Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Silva Lusitana
versão impressa ISSN 0870-6352
Silva Lus. vol.22 no.1 Lisboa jun. 2014
ARTIGOS
Presence of Glycaspis brimblecombei and its Parasitoid Psyllaephagus bliteus in Tunisia and Portugal
Presença do Glycaspis brimblecombeie e do parasitoide Psyllaephagus bliteus na Tunísia e em Portugal
Présence de Glycaspis brimblecombei et son parasitoïde Psyllaephagus bliteus en Tunisie et au Portugal
*S. Dhahri, **M.L. Ben Jamaa, ***A. Garcia, ****C. Boavida and *****M. Branco
*Master E-mail: dhahrisamir@yahoo.fr
**PhD, Laboratoire de Gestion et de Valorisation des Ressources Forestieres, INRGREF. 2080 Ariana, TUNISIE. E-mail: benjamaami@gmail.com
***Master E-mail: andregarcia@isa.ulisboa.pt
*****PhD, Universidade de Lisboa, Instituto Superior de Agronomia, CEF. Tapada da Ajuda, 1349-017 Lisboa, PORTUGAL. E-mail: conceicao.boavida@iniav.pt
****PhD, Instituto Nacional de Investigação Agrária e Veterinária, IP (INIAV). Tapada da Ajuda, Edificio 1, 1349-018 Lisboa, PORTUGAL. E-mail: mrbranco@isa.ulisboa.pt
ABSTRACT
We report here for the first time the presence of Glycaspis brimblecombei Moore (Hemiptera: Psyllidae) and its parasitoid Psyllaephagus bliteus Riek (Hymenoptera: Encyrtidae) in Tunisia, as well as the first record of the presence of P. bliteus in Portugal. Data regarding their geographical distribution, host plants and parasitism rates in Tunisia and Portugal, were collected. G. brimblecombei was observed for the first time in Tunisia in 2010 in the North coastal regions where it was found only on Eucalyptus camaldulensis Dehnh. Main host species in Portugal were E. camaldulensis, E. tereticornis Sm., E. rudis Endl. and E. ovata Labill.. Nine other species, including E. globulus Labill. were found to be resistant. In Tunisia, infestation levels were higher on the West regions (80.5%±4.0) compared to those on the East (8.8% ±4.4). Overall, parasitism rates were found to be low, on average 6.5%±0.8. In Portugal infestation levels varied from 5 to 75% but no geographical pattern emerged. Parasitism rates varied seasonally, from 1% in May to 35% in October, corresponding to the period of build-up and collapse of G. brimblecombei population. A density-dependence pattern is suggested by three weeks delay on the parasitized nymphs in relation to the total nymphs of G. brimblecombei. Further work will be needed to evaluate the efficacy of the parasitoid in controlling this psyllid.
Key words:Eucalyptus, biological control, biological invasions, Red Gum Lerp, Psyllid
RESUMO
Descreve-se aqui pela primeira vez a presença de Glycaspis brimblecombei Moore (Hemiptera: Psyllidae) e do seu parasitóide Psyllaephagus bliteus Riek (Hymenoptera: Encyrtidae) na Tunísia, assim como a presença de P. bliteus em Portugal. Foram recolhidos dados relativos à sua distribuição geográfica, plantas hospedeiras e taxas de parasitismo. G. brimblecombei foi observado pela primeira vez na Tunísia em 2010 nas regiões costeiras do norte, tendo sido observado unicamente em Eucalyptus camaldulensis Dehnh.. As principais plantas hospedeiras em Portugal são E. camaldulensis, E. tereticornis Sm., E. rudis Endl. e E. ovata Labill. Nove outras espécies amostradas, incluindo E. globulus Labill., mostraram-se resistentes. Na Tunísia, as taxas de infestação foram superiores na região ocidental (80,5%±4,0) por comparação com a região oriental (8,8% ±4,4). Em geral, as taxas de parasitismo encontradas foram fracas, em média 6,5%±0,8. Em Portugal, as taxas de infestação variaram de 5 a 75%, mas sem uma tendência geográfica notória. As taxas de parasitismo variaram em função da estação, de 1% em Maio até 35% em Outubro, correspondendo ao período de desenvolvimento e colapso da população de G. brimblecombei. Um modelo de resposta dependente da densidade é sugerido pelo atraso de três semanas entre o pico de parasitismo das ninfas em relação ao número total de ninfas de G. brimblecombei. Serão necessários trabalhos adicionais para avaliar a eficácia deste parasitóide no controlo da psila do eucalipto.
