Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Corrosão e Protecção de Materiais
versão On-line ISSN 2182-6587
Corros. Prot. Mater. vol.34 no.1 Lisboa jun. 2015
ARTIGO
Comparative study of polypyrrole films electrosynthesized on zinc surfaces from organic and aqueous solutions
Estudo comparativo de filmes de polipirrol electrosintetizados em superfícies de zinco obtidos em meio orgânico e aquoso
J. I. Martins*1,2, and M. Bazzaoui1,3
(1) Faculdade de Engenharia, Departamento de Engenharia Química, Universidade do Porto, Rua Roberto Frias, 4200-465 Porto, Portugal
(2) LAB2PT, Instituto de Ciências Sociais, Universidade do Minho, Campus de Gualtar, 4710-057 Braga, Portugal, e-mail: bazzaoui@fe.up.pt
(3) Equipe de Chimie de Coordenation, Laboratoire des Matériaux et Environnement, Département de Chimie, Faculté des Sciences, Université Ibnou Zohr, B.P. 8106, Agadir, Morocco
(*) Corresponding author, e-mail: jipm@fe.up.pt
ABSTRACT
Homogeneous and adherent polypyrrole coatings have been achieved on zinc electrodes previously treated with an aqueous solution of sulfide in the acetonitrile medium with tetraethylammonium p-toluene sulfonate, and in the tartrate aqueous medium without any chemical or electrochemical pretreatment of the working electrode. The XPS and AES analysis showed that the dissolution of the working electrodes is inhibited in the first polarization medium by the oxide and zinc sulfide formed on the surface during the pretreatment in 0.2 M Na2S after 12 h, or by the zinc tartrate film developed through the electrode polarization in the second. The adherence, compactness and corrosion performance in 0.1 M HCl of the polypyrrole coatings from the tartrate solution are higher than those obtained from acetonitrile medium.
Keywords: Polypyrrole, Conducting Polymers, Tartrate, Electropolymerization, Zinc Corrosion
RESUMO
Revestimentos de polipirrol homogéneos e aderentes foram obtidos sobre eléctrodos de zinco previamente tratados numa solução aquosa de sulfureto em meio de acetonitrilo com p-tolueno-sulfonato de tetraetilamónio, e em meio aquoso de tartarato sem qualquer pré-tratamento químico ou electroquímico do eléctrodo de trabalho. As análises por XPS e AES mostraram que a dissolução dos eléctrodos de trabalho é inibida no primeiro meio de polarização pelo óxido e sulfureto de zinco formados sobre a superfície durante o pré-tratamento em Na2S 0,2 M após 12 h, ou pelo filme de tartarato de zinco desenvolvido sobre o eléctrodo no segundo meio de polarização. A adesão, compacidade e comportamento à corrosão em HCl 0,1 M dos revestimentos de polipirrol obtidos a partir da solução de tartarato são melhores do que os provenientes do meio de acetonitrilo.
Palavras-chave: Polipirrol, Polímero Condutor, Tartarato, Electropolimerização, Corrosão do Zinco
1. INTRODUCTION
Since their discovery that conducting polymers rapidly attracted the attention of scientists, taking into account their interesting physical and chemical properties and vast range of applications [1-3]. The electrosynthesis of conducting polymers on oxidizable metals are a good alternative to primaries based on chromate, widely used in metallurgical industry in the fight against corrosion. The electrodeposition of conducting polymers in oxidizable metals is not easy. The anodic dissolution of working electrode before the electropolymerization of the monomer inhibits oxidation. It is therefore necessary to define a strategy to reduce corrosion of the metal substrate without, however, inhibit the electropolymerization.
The pyrrole is one of the most investigated organic compounds in view of its good solubility in organic and aqueous media, easy handling and high environmental stability of the polymer [4-10]. The electrosynthesis of polypyrrole (PPy) on zinc electrodes in an aqueous medium of sodium oxalate has been achieved, but only after a pre-treatment of the electrode in a sodium sulphide solution for a few hours [11]. This two-step process was reduced to one step by introducing a small amount of Na2S in the sodium oxalate solution [9]. Later, some researchers showed to be possible to obtain homogeneous and adherent PPy film on the zinc surface without any chemical or electrochemical pre-treatment, from aqueous media using sodium salicylate [12] or sodium tartrate [13].
