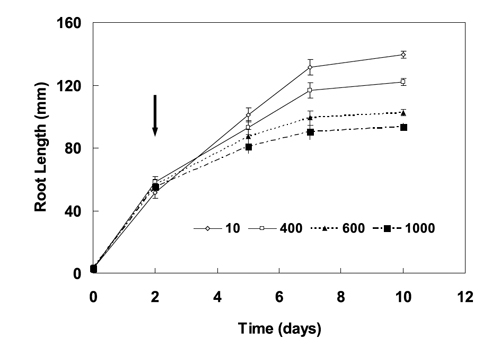
Figure 1 - Mean root growth of six wheat cultivars expressed by root length, in mm. The seedlings were grown in a complete nutrient solution for two days and than transferred to iron excess in the root media. The measurements were made successively on the second, fifth, seventh, and tenth days of a preliminary experiment. The arrow indicates the beginning of treatment with nutrient solutions containing 10 (control), 400, 600 or 1000 mM of soluble iron, and the bars represent mean standard error (total number of plants for each point n=18).
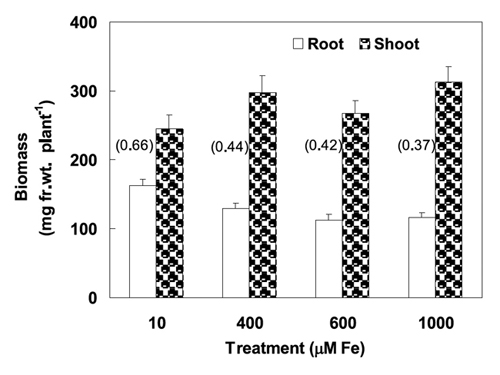
Figure 2 – Mean root and shoot fresh biomass produced by seedlings after ten days of a previous experiment (see legend of Figure 1). Values of root to shoot ratio are presented inside brackets for each iron treatment.
In the present work, all seeds were carefully disinfected for eight minutes by immersion in 5% sodium hypochlorite and rinsed with abundant distilled water. Seeds were germinated for three days at 25º C in trays lined with cotton, imbibed in distilled water and covered with filter paper. The selected uniform seedlings were grown hydroponically on floats in complete nutrient solution, with adjusted pH 4.3 and following composition, expressed as mM (Slaski et al., 1996): 1000 Ca(NO3)2.4 H2O, 300 Mg(NO3)2.6 H2O, 300 NH4NO3, 100 K2HPO3, 100 K2SO4, 400 KNO3, 2 MnCl2.4 H2O, 6 H3BO3, 0.5 ZnSO4.4 H2O, 0.15 CuSO4.5 H2O, 0.1 Na2MoO4.2 H2O, 10 FeCl3.6 H2O, and 10 Na2EDTA. The initial period of seedling growth in complete nutrient solutions (at pH= 4.3) prevented major interferences associated with the inductive mechanisms of root uptake and allowed the minimization of different germination vigour of the seeds of the different wheat genotypes. After two days, the plants were transferred to treatment solutions containing (in mM) 2900 NO3-, 300 NH4+, 1000 Ca2+, 300 Mg2+, 100. 65 SO42- and the micronutrients (34 Cl-, 20.20 Na+, 10 Fe2+, 6 BO33-, 2 Mn2+, 0.50 Zn2+, 0.15 Cu2+, and 0.10 MoO42-). Iron was introduced in treatment solutions by adding the proper volume of a Fe stock containing 200 mM FeCl3.6 H2O and 200 mM Na2EDTA. Potassium was not included in order to avoid amelioration of toxic effects by reducing excess Fe uptake and by alleviating physiological injuries (Cakmak, 2005). Phosphorus was also omitted to prevent precipitation of iron phosphates in treatment solutions. All treatment solutions were continuously aerated and maintained at pH 4.1 ± 0.2. About 33 ml of solution was available per plant and renewal of the total volume of nutrient solutions was done, so that variation of ionic conductivity was less than 10 %. Solutions pH, conductivity (mS), and total dissolved solids (TDS, equivalent to ppm KCl) were measured twice to four times per day with a Cyberscan PC300 potentiometer.