Palavras-chave: Eucalyptus, luta biológica, invasões biológicas, Psila do Eucalipto, Psyllidae
RÉSUMÉ
Nous signalons la présence, pour la première fois, de Glycaspis brimblecombei Moore (Hemiptera: Psyllidae) et son parasitoïde Psyllaephagus bliteus Riek (Hymenoptera: Encyrtidae) en Tunisie et, également, la présence pour la première fois de P. bliteus au Portugal. Des données relatives à leur distribution géographique, leurs plantes hôtes et les taux de parasitisme son collectées. G. brimblecombei a été observé pour la première fois en Tunisie en 2010 dans les régions côtières du Nord où il a été noté, uniquement, sur Eucalyptus camaldulensis Dehnh.. Ses principales espèces hôtes au Portugal sont E. camaldulensis, E. tereticornis Sm., E. rudis Endl. et E. ovata Labill.. Neuf autres espèces, incluant E. globulus Labill., semblent être résistantes. En Tunisie, les taux d'infestation sont élevés dans les régions de l'Ouest (80,5%±4,0) comparés à ceux des régions de l'Est (8,8% ±4,4). En général, les taux de parasitisme trouvés sont faibles, avec une moyenne de 6,5%±0,8. Au Portugal, les niveaux d'infestation varient de 5 à 75% mais aucune tendance géographique n'émerge. Les taux de parasitisme varient en fonction de la saison, de 1% en Mai à 35% en Octobre, correspondant à la période de formation et celle de l'effondrement de la population de G. brimblecombei. Un modèle de densité-dépendance, d'une durée de trois semaines, est suggéré pour le parasitisme des nymphes en relation avec le nombre total des nymphes de G. brimblecombei. D'autres travaux seront nécessaires pour évaluer l'efficacité du parasitoïde dans le contrôle de ce psylle.
Mots-clés: Eucalyptus, lutte biologique, invasions biologiques, Psylle du Gommier Rouge, Psyllidae
Introduction
Originating from Australia, Eucalyptus are one of the most used trees worldwide for forest plantations, mainly for wood, pulp, biomass, ornamental foliage, soil conservation and honey production. In Tunisia, Eucalyptus plantations were introduced in the thirties, with 117 Eucalyptus species planted in different climatic areas (KHOUJA et al., 2001). Currently, Eucalyptus plantations in Tunisia comprise 41000 ha (DGF, 2010), mainly in the North of Tunisia with two main species, E. camaldulensis Dehnh. and E. gomphocephala DC. (DGF, 2005). In Portugal, E. globulus Labill. is the main species used in forest plantations, occupying about 26% of the country forest area (ICNF, 2013). Other species, mainly E. camaldulensis are used mostly for amenity purposes in urban parks and road sides.
In both regions the number of alien insect pests in Eucalyptus trees has increased steeply in the last 50 years (KENIS and BRANCO, 2010; DHAHRI and BEN JAMÂA, 2008). Understanding possible pathways, susceptibility of Eucalyptus species and management strategies to deal with these new problems has become a major concern.
The red gum lerp psyllid Glycaspis brimblecombei Moore was one of the most recent introductions in both countries. This psyllid is distinguished by two cephalic projections between the eyes of the adult (LAUDONIA and GARONNA, 2010) and the protective conical white covers made by the nymphs on the leaves (Figures 1a and 1c). Females deposit the eggs on the leaf surface, attached to the leaf (Figure 1b). The nymphs feed, grow and develop under a conical white waxy cover. The last moult occurs outside the cover, originating the adult (Figure 1c). Both nymphs and adults are phloem feeders. Heavy attacks result on the production of copious amounts of honeydew and premature leaf shedding, leading to loss of tree vigour and growth (BRENNAN et al., 1999).