The aim of this study is to compare some properties of PPy films electrosynthesised on zinc surfaces from the organic and aqueous media.
2. EXPERIMENTAL
2.1. Chemicals
All chemicals compounds were reagents grade. Tetraethylammonium p-toluene sulfonate (NEt4TsO), sodium sulphide (Na2S), acetonitrile (CH3CN), sodium citrate (Na3C6H5O7.2H2O, 99%), sodium tartrate (Na3C6H5O7.2H2O, 99%), malic acid (C4H6O5, 99%), sodium succinate (Na2C4H4O4, 99%), sodium oxalate (Na2C2O4, 99%) and sodium hydroxide (99%) were purchased from Aldrich and used as received. The pyrrole monomer (Aldrich) was distilled twice under nitrogen and water was distilled twice.
Before each experiment the electrochemical organic electrolytic solution was deoxygenated by nitrogen bubbling, but aqueous solutions were used as such.
2.2. Electrochemical apparatus
The electrochemical experiments were performed in a onecompartment cell with three electrodes connected to Autolab model PGSTAT20 potentiostat/galvanostat with pilot integration controlled by GPES 4.4 software. The working electrode was constituted of Zn (99% purity) rectangular sheets (20 mm2 x 50 mm2). The electrodes were mechanically polished with abrasive paper (1200-grade), and rinsed in acetone before each trial. A stainless-steel plate was used as auxiliary electrode. The potentials were measured versus SCE or Ag/ AgCl reference electrodes.
2.3. SEM, XPS measurements
Scanning electron microscopy (SEM) micrographs were obtained on a JEOL JSM-6301F equipment. The microscope chamber was maintained at a pressure between 4 and 10 Pa. The distance between the sample and the objective lens is remained at 15 mm (working distance). The SEM filament was operated at variable current and a voltage of 15 kV.
X-ray photoelectron spectroscopy (XPS) analysis was carried out with a Vacuum Generators VG Scientific Escalab 200A spectrometer with Pisces software for data acquisition and analysis. For analysis, an achromatic Al (Kα 1486.6 eV) X-ray source operating at 15 kV (300 W) was used. Pressures in the analysis chamber ranged from 10−6 to 10−7 Pa and the analyzed area is about 10 mm2 size. The spectrometer, calibrated with Ag 3d5/2 (368.27 eV) reference, was operated in CAE mode with 20 eV pass energy. The spectra was corrected for the surface charging effect by taking the C 1s peak (EB = 285 eV) as an internal reference. Spectra analysis was performed using peak fitting with Gaussian–Lorentzian peak shape and linear type background subtraction.
2.4 Adherence
Adherence measurements were based on the standard Sellotape test, using the TESA-4204 BDF tape, which consists in cutting the film into small squares, sticking the tape and then stripping it. The ratio between the number of adherent film squares remaining and their total number gives the percentage adherence.
2.5 Corrosion measurements
Electrochemical corrosion tests were performed at room temperature in 0.1 M HCl solution on working electrode with area of 10 cm2. DC polarization experiments were always started from the open circuit potential at the scan rate of 2 mV s-1.
The galvanostatic weight loss of the naked and PPy-coated bars electrodes (AA analysis of zinc ions in the solution) was performed for applied current densities in the range of 0.1 – 10 mA cm-2.
3. RESULTS AND DISCUSSION
3.1. Electrochemical behaviour of zinc electrodes in different solutions
Before the electrosynthesis, the electrochemical behaviour of zinc electrode in organic and aqueous media, in the absence of the monomer, has been monitored.
