Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Corrosão e Protecção de Materiais
versão On-line ISSN 2182-6587
Corros. Prot. Mater. vol.32 no.3 Lisboa set. 2013
ARTIGO
Strengthening mechanisms of materials for high temperature application
Mecanismos de endurecimento dos materiais para aplicações a altas temperaturas
C. A. C. Sequeira(1)(*) and L. Amaral(1)
(1) Materials Electrochemistry Group, ICEMS, Instituto Superior Técnico, Technical University of Lisbon, Av. Rovisco Pais, Nº 1, 1049-001 Lisbon
(*) Correspondig author, e-mail: cesarsequeira@ist.utl.pt
ABSTRACT
Metallic materials relevant for high temperature application include the nickel and cobalt base alloys and, to some extent, the titanium alloys and steels. In this paper, the mechanisms by which these alloys are strengthened and, more specifically, the strength achievements in these basic structural materials are discussed.
Keywords: Strengthening Mechanisms, Titanium Alloys, Steels, Nickeliron Alloys, Nickel Superalloys, Cobalt Superalloys
RESUMO
Os materiais metálicos para aplicações em sistemas que operam a altas temperaturas incluem ligas de níquel e de cobalto, bem como ligas de titânio e os aços. Neste artigo discutem-se os mecanismos de endurecimento desses materiais e, mais concretamente, analisam-se os procedimentos que conduzem a esse estado.
Palavras-chave: Mecanismos de Endurecimento, Ligas de Titânio, Aços, Ligas de Níquel-ferro, Superligas de Níquel, Superligas de Cobalto
1. INTRODUCTION
A significant number of industrial processes take place in very aggressive environments. Industrial components are often subjected to extreme conditions, which induce erosion and impact damage.
Some examples of such conditions are high temperature, increased temperature gradients, high pressure, large stresses, or the presence of oxidizing and corroding atmospheres, as well as internally created or externally ingested particulate material. Fig. 1 illustrates several high temperature processes, showing the range of temperatures and required lives for the respective components.
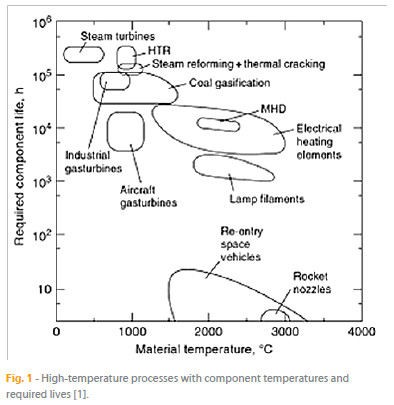
Machines like aircraft gas turbine engines, steam turbines, industrial gas turbines, or processes such as coal conversion, petroleum refining, and nuclear power generation clearly involve the generation of large amounts of heat and the associated high component temperature. In the case of jet engines, fuel is mixed with highly compressed air and the mixture ignited. The turbine is rotated as a result of the air expansion due to the heat generated. On the other hand, the turbine forces the compressor to rotate, leading to the compression of the incoming air, while the existing exhaust gas creates thrust for propulsion. In modern gas turbine engines, the gas temperature may well exceed 1650 ºC in the turbine section, with cooled parts reaching temperatures as high as 1200 ºC. In coal gasifiers, coal is converted into usable gas by reacting with steam at high temperatures. In this case, process temperatures may be as high as 1650 ºC with component temperatures reaching 1090 ºC. Regarding the process of petroleum refining, crude petroleum is catalytically cracked at high temperatures and fractioned into usable petroleum products such as liquid petroleum gas, gasoline, kerosene, diesel, heavy oils, plastics, asphalt and coke. In nuclear power plants, the controlled fission of fuel elements, such as uranium and thorium, generates heat used to produce steam, which is by its turn fed to turbines to generate electrical power.