Glycaspis brimblecombei was detected outside Australia for the first time in North America, in California in 1998, where it was considered the most damaging psyllid introduced in the country (BRENNAN et al., 1999; 2001). In 2001, it was detected for the first time in Chile in the vicinity of the International Airport of Santiago (SANDOVAL and ROTHMANN, 2002). It was included in the Alert List of the European and Mediterranean Plant Protection Organization (EPPO) between 2002 and 2006, when it was deleted. In 2003 was first detected in Brazil (SANTANA and BURCKHARDT, 2007) and in 2005 in Argentina (BOUVET et al., 2005). In Europe, it occurred for the first time in the Iberian Peninsula in 2007 (BORRAJO et al., 2009; VALENTE and HODKINSON, 2009). In Morocco it was first found in 2009 (MAATOUF and LUMARET, 2012) and in Italy in 2010 (LAUDONIA and GARONNA, 2010; PERIS-FELIPO et al., 2011; LO VERDE et al., 2011). This historical sequence of events suggests a first introduction outside Australia in North America or Chile, a subsequent spread to other countries in South America, Brazil and Argentina, where Eucalyptus plantations represent a significant part of the forest plantations, and then to Europe and North Africa.
In the present work we analyze the geographical distribution of the red gum lerp psyllid in the two countries and the occurrence and parasitism rates of its exotic natural enemy Psyllaephagus bliteus Riek (Hymenoptera: Encyrtidae) (Figure 2).InPortugal we further analyze differences between Eucalyptus species susceptibility.

Material and methods
Field surveys
Portugal
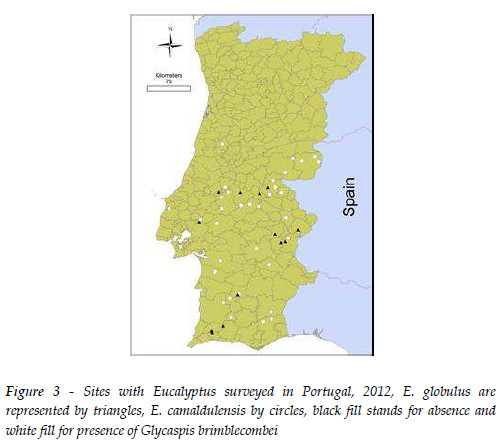
In Portugal, the geographical distribution and infestation levels of G. brimblecombei, were evaluated in 37 sites with E. camaldulensis, plus 16 sites with E. globulus, located along coastal and interior zones of mainland Portugal, in June 2012 (Figure 3). As the Eucalyptus species most susceptible to G. brimblecombei are E. camaldulensis and E. tereticornis Sm., found in Portugal along road sides and in city parks, these were the sites and tree species chosen for the surveys. At each site, four trees were evaluated for the presence or absence of G. brimblecombei. When the insect was found the infestation level was evaluated by sampling on each tree, at observer height (1.5-2m), four branches, one in each cardinal direction: North, South, East and West. The infestation level on each branch was estimated visually on four categories (0; 1-20%; 20-50%; >50%) and then mid-classes infestation levels were assigned as follow: 0; 10%, 35% and 75%.
Tunisia
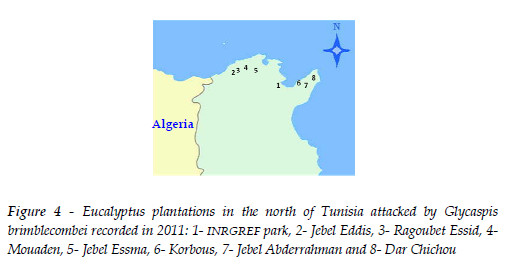
In Tunisia samplings were conducted in eight eucalyptus forest located from 10 to 228 m above sea level, near the Mediterranean Sea (Figure 4): INRGREF Park, Jbel Eddis and Dar Chichou on E. camaldulensis and E. gomphocephala DC. trees, Moauden on E. camaldulensis and E. astringens (Maiden) trees, Ragoubet Essid and Jebel Essma on E. camaldulensis, Korbous on E. camaldulensis and E. rudis Endl. trees and Jebel Abderrahman on E. camaldulensis and E. tereticornis. In August 2012, ten trees of each Eucalyptus species present in each site were evaluated except for the INRGREF Park where only three trees of each species were evaluated. To estimate the presence and the intensity of the attack, from each tree four attacked branches of 70 cm in length were cut, one of each cardinal direction, and brought to the laboratory where the number of total and attacked leaves were counted.