3.1.1. Organic medium {CH3CN+ 0.1 M N(Et)4TsO}
Influence of the electrode pretreatment
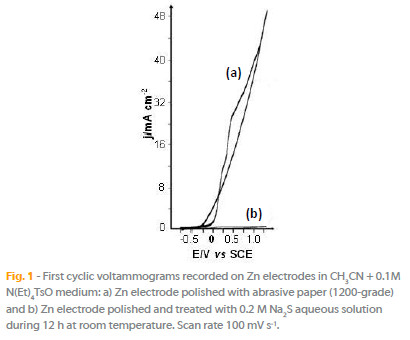
Two kinds of zinc working electrodes have been used in the trials: i) electrodes mechanically polished with abrasive paper (1200-grade) and rinsed with acetone under ultrasonic waves; ii) and electrodes polished and chemically treated in a solution of 0.2 M Na2S during 12 hours at room temperature.
On the Fig. 1 were superposed the first potential sweeps obtained in the electrolytic medium (CH3CN + 0.1M N(Et)4TsO) on Zn plates, without (Fig. 1a) and with (Fig. 1b) a pretreatment in sulfide solution. The potentiodynamic curve of the non-treated zinc electrode shows an oxidation anodic wave characterized by an increase of current density, which reaches a value of approximately 40 mA cm-2 at the potential 1.0 V vs. SCE. In the case of pretreated Zn electrode, the current density does not exceed about 1 mA cm-2.
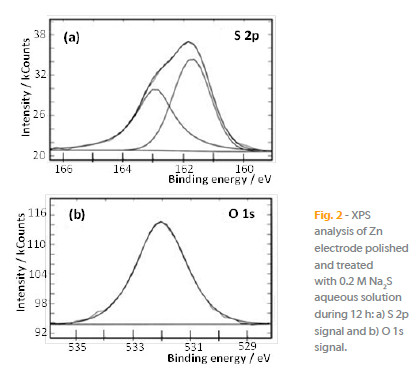
The XPS analysis of sulfur S 2p (Fig. 2a) and oxygen O 1s (Fig. 2b) on zinc electrode surface pretreated with 0.2 M Na2S reveals the presence of a S 2p doublet at 161.7 and 162.9 eV characteristic of zinc sulfide ZnS [14, 15], and a peak O 1s at 532.0 eV related with zinc oxide and hydroxide [14, 16], in full agreement with the results reported in the literature [5] for zinc and zinc-coated electrodes. These data show that the passivation layer developed on the electrode surface after pretreatment with sodium sulfide are composed by a mixture of zinc sulfide, zinc oxide and zinc hydroxide, which inhibits successfully the electrode dissolution.
3.1.2. Aqueous media {H2O + 0.2 M carboxylate salt (Na3C6H5O7, Na2C4H4O6, Na2C4H4O5, Na2C4H4O4 and Na2C2O2)}
The Fig. 3 presents the polarization curves of zinc electrodes in different aqueous solutions of carboxylate salts of linear chain at pH @ 6.9.
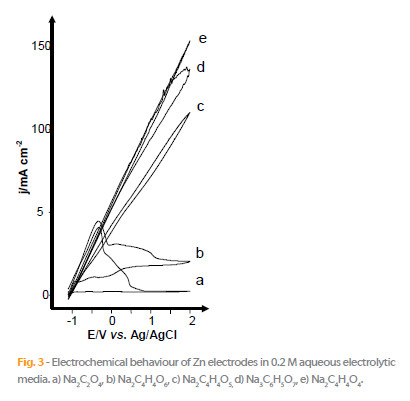
The j-E curves recorded in aqueous solutions of succinate, malate and citrate show the oxidation waves that beginning at -1 V vs. Ag/AgCl with high current densities that correspond to the metal dissolution, since a white deposit of zinc oxide covered the electrode surface. Conversely, with the oxalate and tartrate supporting electrolytes, the potentiodynamic curves show a passivation phenomenon. The electrochemical behaviour begins with complex anodic peaks attributed to the electrode dissolution and the formation of a grey passivation layer, leading to the following reactions:
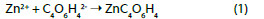
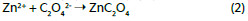
The elemental composition of the passivation layer formed on Zn electrode surface after polarization during two scans between -1 and 2 V vs. Ag/AgCl in 0.2 M Na2C4H4O6 has been investigated by XPS and AES, Fig. 4.