Temperature of air and water bath was controlled at 23 ± 3ºC, while air relative humidity was kept low at 50 to 60 %. The irradiance at leaf level was 150 mM m-2 s-1 and was provided by fluorescent Phillips lamps TL 40w/54 for 16 hours per day.
For the evaluation of iron tolerance, the length of the longest primary root of each plant was measured with accuracy of 0.5 mm at the end of two days in complete nutrient solution (initial value) and at the end of the treatment period (final value). The relative root increment (RRI) was calculated as the ratio between the absolute increment of root length after Fe treatment (n= 28) and the average increment of root length of control plants of each wheat accession (with total number of control plants n= 14), expressed as percentage. The relative elongation rate of the primary root (RER) of seedlings of each accession was also calculated for both modalities, control and excess iron, during the period of treatment, expressed as mm mm-1 day-1.
The screening was performed with two factors, accessions and Fe treatments, and seven blocks. Each block consisted of three randomized vessels with treatment solutions, including one control (10 mM Fe) and two containing 600 mM Fe. Each vessel included two plants of the selected accessions.
Analysis of variance and definition of homogenous statistical groups with least significance difference at P = 95 % (a < 0.05), were performed to compare the wheat accessions using the STATGRAPHICS program.
Results
Sixty traditional wheat landraces have been screened for their tolerance to the excess of iron in root media. The root relative increment (RRI) was found to be statistically significant criterion to measure the iron toxicity by the inhibition of root elongation of wheat seedlings under iron stress (Table 1).
In a previous experiment performed with six wheat accessions, three days treatment with 1000 mM provoked about 50% reduction of mean root elongation of five days old seedlings. And the treatment with 600 mM Fe induced about 50% reduction of mean root elongation of seven (to ten) days old seedlings, during five (to eight) days of treatment (Figure 1). However, the different accessions showed more differential behaviour in the 600 mM Fe relative to 1000 mM Fe. The exposure to 600 mM Fe for five days treatment was identified as optimal conditions to better discriminate Fe resistance among the Madeiran landraces, because after that the rate elongation of plant roots decreased (Figure 1). The duration of the experiment was short and it was easy to perform. For longer periods of treatment with older plants, the nutrient solutions evidenced more quickly changes in pH and conductivity, so they had to be substituted more often. The results for root length, absolute root elongation and root increment relative to the correspondent control (RRI) after five days treatment with 600 mM Fe were statistically different among accessions. To compare distinct accessions exhibiting different germination vigour in the period considered, the percentage of RRI showed to be a parameter more independent than the others.
The root biomass accumulation was affected by Fe stress, but lesser than the root elongation. The shoot biomass accumulation showed inconsistent trends, so, the root to shoot ratio significantly decreased after eight days under Fe stress in comparison with control plants (Figure 2).
Some of the young wheat plants suffered to some extent from H+ toxicity (at pH 4.1 ± 0.2), which possibly affected the root growth. In few plants, the apical two mm of some of the thin and weak roots were white vitreous and lost their positive geotropism, or became dark and necrotic.
The distribution of individual values of RRI from wheat plants (n= 28) of four landraces are shown in Figure 3 to illustrate the genotypic differences in response to Fe excess present in solution, under the experimental conditions.