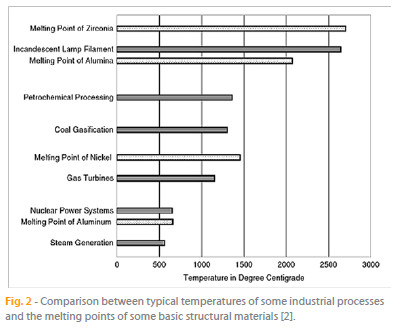
All the mentioned processes involve high temperatures and therefore require construction materials with high-temperature capability under load to meet performance and durability requirements. During operation, in addition to fatigue and creep damage of structurally loaded components, the materials undergo oxidation, corrosion, and erosive wear. In Fig. 2, typical temperatures of some industrial processes are compared with the melting points of some basic structural materials.
The variation with the temperature of the strength ranges of a few potential materials of construction for high-temperature processes is shown in Fig. 3 [3]. Several properties of many of these engineering materials, such as tensile, creep, and fatigue strength, are generally optimized for maximum load-carrying capability but less attention is paid to the environmental resistance. For example, turbine blades for jet engines are made of precipitation-strengthened nickel base superalloys, consisting in gamma prime (? ) phase precipitates in a gamma (?) phase matrix. Aluminum is one of the constituents of the alloys, which participates in the formation of strengthening precipitates and provides for oxidation resistance. The aluminum content is kept at a level below 6 % in order to maximize creep strength, although higher aluminum content increases high-temperature oxidation resistance. If such bare alloys are exposed to the environment of high-pressure turbine of modern gas turbine engines, they will degrade fast by several processes including oxidation [4], high-temperature corrosion [5], and heat damage [6].
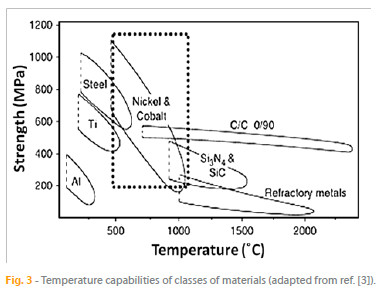
Despite the fact that non-metallic materials such as monolithic ceramics and ceramic composites are becoming increasingly important in some of the above mentioned industrial processes, the demand for materials for those applications is met predominantly by metallic alloys. Properties such as high melting point, strength (tensile strength, creep strength, fatigue strength), ductility and toughness, and low density wherever possible, are the reason for the attractiveness of these alloys. However, the environmental resistance of the metallic alloys is rarely adequate.
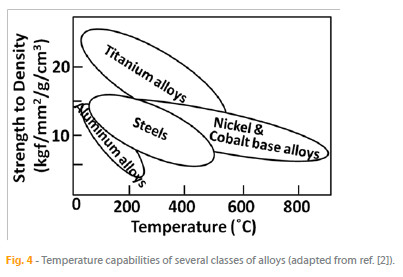
Four groups of alloys support the bulk of the demand in industrial processes. These groups are based on the metals aluminum, titanium, iron, and nickel and cobalt. The dependence on the temperature of the range of density-corrected strengths of these classes of alloys is schematically depicted in Fig. 4. It is clear that aluminum, with its low melting point of 660 ºC and its commercially available alloys with temperature capability limited to about 150 ºC, is not adequate for high-temperature applications. The only alloys relevant to such hightemperature applications include the nickel and cobalt based alloys and, to some extent, the titanium alloys and steels. The mechanisms by which these alloys are strengthened and, more specifically, the strength achievements in the basic structural high-temperature materials are discussed in this paper.
2. STRENGTHENING MECHANISMS
Pure metals are weak and usually have poor environmental resistance. The strengthening of materials is therefore of great importance for engineering applications. Construction parts must be designed not only to endure the anticipated forces (which materials are expected to withstand during service), but also any sudden, short-duration forces that, if not considered, might cause catastrophic failure. Liberal safety factors are generally adopted by designers, to ensure the safe use of a construction part, even in the case that a sudden force of larger magnitude appeared during service. For that purpose, various strengthening mechanisms are usually employed, as well as surface treatments to increase the protection from the environment. Strengthening generally involve dislocation generation, dislocation movement and the interactions of dislocations with each other or with other entities that might hinder their free motion. A summary of metals strengthening processes is presented in Table 1.