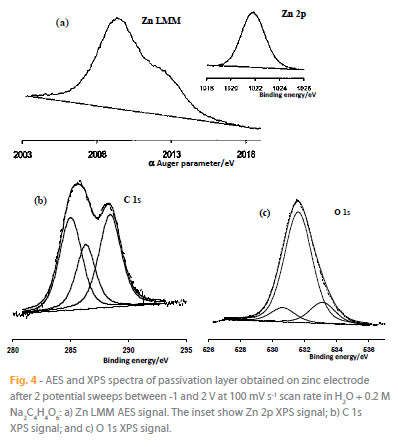
The analysis reveals the presence of Zn, C and O. The use of AES technique was to cross the difficulties encountered in the decomposition of Zn 2p XPS signal. Indeed, the difference between the two oxidation states Zn (0) and Zn (II) binding energies is too small [17-19], whereas the kinetic energies of these chemical states presented in Fig. 4a are largely distinguished in Auger electron spectroscopy spectrum. In this context, the Auger parameter a is defined in the following equation:
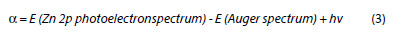
Where E is the binding energy (eV), and hv the incident wave energy (in our case hv = 1253.6 eV). In the basis of the data reported in the literature relating to values of the a Auger parameter for the oxidation states of zinc [19], the whole passivation layer encloses the Zn(II). The same result has been obtained for zinc electrode polarized in aqueous solution of sodium salicylate [20].
On the other hand, the C 1s (Fig. 4b) signal consists of three peaks at 285, 286.3 and 288.4 eV attributed to carbon atoms of C-C, C-O and C=O groups, which belonging to tartrate supporting electrolyte.
The oxygen signal O 1s (Fig. 4c) is composed of three peaks at 530.6, 531.7 and 533.6 eV, whose relative intensities are 0.24:1:0.30, respectively. The main peak at 531.7 eV corresponds to oxygen from tartrate anion and comprises two shoulders: the first at 530.6 eV is associated with zinc oxide, and the second at 533.6 eV could be due to some water trapped inside of the passivation film [21].
As conclusion, the spectroscopic analysis shows that the passivation layer obtained on zinc electrode in tartrate medium is composed by zinc tartrate and some amount of zinc oxide. In aqueous medium of oxalate, according with previous works [14], the passive film is composed exclusively by zinc oxalate.
3.2. Electropolymerization of pyrrole on zinc electrodes in organic and aqueous media
The electrosynthesis of polypyrrole in {CH3CN + 0.1 M N(Et)4TsO + 0.3 M Pyrrole} and aqueous media using different carboxylate salts of linear chain (Na2CxHyOz) of 0.2 M concentration and with 0.5 M pyrrole, was investigated. The behaviour of the voltamperometric curves of zinc plates changes as compared to that obtained in the absence of the monomer.
In organic medium j-E curves recorded on unpretreated Zn electrode show a competition between the reaction of the electrode oxidation and the pyrrole electropolymerization, Fig. 5a. The electrochemical process begins in the first sweeps by the oxidation of the Zn electrode surface, and on the followings appear two peaks at 0.5 V vs. SCE and -0.65 V vs. SCE attributed to oxidation and reduction of the polymer. According to the results of XPS analysis, a composite film of PPy and Zn oxide is formed on the metallic surface. Meanwhile, when the electrode undergoes a preliminary treatment with sulfides, the surface of the electrode is stabilized by the ZnS passivation layer, and the zinc oxidation is prevented. The j-E curves recorded in Fig. 5b are comparable to those obtained in the same electrolytic medium on an inert platinum electrode, Fig. 5c, and the film formed on the Zn surface is adherent and uniform. The oxidation and reduction peaks of polymer are well defined, and their intensity increases with the thickness.