Host range
In Portugal an additional survey was conducted in August 2012 to assess susceptibility of different Eucalyptus species planted in a Eucalyptus arboretum located in the campus of the Faculty of Agronomy (ISA), Lisbon. The infestation level of G. brimblecombei was assessed in 30 Eucalyptus species that are present at ISA's arboretum. For each Eucalyptus species four trees were selected, at 10 m distance along a line, whenever possible. In each tree four branches were selected, from each cardinal direction and the infestation level was determined as described above, for the field surveys in Portugal.
Parasitism rates
Portugal
The seasonal evolution of the populations of G. brimblecombei and its parasitoid P. bliteus was followed by monthly sampling on E. camaldulensis trees in the ISA's arboretum. This study was conducted between May and October 2012.
On each month 40 leaves per tree were collected, at about 1.5-3m height, from five trees. The leaves were placed into separate paper bags by tree. In the same day, the leaves were inspected in the laboratory and the immature stages of G. brimblecombei were identified and counted separately by the following groups of developmental stages: i) eggs; ii) young nymphs: first, second and third instars; ii) fourth instar nymphs (N4); iii) fifth instar nymphs (N5); iv) parasitized alive nymphs, "live mummies" and v) parasitized nymphs with exit hole from where a parasitoid had already emerged "empty mummies". The nymphal stages were determined by the number of antennal segments, according to FIRMINO-WINCKLER (2009). The "live mummies" were placed separately, inside gelatine capsules for posterior identification of the emerged parasitoids. The sex ratio of P. bliteus emerged from the mummies was calculated.
To estimate parasitism, it was considered that the alive N4 and N5 were the fraction of the nymphal population that survived to the parasitoid attack. The parasitism rate was then calculated by the formula:
Parasitism rate = number of "live mummies" / (number of "live mummies" + number of nymphs N4+N5 belonging to the same cohort of the mummies) x 100
Tunisia
In Tunisia, samplings of parasitism rates were conducted on two sites, Moauden and Jbel Eddis, and on two years, August 2012 and July 2013. In each site, on four E. camaldulensis trees,four attacked branches of 50 cm in length were covered with screen bags (60 cm x 45 cm and less than 1 mm mesh size). After 30 days, all emerged insects were counted under binocular. The number of parasitoids and of adult G. brimblecombei was registered. The parasitism rate was determined by the following expression: (Total number of parasitoids emerged per bag/ (Total number of adult psyllid + total number of parasitoids)) x 100.
Data analysis
Data is presented in average values ± standard error (SE). Relationship of infestation levels with latitude and longitude were assessed by using Pearson correlation. A two-way ANOVA, considering the factors year and site was performed to assess differences on the percentage parasitism, as dependent variable, in Tunisia. Statistical analyses were performed using IBM SPSS statistic 21 (MORGAN et al., 2011).
Results
Distribution of Glycaspis brimblecombei in Portugal and Tunisia
Portugal
In the field survey conducted in 2012, the red gum lerp psyllid was found on all regions where E. camaldulensis was present, whereas it was never observed on E. globulus (Figure 3). The mean percentage leaves (±SE) infested on each site varied from 5.0% ± 1.5 to 75.0% ± 0.0. No relationship with latitude or with longitude emerged significant as determined by the Pearson correlation, r =0.328, p =0.051 and r =-0.103, p=0.550, respectively.