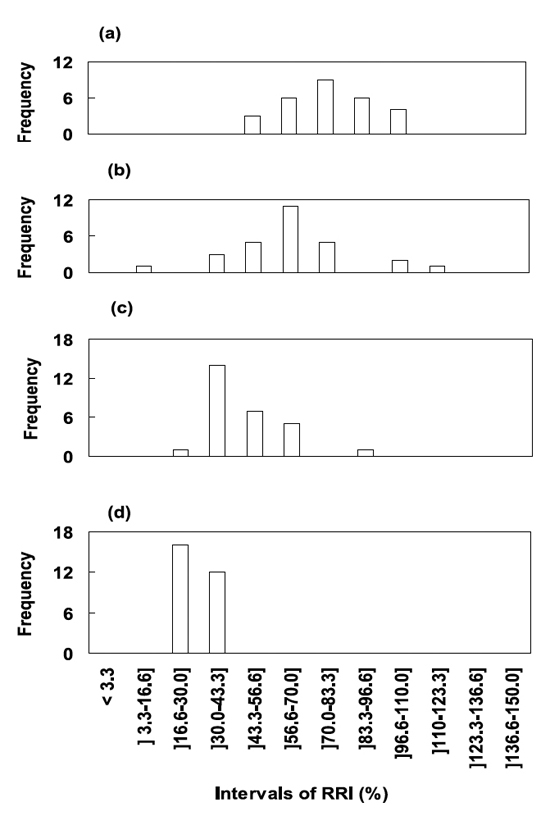
Figure 3 - Histograms of relative root increment (RRI, as percentage of control) of four Madeiran wheat cultivars (a) 0245 Trigo Branco do Sítio do Amparo, (b) 0082 Trigo Vermelho, (c) 0118 Trigo Rapado, and (d) 0101 Trigo da Mata, with the frequency values (total number of plants n= 28 for each landrace) distributed by statistically defined intervals. In each interval of values considered the lower limit is excluded, while the upper limit is included. Plants were grown for five days in nutrient solution containing 600 mM of soluble iron.
The ISOP 0245 appeared to be the most Fe tolerant accession. Four individuals from this landrace showed more than 96.6 % RRI and 33% exceeded 83.3 % RRI (Figure 3a). Sampled individual plants showed normal-like distribution in the variability of their response to Fe stress, which is consistent with the population landrace adaptation to this limiting factor. Inside all wheat landraces there were significant differences among individuals. Particularly pronounced was the variability found in the ISOP 0082 (Figure 3b): three individual plants had values that were higher than 96.6% RRI, twenty-four plants belonged to four continuous classes of RRI between 43.3 and 83.3 % RRI, and only one plant exhibited 12.4 % RRI; but this accession showed globally good tolerance to Fe excess (62.6 % mean RRI in Table 1). The normal-like distribution is disrupted in ISOP 0118 and skewed towards lower RRI values and over half of individuals ranked below 43.3% (Figure 3c). While the ISOP 0101 did exhibit the highest susceptibility to excess of iron in the growing media with all individuals clustered in two classes comprising the RRI values lower than 43.3% (Figure 3d).
Figure 4a shows the distribution of individual values of root absolute elongation (mm) of all landraces (n= 1820) during five days of treatment with 600 mM Fe. These values ranged from 3 to 102 mm, within eleven statistical classes of 10 mm intervals. Only 4.9% of the individuals showed root elongation shorter than 20 mm and about 53% have grown more than 40 mm under Fe stress (Figure 4a). Besides the genotypic tolerance to Fe excess, this parameter depends on the seed germination vigour of the different genotypes and particularly of the duration of experiment. The frequency of individual RRI (%) distributed among eleven discrete classes, expressed as number and as percentage of total (in brackets), is presented in Figure 4b. The RRI criterion reflects the roots response to Fe stress and in comparison with absolute root elongation is less dependent on the rate of root potential growth without stress (and on the experiment duration), and allowed different wheat genotypes comparisons. It revealed that over half of wheat individuals sampled from the accessions of Madeira exceeded 50% RRI and allowed very large range from 3.6% to 141.9% RRI (respectively, from one plant from ISOP 0112 and another one from ISOP 0077). Additionally, ten extremely tolerant individuals (RRI over 110%) and nine sensitive ones (RRI less than 16.6%) have been identified (Figure 4b).