In aqueous media, the voltammograms recorded between -1.1 and 2.0 V vs. Ag/AgCl with 100 mV s-1 scan rate show an anodic wave that begins at -1.1 V vs. Ag/AgCl with a peak at -0.6 V vs. Ag/AgCl during the first potential sweep (Fig. 5d), which appears also in the absence of pyrrole and consequently is attributed to the oxidation of zinc. During the later potential sweeps, the oxidation peak of zinc disappears and anodic waves similar to that observed in the case of platinum electrode (Fig. 5e) occur, and correspond to the pyrrole electropolymerization with adherent and homogeneous films. As it is seen, the reduction peak of the polymer film does not appear in the voltammetric curves of electropolymerization (Fig. 5d), contrary to the organic medium. This behavior seems to indicate that the PPy layer obtained from the tartrate aqueous medium is more compact than from the acetonitrile organic medium. So, the low mobility of the tartrate anion (C4H4O6 2-) in comparison with the para-toluene sulfonate (TsO-) through the PPy backbone, leads to migration of cation (Na+) to inside the film to maintain the charge neutrality during the reduction step. In the organic medium the porosity of the PPy film allows both the migration of the doping anion (TsO-) to into as to out the PPy film during its oxidation and reduction, respectively. A similar phenomenon has been observed with PPy doped with oxalate or salicylate in aqueous medium [14, 20].
The systems using oxalate as supporting electrolyte have difficulty to electropolymerize the pyrrole due to blocking of passivation promoted by zinc oxalate layer. However, some authors [14] by introducing sodium sulfide into the electrolysis solution at pH = 6 have activated this layer allowing the pyrrole electropolymerization.
Finally, with succinate, malate and citrate salts, only have been observed the electrode oxidation. This is in agreement with strong electrode dissolution in the absence of the monomer, as it was reported above.
Furthermore, the electropolymerization of pyrrole was also successfully achieved on zinc electrodes under the galvanostatic mode using the same electrolytic conditions of cyclic voltammetry. The chronopotentiometric curves exhibit an induction time related with the electrode dissolution, and followed by a plateau of potential where the monomer is oxidized. The induction time decreases with increase in the current density, and is shorter for the organic medium comparing to the aqueous, since the working electrode was previously passivated by sodium sulfide that inhibits partially its dissolution.
The Fig. 6 shows the evolution of the potential plateau (Ep) with the applied current density: the polymerization potential increases with the applied current density. It is clear that the current density has to be higher than 1 mA cm-2 and 5 mA cm-2, respectively, for the acetonitrile and aqueous media, to obtain the electropolymerization of pyrrole.
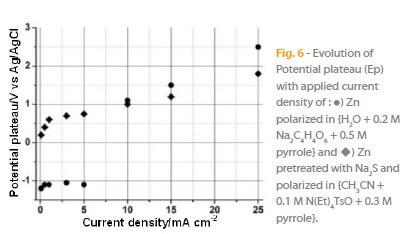
3.3. Characterization of the PPy films
3.3.1. Morphology and adherence
In acetonitrile and in the presence of 0.1 M N(Et)4TsO and 0.3 M pyrrole the surface properties of the PPy films obtained at 2.5 mA cm-2 on Zn electrodes during 10 minutes with and without pretreatment are very different. Homogeneous and adherent films are obtained on pretreated Zn surface (Fig. 7A and Fig. 7B), and the quality of the polymer is so excellent that no crack or detachment of the film is observed. Conversely, the PPy coating from Zn electrodes without chemical pretreatment is inhomogeneous, poorly adherent and with cracks (Fig. 7C). Moreover, the coating becomes also brittle with the increasing of current density to a value of 10 mA cm-2 for the pretreated electrodes with Na2S (Fig. 7D).
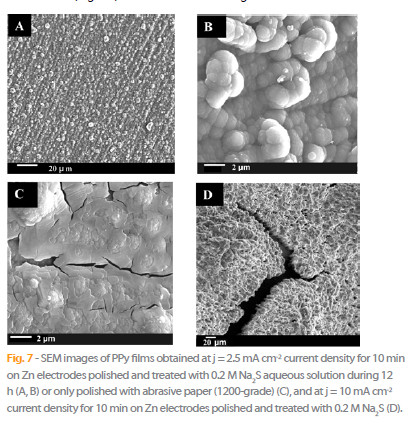
On the other hand, the surface structure of PPy films galvanostatically prepared on zinc electrode at 15 mA cm-2 in {H2O + 0.2 M Na2C4H4O6 + 0.5 M pyrrole} during 10 min is homogeneous, strongly adherent and present a cauliflower structure constituted of clusters of globules (≈ 5-10 μm diameter), Fig. 8.