Tunisia
In Tunisia, the red gum lerp psyllid G. brimblecombei was first observed in September 2010 on the foliage of E. camaldulensis in the Park of INRGREF in Tunis. In a survey conducted in 2012, the psyllid was found present all over the North of Tunisia (Figure 4). The proportion of attacked leaves varied from 4.9%±2.6 to 95.4%±1.1. Infestation level was not correlated with latitude, r= 0.63, p = 0.092, but was significantly correlated with longitude, r= -0.93, p < 0.001. The percentage of attacked leaves was highest on the West sites: Jebel Eddis, 72.6%±1.2, Ragoubet Essid, 95.4% ±1.1, Mouaden, 87.3%±9.7 and Jebel Essma, 66.7%±12.3. Defoliation due to severe sap sucking activity was recorded in all these localities. Yet, on the East sites, infestation level was about ten times lower: Dar Chichou 13.5% ±4.3), INRGREF, 9.7% ±1.2, Korbous, 7.7% ± 3.2, Jbel Abderrahmane, 4.9% ± 2.5.
Host plants
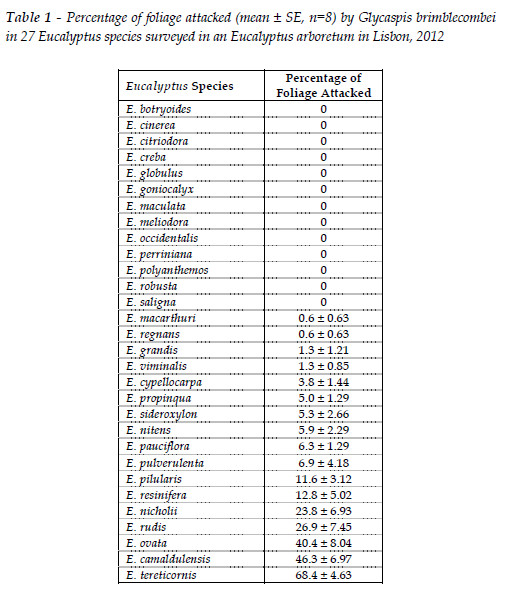
According to the infestation levels observed on an arboretum in Lisbon, E. tereticornis, E. camaldulensis and E. ovata were the most susceptible Eucalyptus species to G. brimblecombei (Table 1). E. globulus, which is the most important planted species in Portugal, was observed to be resistant to G. brimblecombei as well as other 12 species (Table 1). The species E. nitens (Deane & Maiden) Maiden also used for forestry purposes and from the same Botanical section as E. globulus showed an intermediate susceptibility. Other 13 species of Eucalyptus also exhibited the presence of the psyllid at low or intermediate densities (Table 1).
Parasitism by Psyllaephagus bliteus
Portugal
From the study conducted in 2012 at ISA's arboretum the parasitism rate was low in May at the beginning of the study, 1.0±1.0%, when the population of the psyllid started to build up, and attained a maximum of 35±15.0% in October, when the population of the psyllid collapsed. The peak of parasitized nymphs followed with about three weeks delay the peak of total nymphs (Figure 5).
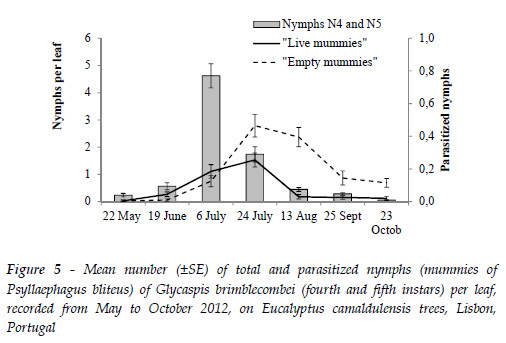
From the collected "live mummies" only P. bliteus emerged (43 males and 54 females). No other parasitoid or hyper-parasitoid was observed.
Mean monthly air temperatures during the observed months varied between 19ºC in May and 23ºC in August. Within the observed period rainfall occurred in May (50 mm), August (8 mm), September (50 mm) and October (110 mm). The collapse of G. brimblecombei started between July and August, before any rainfall occurred.