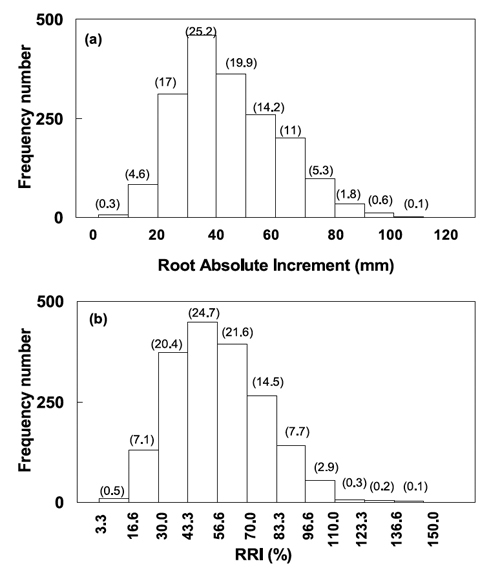
Figure 4 - Distribution of (a) root absolute increment (in mm) and (b) relative root increment (RRI, as percentage of control) of sixty-five wheat genotypes, by statistically defined intervals. Plants (n= 1820) were grown during five days in nutrient solution containing 600 mM of soluble iron. Values of frequency percentage are presented in brackets for each column.
Table 1 presents all wheat samples tested (the Madeiran landraces, the Portuguese cultivars and one Brazilian inbreed line) ranked by to the increasing order of the mean RRI. The majority of accessions exhibited RRI between 50 and 70% RRI suggesting an intermediate tolerance to Fe excess of the wheat germplasm from Madeira.
For instance, with the common designation of Trigo Vermelho (Red Wheat), we identified five ISOPs including 0107, 0183, 0082, 0077, and 0121 were characterized by different RRI of 35.8%, 59.4%, 62.6%, 67.3% and 74.8% respectively. From these landraces, the ISOP 0121 (Table 1) was the most tolerant among them, revealed by the highest mean relative root increment. The ISOP 0082 (Table 1 and Figure 3b) was not significantly different from the ISOPs 0077 and 0183, as all three exhibited intermediate tolerance. But statistical significant difference was found between the last two landraces. The ISOP 107 was the Red Wheat statistically most susceptible to Fe toxicity (Table 1).
Among the seven wheat landraces commonly designated as Trigo Rapado (Table 1), the ISOP 0208 was significantly superior to the ISOPs 0074, 0110, 0116, and ISOP 0118 (Figure 3c; and with 46.2% RRI, in Table 1), while statistically the most Fe-tolerant Trigo Rapado was the ISOP 0089 (75.4% RRI).
The seven accessions with RRI exceeding 70 % might be considered as the most Fe-tolerant, and ISOP 0245 (Trigo Branco do Sítio do Amparo; Figure 3a) was significantly more Fe-tolerant than ISOP 0098 (Trigo Peladinho) as revealed by the RRI indicator.
Discussion
The Fe2+-toxicity syndrome rises together with plant nutritional disorders, which are usual in acid soils (Marschner, 1991), particularly toxicities of Al and Mn, and deficiencies of P, Ca, Mg, Zn, and SiO2. So, the symptoms alone are difficult to identify in many plant species. In field conditions, the soybean plantlets developed precociously an overall impaired physiology in very wet, acid and Fe2+-toxic soils, but the visible shoot symptoms of toxicity disappeared completely when the soils were well aerated (Bataglia and Mascarenhas, 1981). In the fields, the expression of Fe2+-toxicity symptoms required the uptake by roots and its acropetal translocation via xylem flow into the leaves (Becker and Asch, 2005). Frequently, these include accumulation of iron in necrotic spots on fully developed leaves and leaf bronzing of rice (De Varennes, 2003; Eco-SSL, 2003).