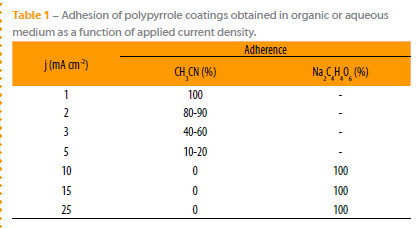
The applied current densities have a great effect on the adherence of coatings electrosynthesized in organic medium. The low applied current densities are the more suitable for attaining a good adherence of PPy film obtained in organic medium. In the case of PPy films produced in aqueous medium of tartrate, the adherence was estimated at 100 % for applied current densities in the range between 10 and 25 mA cm-2, Table 1.
3.3.2. XPS analysis
The XPS data for PPy films obtained under the organic and aqueous conditions described, exhibit the same features like those of standard PPy films of the literature [22, 23]. The assignments of the binding energies are summarized in the Table 2 [24-26].
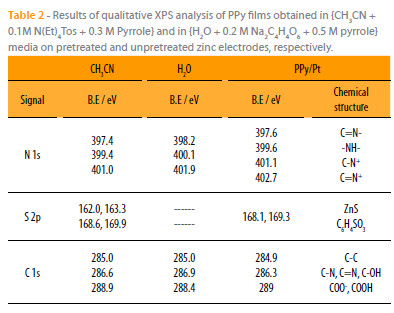
In spite of the homogeneity of PPy coatings observed by scanning electron microscopy, the X-ray photoelectron spectroscopy results of PPy films synthesized in organic medium show some cracks on the polymer surface. Indeed, the XPS signal of sulfur S 2p comprises in addition to the intense doublet at 168.6 and 169.9 eV belonging to the doping tosylate anions, another very weak located at 162.0 and 163.3 eV attributed to some traces of zinc sulfide. However, any XPS signal of Zn 2p was detected on the outer side of the PPy films electrosynthesized in tartrate aqueous medium, which clearly indicates a better homogeneity and compactness.
3.3.3. Corrosion performance of PPy films
The coated electrodes to test, with a thickness of the order of 12 μm, have been obtained galvanostatically in organic ({CH3CN + 0.1 M N(Et)4TsO + 0.3 M Pyrrole}) and aqueous ({H2O + 0.2 M Na2C4H4O6 + 0.5 M pyrrole}) media.
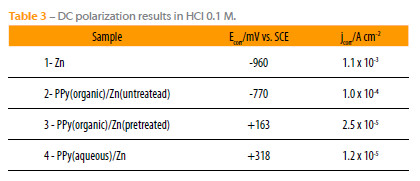
The Tafel plots from the DC polarization tests in 0.1 M HCl, for the PPy-coated and uncoated zinc surface, Fig. 9, allowed estimating the potentials and current densities of corrosion, Table 3. The calculated values show that PPy coatings significantly increase the corrosion potential and sharply reduce the corrosion current densities. The corrosion potential of zinc coated with PPy films obtained from aqueous solution is nobler than that obtained from organic medium with a pretreatment of the surface, while the corrosion current density is lesser.