Solid solution strengthening and precipitation hardening are effective mechanisms at high temperatures and, therefore, are used in strengthening high-temperature alloys. Grain-size control is often used in conjunction with these mechanisms. By its turn, oxide dispersion strengthening (ODS) is sparsely used in some cases for moderate-temperature applications. The remaining mechanisms, martensitic transformation and work hardening, are not effective at temperatures of interest, being used only for low temperature applications.
Solid solution strengthening and precipitation hardening mechanisms involve hindering the movement of dislocations in the metallic structure. In a solid solution, appropriate alloying elements (the solute) are dissolved in a metal (the solvent). The solute atoms randomly substitute for the solvent atoms (Fig. 5 (a) ) without altering the phase, i.e., the crystal structure. Depending on whether the substituting atom is larger or smaller than the solvent atom, compressive or tensile stresses, respectively, are generated in the vicinity of the solute atoms. The motion of the dislocations is then impeded by the presence of stress fields. The size differences between the solvent and the solute atoms and the volume fraction of the solute are determinant in the effectiveness of the strengthening. For this mechanism to be effective, the solute atoms must be reasonably soluble in the solvent metal, and the atomic size difference must be significant.
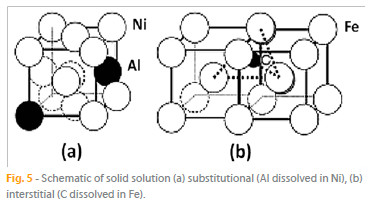
In addition to the substitutional solid solution in which solute atoms randomly replace solvent atoms, the solute atoms may also occupy interstitial sites in the solvent (Fig. 5 (b)). For example, carbon, which is a small atom, forms an interstitial solid solution with iron. The stress fields originated by the interstitial atoms also interfere with dislocation motion, thus influencing strength.
In the case of precipitation hardening, the solute atoms promote the formation of a fine and uniformly dispersed second phase. To achieve this condition, the solvent metal needs to form a supersaturated solid solution at high temperature. On cooling, the solute atoms are rejected from solution in the form of fine precipitates, due to the decrease of solubility. These precipitates then impede dislocation motion, with the strength achieved by this process being controlled by the size, strength, coherence and volume fraction of the precipitates. These parameters may be controlled through appropriate heat treatment of the alloys.
Following this brief discussion, more detailed aspects concerning strengthening in the metallic materials for high temperature applications are analysed hereafter.
3. STRENGTH ACHIEVEMENT IN HIGH TEMPERATURE ALLOYS
3.1 Titanium alloys
Titanium and its alloys present very interesting structural properties [7]. The metal allies a high melting point of 1668 ºC and excellent corrosion resistance to a low density, 4.54 y/cm3 (about 60 % of that of steel and nickel and cobalt base superalloys). Titanium exhibits a hexagonal close packed (hcp) structure called a which transforms on heating to above 883 ºC into a body-centered cubic (bcc) structure b. The material, on a density-corrected basis, forms some of the strongest alloys available. Titanium alloys are extensively used in gas turbine engines as fan blades, compressor blades and vanes, disks, and cases, which is possible due to their high strength and low density. On the other hand, their susceptibility to interstitial formation during processing or application above 600 ºC, stabilizing the a phase, constitutes the major limitation for their use. This occurs by the spontaneous capturing of oxygen (as well as carbon and nitrogen). The a phase forms a brittle skin, known as the a case, which debits the structural properties of the alloys. Prior to any processing, the a case, therefore, must be chemically or mechanically removed.