Many authors screen metal tolerance by the root response to metal excess in hydroponics (Camargo et al., 1995). In this work, the selection of iron concentration to root media was supported by the observation of morphological features in the preliminary experiments, besides the preliminary results stated. The iron stress led to diminished uptake of water and nutrients (data not shown). After ten days of growth in control medium (with 10 mM Fe added), the healthy and vigorous plants had bright green leaves and the roots were long, firm, white coloured and exhibited abundant secondary ramification. The vigorous plants grown with 400 mM Fe in root media had slightly chlorotic leaves and shorter primary and secondary roots. The 600 mM Fe treatment was more debilitating to wheat plants, which showed speckled yellowish green leaves with rare necrotic tips and, generally, white to orange coloured, shorter and feeble roots, that occasionally get broken easily. Under 1000 and, especially 2000 mM Fe treatments, the leaves had intense intervein chlorosis, resembling light greenish yellow marbling, numerous plants showed apical necrosis and some exhibited dehydration symptoms. The roots were remarkably shorter and had orange coloured apices. According, other authors (Eco-SSL, 2003) also referred that the root apices showed intense orange coloured surfaces, possibly due to Fe2+ oxidation to Fe3+ promoted by the rice roots.
All accessions showed lower RER (as mm mm-1 day-1) in the Fe excess treatment compared to the control, but the genotype discrimination by this criteria was poor (data not shown). During the five days treatment with 10 and 600 mM Fe the mean values of RER of all tested accessions were 0.342 (ranging from 0.079 to 1.340) and 0.188 (0.020-0.673), respectively. Drastic reduction of RER was observed in response to Fe treatment in the most Fe-susceptible ISOP 0101 (0.235 to 0.084) while only slight decrease in RER (0.219 to 0.174) was recorded in the most Fe-tolerant ISOP 0245. However, in a two weeks experiment with wetland plant species, Snowden and Wheeler (1993) found negative correlations between the tolerance to iron stress (expressed as RGR, as g g-1 day-1) and few other variables as: the relative growth rates in control nutrient solutions, the soil fertility and the extractable phosphorus concentrations of original sites of plant collection. In that short experiment, the root fresh biomass of young wheat plants from control was statistically the highest compared with that from all other treatments undistinguished among accessions, which indicated that the Fe toxicity affected mainly the roots. The shoot biomass from control plants tended to be lower than that from the plants submitted to excess Fe in root media (Snowden and Wheeler, 1993). Iron is a required nutrient for plant normal growth and the ferric iron supplied to nutrient solutions could be efficiently uptaken by roots using ferroxidases or phytosiderophores (Guerinot and Yi, 1994), and than transported to shoots and transformed into bioactive molecules or become stored, within a well regulated iron metabolism of Graminaceous plants. In accordance with these authors, apparently Fe accumulation have occurred at the root surface and a putative surplus of Fe uptake could have induced locally oxidative stress, which affected the root growth. And, possibly the allocation of seed reserves and of de novo biosynthesized molecules priority into shoots might be involved in the inconsistent trend of increased shoot growth of wheat plants submitted to Fe excess treatments for a few days (Figure 2).
Curiously, among the most Fe-susceptible wheat genotypes (with RRI lower than 43.3%) we found the ISOP 107 and the Brazilian inbreed IAS-, which were found very Al-tolerant (Domingues et al., 2006). Under acid soil conditions containing high amounts of iron and aluminium soluble compounds, these genotypes would suffer from Fe2+ toxicity, during the rainy season, but this agronomic constrain could be overcome with good soil drainage. Particularly in iron-rich soils and rainfed conditions of extensive crops, probably, plants of Fe-susceptible genotypes would have shallow roots, impaired water and nutrient uptake, and yield reduction.
The RRI biotest is a non-destructive, macro-visual method that might be used with large number of accessions. As for aluminium screening (Domingues et al., 2006), it provides fast, reproducible, and inexpensive results, and consents the best discrimination among precise mean values of many genotypes, since the controlled experimental conditions were maintained. However, the agronomic value of potentially interesting material needs be confirmed under field conditions containing toxic levels of the metal.