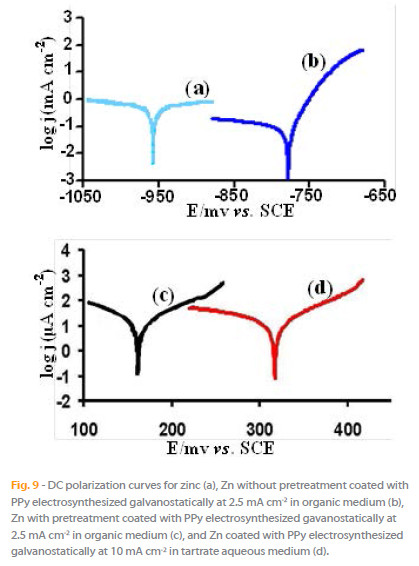
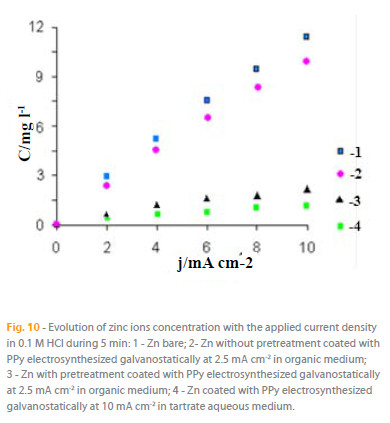
In order to understanding ionic mass transfer through the micropores of the PPy structure, we have studied the behaviour of the naked and PPy-coated Zn bar in 0.1 M HCl medium under different applied current densities, analysing the ionic metallic content in the solution (Fig. 10) after five minutes of the experiment. The results show the following: 1) the bar zinc corrosion is of the same or der to that of PPy(organic medium)- Zn(without pretreatment); 2) the corrosion of zinc elec trode is about ten times higher than that observed for both PPy coatings, from aqueous or organic medium with a sur face pretreatment; 3) the content of zinc in the acid solution f or PPy(organic medium)- Zn(preatread in H2S solution) is twice the values observed for the PPy(tartrate aqueous medium)-Zn, in fully agr eement with that predicted from the Tafel results.
3.3.4. Application of the PPy electrosynthesis from aqueous tartrate solution
To concretize our work concerning the pyrrole electropolymerization on zinc substrates, we present handfuls of door brass (33 % Zn, 67 % Cu) covered with PPy from tartrate solutions.
The samples were mechanically polished, cleaned with an alkaline solution at 60 º C and rinsed with water. The electrosynthesis of PPy was performed galvanostatically by imposing a current density of 20 mA cm-2 during 15 minutes in {H2O + 0.2 M Na2C4H4O6+ 0.5 M pyrrole}.
The PPy coating has been soft polished with abrasive paper (200-grade) followed by diamond paste (1 μm). The handfuls exhibit a remarkable looking black good as is shown in Fig. 11. This application is not limited only to the handfuls of door, but can be extended to others oxidizable objects used in our life.

4. CONCLUSION
The electrosynthesis of PPy on zinc electrodes was achieved successfully in sodium tartrate aqueous solution and in acetonitrile in the presence of para-toluene sulfonate salt. A previous chemical treatment with Na2S during 12 hours at room temperature was necessary in the case of organic medium.
The adherence of the coating synthesized in aqueous medium was estimated at 100% at high current densities. In organic medium the adherence and compactness decreases with the applied current density even after a surface pretreatment.
In brief, the pyrrole electropolymerization on zinc electrode from tartrate aqueous medium is better than that from acetonitrile organic solution for the following reasons:
• One step in the electropolymerization procedure is enough, while two steps are necessary for the elaboration of PPy in acetonitrile medium.
• Better adherence, compactness and corrosion performance concerned the galvanostatically applied current densities.
• The aqueous medium is cheaper, more ecological and stable.