Tunisia
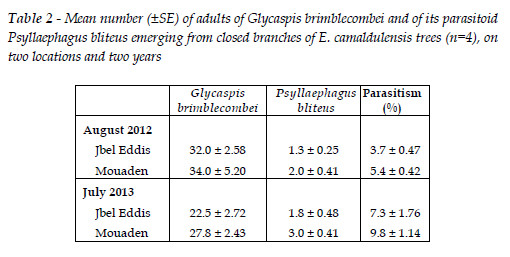
In 2011, a team of researchers prospecting the eucalyptus plantation attacked by G. brimblecombei found nymphs parasitized by P. bliteus in the north of Tunisia. In the evaluations made in 2012 and 2013 in two sites, the proportion of P. bliteus emerging in relation to adult psyllids was found to be low, overall mean 6.5%±0.8 (Table 2). Results from the ANOVA show significant differences between the two years (F1,12 =13.00, p =0.004) but not between sites (F1,12 =3.85, p =0.073) and no interaction site*year (F1,12 =0.133, p =0.721). Parasitism was higher on 2013 in comparison with 2012 (Table 2).
Discussion
In this work the first record of G. brimblecombei in Tunisia and its parasitoid P. bliteus in Portugal and Tunisia is presented. The spatial and temporal sequence of events suggests a possible introduction of G. brimblecombei into Western Europe from North or South America, where the species was present since the nineteens' (BRENNAN et al., 1999) and subsequent spread to eastern Mediterranean countries. In Tunisia higher densities were found on the West region compared to those on the East, which might be related with the invasion process starting from West.
A rapid spread of the red gum psyllid was observed in Portugal and Tunisia following its introduction. In Portugal, five years after its first detection in 2007 (VALENTE and HODKINSON, 2008), the psyllid was now found on all the sites surveyed. The most susceptible Eucalyptus species were found to be E. camaldulensis, E. tereticornis, E. rudis and E. ovata as reported by previous works (BRENNAN et al., 2001). Yet, the presence of the pysllid was here recorded in 11 other species, including E. nitens, one important planted species in Portugal in use to replace E. globulus in areas where Eucalyptus plantations are severely affected by the Eucalyptus weevil Gonipterus platensis Marelli (Coleoptera: Curculionidae) (REIS et al., 2012). Still, E. globulus was found to be resistant to G. brimblecombei, which implies a lower concern about the economic impact of the pest species in Portugal. In Tunisia as well as in other North African countries, E. camaldulensis and E. rudis are among the most used trees for soil protection, biomass and firewood production. Thus higher social and economic impacts of this psyllid may occur in these countries, comparatively to Portugal.
The parasitoid P. bliteus was observed in both countries although it was not imported or released in neither of them. The presence of P. bliteus in Portugal, which is reported here for the first time, was first noticed in June 2011, in Coimbra and Lisbon area (CARLOS VALENTE and CONCEIÇÃO BOAVIDA, pers. communication). In Spain it was detected, in 2010, in the region of Huelva (PÉREZ-OTERO et al., 2011). In 2011, P. bliteus was also detected in Italy without being intentionally imported and released (CALECA et al., 2011). Altogether these observations suggest that some individuals of the parasitoid might have arrived together with the psyllid, accompanying its spread. In some regions, namely in California (EUA) and Brazil, this biological control agent was considered of great interest to control the populations of G. brimblecombei and was mass reared and released (DAHLSTEN et al., 2005; SANTANA and BURCKHARDT, 2007). The parasitism rates found in both Tunisia and Portugal were relatively low. In Portugal, as expected, it was observed that the parasitism rates changed through time and season with the host population dynamics, showing a density-dependent relationship, slightly delayed in time. As a consequence, the calculated parasitism rates were lowest in the beginning of the season, when the psyllid population density was highest but the parasitoid population was still developing. Conversely, the parasitism rates were highest at the end of the season, contributing for the collapse of the psyllid population, together with climate and plant quality. Therefore, present data shows high seasonal variability on the parasitism rate and a density-dependence pattern. As a consequence, during six sampling months, the parasitized mummies represented 1 to 35% of total nymphs. The method used is not as precise as dissecting live psyllids, and might slightly underestimate the total parasitism. A relationship of the parasitism rates with the climate were found by DAANE et al. (2012) in California, with lower parasitism rates observed on drier and hot continental sites (1.2%) than the average parasitism rate at coastal sites (29.7%). Lisbon is located near the coast, as well as the surveyed regions in Tunisia, thus, according to the climate hypothesis, higher parasitism rates are expected to occur in the surveyed regions compared to those in the interior regions of both countries. It is also possible that due to its recent introduction the populations are still increasing.