Wheat germplasm from Madeira seems to be well adapted to the prevalent high levels of aluminium and/ or iron present in the acid and very acid soils (Ganança et al., 2007). The results obtained in this work indicate that at least the most tolerant seven accessions could be used as a source of Fe tolerance in wheat breeding programs to tropical and subtropical regions with Fe-toxic soils. ISOP 098 (Trigo Peladinho) collected in a field with the soil pH 4.3 was found to be Al-tolerant in a previous experiment (Domingues et al., 2006). Also, Camargo and collaborators (1995) found tolerance to different metals (Al, Mn, and Fe), alone or combined, in Triticum species. The ISOP 098 could be included in the plant breeding of new wheat cultivars for regions suffering from phytotoxic levels of Al and Fe in acid soils. The most promising plants of wheat accessions after submitted to Fe excess screening could be transplanted and grown to maturity for grain. The descendents originated by the self-pollinated Fe-tolerant individuals would inherit the particular character of maintaining roots increment under Fe toxic conditions. Finally, further cereal breeding programs must be implemented by the crosses of this potentially Al- and Fe-tolerant germplasm with other most desirable inbreeds, exactly in the proper agricultural environment, during several years, to select new high yield cultivars improved for the locally prevalent abiotic and biological constraints (Foy, 1993).
Conclusions
The assessment of the mean relative root increment (RRI) of wheat seedlings exposed to 600 mM Fe for five days allowed a good evaluation of the degree of Fe tolerance and good discrimination among landraces from the Archipelago of Madeira. Fifty-three percent of all individuals tested exceeded 40 mm of root elongation under experimental conditions, but remarkable heterogeneity for Fe tolerance was found inside some wheat landraces of Madeira.
The seven ISOPs 0078, 0089, 0098, 0109, 0121, 0244, and 0245 were the most Fe-tolerant and were characterized by more than 70 % of mean RRI (Table 1). They correspond to about 12 % of the landraces studied, while 85 % of all ISOPs showed more than 50 % of mean RR, which indicates there was found good tolerance to Fe excess in root media in the majority of wheat germplasm collected on the Island. The ISOP 098 should be considered for development of new cultivars for regions suffering from phytotoxic levels of Al and Fe in acid soils.
This work constitutes a first evaluation of iron tolerance from wheat landraces in Portugal, and it should proceed by confirming the superior performance of the Madeiran wheat landraces in field trials with iron enriched soils. Also, research is needed to identify the physiological and genetic basis of this trait in wheat. This screening indicates that some landraces are Fe tolerant and might be of particular interest for cultivation under acid rich iron soils of tropical and subtropical regions.
Acknowledgements
Thanks to Dr. Maria Antonieta Nunes, who has in the meantime retired of I.I.C.T.(Tropical Research Institute, Lisbon, Portugal); to Dr. Jan Slaski, from Alberta Research Council (Canada); and to Dr. Benvindo Maçãs, from I.N.R.B. (National Institute for Biological Resources, Elvas, Portugal). This work has been supported by Program SAPIENS, from Fundação para a Ciência e Tecnologia (P.O.C.T.I. nº 35003/ AGR/2000).
References
Bataglia, O.C. and Mascarenhas, H.A.A. (1981) - Toxicidade de ferro em soja. Bragantia, 40: 199-203. [ Links ]
Becker, M. and Asch, F. (2005) - Iron toxicity in rice – conditions and management concepts. Journal of Plant Nutrition and Soil Science, 168: 558-573. [ Links ]
Boudot, J.-P., Brahim, A.B.H.; Steiman, R. and Seigle-Murandi, F. (1989) - Biodegradation of synthetic organo-metallic complexes of iron and aluminium with selected metal to carbon ratios. Soil Biology and Biochemistry, 21: 961-966. [ Links ]
Cakmak, I. (2005) - The role of potassium in alleviating detrimental effects of abiotic stresses in plants. Journal of Plant Nutrition and Soil Science, 168: 521-530. [ Links ]
Camargo, C.E.O.; Felício, J.C.; Freitas, J.G. and Ferreira Filho, A.W.P. (1995) - Trigo duro: tolerância à toxicidade de alumínio, manganês e ferro em soluções nutritivas. Bragantia, 54: 371-383. [ Links ]
Chatterjee, C.; Gopal, R. and Dube, B.K. (2006) - Impact of iron stress on biomass, yield, metabolism and quality of potato (Solanum tuberosum L.). Scientia Horticulturae, 108: 1-6. [ Links ]
Craw, D. (2005) - Potential anthropogenic mobilisation of mercury and arsenic from soils on mineralised rocks, Northland, New Zealand. Journal of Environmental Management, 74: 283-292. [ Links ]
De Varennes, A. (2003) - Produtividade dos solos e ambiente. Escolar Editora, Portugal. [ Links ]
Domingues, A.M.; Ganança, J.F.T.; dos Santos, T.M.M.; Pinheiro de Carvalho, M.Â.A. and Nunes, M.A. (2006) - Prospects for aluminium tolerance in traditional wheat cultivars from Madeira Archipelago. Revista de Ciências Agrárias, 29: 69-79. [ Links ]
Eco-SSL , (2003) - Ecological soil screening level for iron. Interim final. OSWER Directive 9285.7-69 (on line). US EPA, 44 pp (Acessed on 2011.05.13). Available at <http://www.epa.gov/ecotox/ecossl/pdf/eco-ssl_iron.pdf> [ Links ].