REFERENCES
[1] P. Metz, G. Alici and G. M. Spinks, Sensor Actuat A-Phys., 130-131, 1-11 (2006). [ Links ]
[2] X. Luo, A. J. Killard, A. Morrin and M. R. Smyth, Anal. Chim. Acta, 575, 39- 44 (2006). [ Links ]
[3] S. Ampuero and J. O. Bosset, Sensor Actuat B - Chem., 94, 1-12 (2003). [ Links ]
[4] E. J. Oh, K. S. Jang and A. G. MacDiarmid, Synthetic. Met., 125, 267-272 (2002). [ Links ]
[5] F. Beck and R. Michaelis, J. Coating Technol., 64, 59-67 (1992). [ Links ]
[6] M. Bazzaoui, L. Martins, E. A. Bazzaoui and J. I. Martins, Electrochim. Acta, 47, 2953-2962 (2002). [ Links ]
[7] J. I. Martins, M. Bazzaoui, T. C. Reis, E.A. Bazzaoui and L. Martins, Synthetic. Met., 129, 221-228 (2002). [ Links ]
[8] M. Bazzaoui, E. A. Bazzaoui, L. Martins and J. I. Martins, Synthetic. Met.,130, 73-83 (2002) [ Links ]
[9] M. Bazzaoui, J. I. Martins, T. C. Reis, E. A. Bazzaoui, M. C. Nunes and L. Martins, Thin Solid Films, 485, 155-159 (2005). [ Links ]
[10] J. I. Martins, T. C. Reis, M. Bazzaoui, M. C. Nunes, L. Martins and E. A. Bazzaoui, Prog. Org. Coat., 65, 62-70 (2009). [ Links ]
[11] B. Zaid, S. Aeiyach, P. C. Lacaze and H. Takenouti, Electrochim. Acta, 43, 2331-2339 (1998). [ Links ]
[12] M. Bazzaoui, L. Martins, E. A. Bazzaoui and J. I. Martins, J. Electroanal. Chem., 537, 47-57 (2002). [ Links ]
[13] J. Petitjean, S. Aeiyach, J. C. Lacroix and P. C. Lacaze, J. Electroanal. Chem., 478, 92-100 (1999). [ Links ]
[14] S. Aeiyach, B. Zaid and P. C. Lacaze, Electrochim. Acta, 44, 2889-2898 (1999). [ Links ]
[15] G. Deroubaix and P. Marcus, Surf. Interface Anal., 18, 39-46 (1992). [ Links ]
[16] C. D. Wagner, W. M. Riggs, L. E. Davis and J. F. Moulder, Handbook of X-Ray Photoelectron Spectroscopy, G. E. Mullenberg Ed., Perkin Elmer Corp., Minnesota (1978). [ Links ]
[17] C. A. Ferreira, B. Zaid, S. Aeiyach and P. C. Lacaze, Organic Coatings, P. C. Lacaze (Ed.), AIP Press, Woodbury, New York, pp. 159-165 (1996). [ Links ]
[18] J. F. Moulder, W. F. Stickle, P. E. Sobol and K. D. Bomben, Handbook of X- Ray Photoelectron Spectroscopy, J. Chastain, Ed., Perkin-Elmer Corporation, Eden Prairie, pp. 204 (1992). [ Links ]
[19] G. Moretti, J. Electron. Spectrosc., 95, 95-144 (1998). [ Links ]
[20] E. Hermelin, J. Petitjean, S. Aeiyach, J. C. Lacroix and P. C. Lacaze, La Revue de Métallurgie /Sciences et Génie des Matériaux 97, 1501-1508 (2000). [ Links ]
[21] T. C. Lin, G. Seshadri and J. A. Kelber, Appl. Surf. Sci., 119, 83-92 (1997). [ Links ]
[22] K. G. Neoh, K. K. S. Lau, V. V. T. Wong, E. T. Kang and K. L. Tan, Chem. Mater., 8, 167-172 (1996). [ Links ]
[23] E. T. Kang, K. G. Neoh and K. L. Tan, Adv. Polym. Sci., 106, 135-190 (1993). [ Links ]
[24] C. Malitesta, I. Losito, L. Sabbatini and P. G. Zambonin, J. Electron. Spectrosc., 76, 629-634 (1995). [ Links ]
[25] G. E. De Benedetto, C. Malitesta, F. Palmisano and P. G. Zambonin, Anal. Chim. Acta, 389, 197-204 (1999). [ Links ]
[26] C. S. Lee, J. Joo, S. Han and S. K. Koh, Sens. Actuators A, 121, 373-381 (2005). [ Links ]
Acknowledgements
This work has financial support from the UID/AUR/04509/2013 Project by FCTMEC through national funds and where applicable, the FEDER cofinancing under the new PT2020 partnership agreement.
Artigo submetido em Dezembro de 2014 e aceite em Fevereiro de 2015