Depending on the predominant phases, three families of titanium alloys may be considered: the 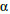 alloys, the
alloys, the 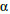 -
-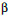 alloys, and the
alloys, and the 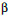 alloys. In the
alloys. In the 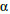 alloys, the
alloys, the 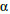 phase is stabilized by the addition of Al, which raises the
phase is stabilized by the addition of Al, which raises the 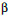 transus (the temperature boundary between the single-phase and two-phase regions, Fig. 6 (a) ). By their turn, the
transus (the temperature boundary between the single-phase and two-phase regions, Fig. 6 (a) ). By their turn, the 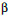 alloys contain elements such as V, Nb, Mo, Cr, W, Fe, Co, and Si, which stabilize the
alloys contain elements such as V, Nb, Mo, Cr, W, Fe, Co, and Si, which stabilize the 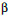 phase by lowering the
phase by lowering the 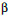 transus (Fig. 6 (b) ).
transus (Fig. 6 (b) ).
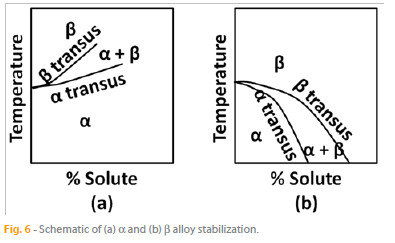
Both phases are present in the case of the 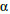 -
-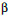 alloys. Some elements, such as Zr, Sn, and Hf, are neutral in the way that they do not stabilize either of the phases but contribute to property improvement.
alloys. Some elements, such as Zr, Sn, and Hf, are neutral in the way that they do not stabilize either of the phases but contribute to property improvement.
The increased strength in 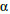 alloys originates from solid solution strengthening due to Al and Sn. Every 1 % Al addition increases strength by about 8 ksi (55 MPa), while every 1 % addition of Sn leads to an increase of 4 ksi (28 MPa). The
alloys originates from solid solution strengthening due to Al and Sn. Every 1 % Al addition increases strength by about 8 ksi (55 MPa), while every 1 % addition of Sn leads to an increase of 4 ksi (28 MPa). The 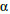 alloys are insensitive to heat treatment mainly due to the lack of second phases. On the other hand, the
alloys are insensitive to heat treatment mainly due to the lack of second phases. On the other hand, the 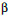 alloys always contain small amounts of the a phase, which size and distribution may be controlled by a combination of thermal and mechanical treatments. This second phase precipitate is responsible for the improved strength. Nevertheless, the most useful titanium alloys are the
alloys always contain small amounts of the a phase, which size and distribution may be controlled by a combination of thermal and mechanical treatments. This second phase precipitate is responsible for the improved strength. Nevertheless, the most useful titanium alloys are the 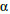 -
-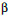 alloys, consisting of both
alloys, consisting of both 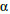 and
and  phases. In these alloys, heat treatments are beneficially used to control both solid solution strengthening and precipitate hardening.
phases. In these alloys, heat treatments are beneficially used to control both solid solution strengthening and precipitate hardening.
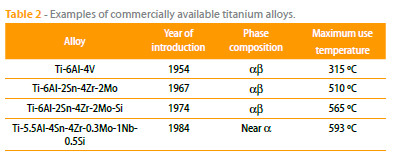
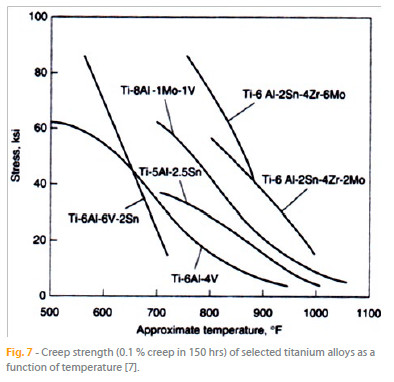
Table 2 presents some of the commercially available titanium alloys and Figure 7 depicts the creep strength of several Ti alloys as a function of the temperature. Considering the presented data, it is clear that the maximum use temperature of these titanium alloys is limited to 593 ºC.
When titanium alloys are exposed to an oxygen-containing atmosphere at elevated temperatures, they not only dissolve oxygen and form a case but also oxidize, producing a titania (TiO2) scale. Unlike alumina on many aluminum-containing alloys, titania is non-protective against continued oxidation, once that oxygen easily diffuses through it. To protect titanium alloys against oxidation at elevated temperatures, several coating systems have been investigated, such as platinum aluminides [8], magnetron sputtered Ti-Al [9], and Ti-Al-Cr [10].