Foy, C.D. (1993) - Role of the soil scientist in genetic improvement of plants for problem soils. In: Proceedings of the Workshop on Adaptation of Plants to Soil Stresses. Lincoln, University of Nebraska, p. 185-205. [ Links ]
Ganança, J.F.T.; Abreu, I.; Sousa, N.F.; Paz, R.F.; Caldeira, P.; dos Santos, T.M.M.; Costa, G.; Slaski, J.J. and Pinheiro de Carvalho, M.Â.A. (2007) - Soil conditions and evolution of aluminium resistance among cultivated and wild plant species on
the Island of Madeira. Plant Soil and Environment, 53: 239-246.
Guerinot, M.L. and Yi Y. (1994) - Iron: nutritious, noxious and not readily available. Plant Physiology, 104: 815-820. [ Links ]
Hoffer, G.N. and Carr, R.H. (1923) - Accumulation of aluminium and iron compounds in corn plants and its probable relation to rootrots. Journal of Agricultual Research, 23: 801-860. [ Links ]
Khabaz-Saberi, H.; Setter, T.L. and Waters, I. (2006) - Waterlogging induces high to toxic concentrations of iron, aluminium, and manganese in wheat varieties on acidic soil. Journal of Plant Nutrition, 29: 899-911. [ Links ]
Madeira, M.; Furtado, A.; Jeanroy, E. and Herbillon, A.J. (1994) - Andisols of Madeira Island (Portugal). Characteristics and classification. Geoderma, 62: 363-383. [ Links ]
Marschner, H. (1991) – Mechanisms of adaptation of plants to acid soils. Plant and Soil 134: 1-20. [ Links ]
Six, J.; Bossuyt, H.; Degryze, S. and Denef, K. (2004) - A history of research on the link between (micro) aggregates, soil biota and soil organic matter dynamics. Soil and Tillage Research, 79: 7-31. [ Links ]
Slaski, J.J.; Zhang, G.; Basu, U.; Stephens, J.L. and Taylor, G.J. (1996) - Aluminum resistance in wheat (Triticum aestivum) is associated with rapid, Al-induced changes in activities of glucose-6-phosphate dehydrogenase and 6-phosphogluconate
dehydrogenase in root apices. Physiologia Plantarum, 98: 477-484.
Snowden, R.E.D. and Wheeler, B.D. (1993) - Iron toxicity to fen plant species. Journal of Ecology, 81: 35-46. [ Links ]
Wu, P.; Hu, B.; Liao, C.Y.; Zhu, J.M.; Wu, Y.R.; Sanadhira, D. and Paterson, A.H. (1998) - Characterization of tissue tolerance to iron by molecular markers in different lines of rice. Plant and Soil, 203: 217-226. [ Links ]
Zeven, A.C. (1998) - Landraces: a review of definitions and classifications. Euphytica. [ Links ]
Recepção/Reception: 2011.03.21
Aceitação/Acception: 2011.09.26