In Portugal, the intensity of tree infestation was not related with longitude, thus a coastal-inland gradient was not observed. Further studies will be needed to confirm the effect of climate and parasitism rates on these Mediterranean regions and in order to fully understand the population dynamics of G. brimblecombei and the impact of P. bliteus. In both Tunisia and Portugal, a national survey program to estimate the evolution of the infestation and to evaluate the impact of the parasitoid P. bliteus on the G. brimblecombei population is being prepared which will help to clarify the relevance of this natural enemy.
References
BRENNAN, E.B., GILL, R.J., HRUSA, G.F., WEINBAUM, S.A., 1999. First record of Glycaspis brimblecombei (Moore) (Homoptera: Psyllidae) in North America: initial observationsand predator associations of a potentially serious new pest of Eucalyptus in California. Pan-Pacific Entomol 75: 55-57. [ Links ]
BRENNAN, E.B., HRUSA, G.F., WEINBAUM, S.A., LEVISON, W.J., 2001. Resistance of Eucalyptus species to Glycaspis brimblecombei (Homoptera: Psyllidae) in the San Francisco bay area. Pan-Pac Entomol 77: 249–253. [ Links ]
BORRAJO, P., LOPEZ, G., RUIZ, F., 2009. Primera cita de Glycaspis brimblecombei Moore (Homoptera: Psyllidae) en SO de España. Bol Sanidad Veg Plagas 35: 355-361. [ Links ]
BOUVET, J.P.R., HARRAND, L., BURCKHARDT, D., 2005. Primera cita de Blastopsylla occidentalis y Glycaspis brimblecombei (Hemiptera: Psyllidae) para la República Argentina Rev Soc Entomol Argent 64: 99-102. [ Links ]
CALECA, V., LO VERDE, G., MALTESE, M., 2011. First record in Italy of Psyllaephagus bliteus Riek (Hymenoptera Encyrtidae) parasitoid of Glycaspis brimblecombei Moore (Hemiptera: Psyllidae). Naturalista Sicil 37: 435-444. [ Links ]
DAANE, K.M., SIME, K.R., PAINE, T.D., 2012. Climate and the effectiveness of Psyllaephagus bliteus as a parasitoid of the red gum lerp psyllid. Biocont Sci Technol 22(11): 1305-1320. [ Links ]
DAHLSTEN, D., DAANE, K., PAINE, T., SIME, K., LAWSON, A., ROWNEY, D., ROLTSCH, W., JOHN, A., KABASHIMA, J., SHAW, D., ROBB, K., GEISEL, P. CHANEY, EMERITUS, W., INGELS, C., VARELA, L., BIANCHI, M., TAYLOR, G., 2005. Imported parasitoid helps control red gum lerp psyllid. Calif Agric. 59: 229-234. [ Links ]
DHAHRI, S., BEN JAMAA, M.L., 2008. Les insectes ravageurs des eucalyptus en Tunisie. Ann. INRGREF 12(2) NS: 363-372. [ Links ]
DGF - Direction Générale des Forêts, 2005. Résultats du Deuxième Inventaire Forestier et Pastoral National, Ministère de l'Agriculture, des ressources en Eau et de la Pêche. Tunisie, 129 pp. [ Links ]
DGF - Direction Générale des Forêts, 2010. Résultats du deuxième inventaire forestier et pastoral national. Ministère de l'Agriculture de Tunisie, 180 p + annexes. [ Links ]
FIRMINO-WINCKLER, D.C., WILCKEN, C.F., de OLIVEIRA, N.C., de MATOS, C.A.O., 2009. Biologia do psilídeo-de-concha Glycaspis brimblecombei Moore (Hemiptera, Psyllidae) em Eucalyptus spp. Rev Bras Entomol 53(1): 144-146. [ Links ]