3.2 Steels
Despite their complexity, steels are among the most versatile and widely used structural materials [11]. On a density-corrected basis, as shown in Fig. 4, the strengths of steels are competitive with nickel based superalloys at lower temperatures, with the advantage of being much more cost-effective. At higher temperatures, however, their strengths fall off below those of superalloys.
The structure of steels is based on the iron-carbon phase diagram depicted in Fig. 8. Due to the non-equilibrium cementite (Fe3C) as a terminal phase, this is not a true equilibrium phase diagram; nevertheless, the diagram faithfully explains the microstructure and phase behaviour for the range of practical thermal exposure. Depending on the carbon content, steels are classified into low-carbon (<0.25 wt % C), medium-carbon (0.25 to 0.60 wt % C), and high-carbon (> 0.60 wt % C) steels. In order to widen the range of structural and physical properties, other alloying constituents besides carbon are also added to steels. In this way, each of the classes may contain additional alloying elements such as Cr, Mo, Ni, V, Nb, Ti and Al to form the socalled alloy steels. At room temperature, pure iron has a bcc structure called a iron or ferrite (Fig. 8) which converts to an fcc (face - centered cubic) y phase called austenite above 911.5 ºC. With increasing temperature, before melting at 1538 ºC, austenite changes to a bcc phase called 8 ferrite at 1396 ºC. As shown in the phase diagram of Fig. 8, the sizes of the various phase fields vary with the carbon content.
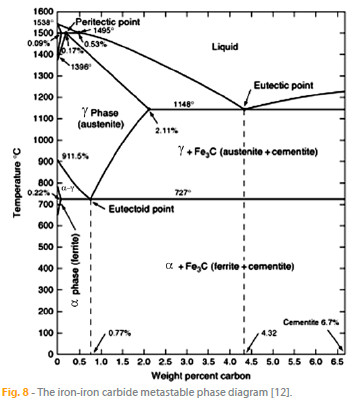
A combination of mechanisms is used to strength steels. Among them are solid solution strengthening by interstitial elements such as carbon and nitrogen, substitutional solid solution, either in the a of the y phase, precipitation hardening from carbides of Nb and V, grain size control, and processing-induced texture of grains by, for example, rolling. Moreover, different phases may be stabilized by different alloying elements. The degree of solid-solution strengthening by substitution is controlled by expanding (with the addition of C, N, Cu, and Zn), contracting (by the addition of Ta, Nb, and Zr), opening (with the addition of Ni, Mn, Co, Ru, Rh, Pd, Pt, etc.), or closing (as in the case of the addition of Si, Al, Be, and P) the y phase field of the phase diagram in Fig. 8.
The austenite phase can dissolve as much as 2.11 wt % (~10 at %) carbon in a solid solution. If this phase is quenched very fast to a temperature in the vicinity of room temperature, instead of changing to ferrite and cementite as predicted by the phase diagram in Fig. 8, it transforms into a phase called martensite supersaturated with carbon. The crystal structure is then distorted to body-centered tetragonal (bct) by the high carbon content. The unique feature of this phase transition is that it does not involve diffusion of atoms, but shear or local displacive transformation at rates close to the velocity of sound in the alloy. The maximum temperature below which martensitic transformation occurs is known as the martensite start temperature, Ms. The presence of martensite grains, which are hard and brittle and show plate or needle-like morphology, significantly increases strength.
Time-temperature-transformation (TTT) diagrams [11] have been developed in order to design the right heat treatment regimens to achieve the optimum combination of microstructure, strength, and ductility of steels. The following list includes some of the commercially available steels:
High Strength Low Alloy Steel (HSLA): These are low carbon (0.03 to 0.15 wt % C) steels which derive their strengthening from a solid solution (C, Mn, Si), precipitation hardening (carbides of Nb, Ti, and V), grain size control stabilized by grain boundary carbides and processing-induced texture. These materials typically show tensile strength and failure strains of 700 MPa (~100 ksi) and 12 to 18 %, respectively.