PERIS-FELIPO, F.J., MANCUSI, G., TURRISI, G.F., JIMENEZ-PYEDRO, R., 2011. New corological and biological data of the Red Gum Lerp Psyllid, Glycaspis brimblecombei Moore, 1964 in Italy (Hemiptera, Psyllidae) Biodiv Jour 2(1): 13-17. [ Links ]
ICNF, 2013. IFN6 -Áreas dos usos do solo e das espécies florestais de Portugal continental. Resultados preliminares. Instituto da Conservação da Natureza e das Florestas, Lisboa, pp. 33. [ Links ]
KENIS, M., BRANCO, M., 2010. Impact of alien terrestrial arthropods in Europe. Chapter 5. BioRisk 4: 51-71. [ Links ]
KHOUJA, M.L., KHALDI, A., RJEB, M.N., 2001. Results of the eucalyptus introduction trials in Tunisia. Pages 163-168. Proceeding of the International conference; Eucalyptus in the Mediterranean basin: perspectives and new utilization. October 15-19, 2000, Centro Promozione Pubblicità, Florence, Taormina- Italy. [ Links ]
LAUDONIA, S., GARONNA, A.P., 2010. The red gum lerp psyllid, Glycaspis brimblecombei, a new exotic pest of Eucalyptus camaldulensis in Italy. Bull Insectol 63: 233-236. [ Links ]
LO VERDE, G., BELLA, S., CALECA, V., RAPISARDA, C., SIDOTI, A., 2011. Presenza in Sicilia di Glycaspis brimblecombei Moore (Hemiptera Psyllidae) su Eucalyptus camaldulensis Dehnh. Naturalista Sicil 35: 425-434. [ Links ]
MAATOUF, N., LUMARET, J.P., 2012. Eco-ethologie des nouveaux ravageurs invasifs des eucalyptus du Maroc.Ann Soc Entomol Fr 48: 289-297. [ Links ]
MORGAN, G.A., LEECH, N.L., GLOECKNER, G.W., BARRETT, K.C., 2011. IBM SPSS for introductory statistics: Use and interpretation. Taylor & Francis. [ Links ]
PÉREZ-OTERO, R., BORRAJO, P., MANSILLA, J.P., RUIZ, F., 2011. Primera cita en España de Psyllaephagus bliteus Riek (Hymenoptera, Encyrtidae), parasitoide de Glycaspis brimblecombei Moore (Hemiptera, Psyllidae). Bol Sanidad Veg Plagas 37: 37-44. [ Links ]
REIS, A.R., FERREIRA, L., TOMÉ, M., ARAUJO, C., BRANCO, M., 2012. Efficiency of biological control of Gonipterus platensis (Coleoptera: Curculionidae) by Anaphes nitens (Hymenoptera: Mymaridae) in cold areas of the Iberian Peninsula: Implications for defoliation and wood production in Eucalyptus globulus. Forest Ecol Manag 270: 216-222. [ Links ]
SANDOVAL, A., ROTHMANN, S., 2002. Detección del psílido de los eucaliptos rojos, Glycaspis brimblecombei Moore 1964, en Chile (Homoptera: Psyllidae). XXIV Congreso Nacional de Entomología. Santiago, Chile (Abstract). [ Links ]
SANTANA, D.L.Q., BURCKHARDT, D., 2007. Introduced Eucalyptus psyllids in Brazil. J For Res 12: 337–344. [ Links ]
VALENTE, C., HODKINSON, I., 2009. First record of the Red Gum Lerp Psyllid, Glycaspis brimblecombei Moore (Hym.: Psyllidae) in Europe. J App Entomol 133: 315-315. [ Links ]
Submitted for publication in November 2013
Accepted in January 2014
Acknowledgements
This study was funded in Portugal by National Funding through FCT- Fundação para a Ciência e a Tecnologia, National Project PTDC/AGR-CFL/111877/2009 and a bilateral cooperation Project Portugal-Tunisia, 43/TP/09. This work was also supported by the 2nd Project 'Integrated management of Forest' TS-P33 financed by JICA.