Bainite Steel: These steels consist in a strained ferrite matrix with a fine dispersion of cementite and are produced by an isothermal heat treatment of low carbon (<0.05 wt % C) steels according to a TTT diagram between 250 ºC and 550 ºC. Bainite steels achieve tensile strength of 600-1200 MPa (around 87-174 ksi) and failure strains of 15-20 %.
Dual-Phase Steels: These are low carbon steels (0.1 to 0.2 wt % C) which contain two phases consisting of a fine dispersion of 10 to 20 vol % martensite in a ferrite matrix. This microstructure and phase contents are obtained by heating the material to the a+y phase field (Fig. 8) to form 10 to 20 vol % austenite which is then converted to martensite by rapid quenching. This method leads to steels with moderate to high tensile strength and high ductility
Transformation Induced Plasticity Steels (TRIP): In this case, austenite (y phase) is retained at room temperature by fast quenching, which is then mechanically deformed during processing. Under load, the austenite phase transforms to martensite, leading to the strengthening of the steel, and to tensile strengths in the range of 600 to 1300 MPa (approximately 87 to 189 ksi), as well as failure strains as high as 25-40 %.
It should be said that the temperature capability of steels in stressed applications is controlled not only by strength but also by the environmental resistance and is generally limited to 650 ºC.
3.3 Nickel-iron alloys
Nickel-iron alloys usually contain 15 to 60 % iron and 25 to 60 % nickel [13]. This class of alloys has the fcc structure of y austenite as a matrix strengthened by precipitates, as well as solid solution and grain boundary strengthening. Ni-Fe alloys are less expensive than Ni and Co base superalloys, and may be classified into five groups, as follows:
Group 1: The first group is relatively Fe-rich with 25 to 35 % Ni and less than 2 % Ti. The strengthening phase consists in coherent fcc y (called gamma prime) precipitates. Commercial alloys in this group include Tinidur, V-57, and A-286, which use temperature is limited to 650 ºC.
Group 2: Alloys in group 2 are rich in Ni (> 40 %) with increased contribution from a solid solution, leading to higher strengths than those of the iron-rich group. Inco X-750 and Inco 901 are examples of commercial alloys of this group.
Group 3: In this group, Ni-rich alloys owe their strength to coherent bct y (called gamma double prime) precipitates. Inco 706 and 718 are commercial alloys representative of this group. The latter is one of the most used alloys of this class. The temperature capability of these alloys ranges from cryogenic to 650 ºC.
Group 4: These are Fe-rich Fe-Ni-Co alloys with low thermal expansion achieved through removal of Cr and Mo, which stabilize ferrite in the structure. Coherent fcc y precipitates are primarily responsible for the strength of this group of alloys. Commercial members of this class are Inco 903 and 909. The temperature capability of these alloys is limited to 650 ºC, and the elimination of Cr makes them more susceptible to oxidation and corrosion.
Group 5: This group of Ni-rich alloys has a particularity that is the absence of coherent precipitates. Some of the alloys of this group derive their strength from precipitates of carbides, nitrides, and carbonitrides. Other alloys, such as Hastelloy X and N-155, are solidsolution strengthened. For non-stressed applications, the latter may be used at temperatures limited to 1093 ºC.
3.4 Nickel and cobalt base superalloys
The name superalloys derives from the fact that this unique class of complex alloys based on Ni and Co exhibit extraordinarily high strength, which is also maintained across a wide elevated temperature range. Indeed, some of these alloys are used in load-bearing applications at temperatures exceeding 80 % of their melting point! Ni and Co are attractive metals because of their high melting point and crystal structures amenable to extensive alloying. Ni exhibits an fcc crystal structure, whereas Co is hexagonal close packed (hcp) at room temperature. Alloying elements are generally added in order to stabilize Co in the fcc form.
As depicted in Fig. 9, a number of strengthening mechanisms are used to produce practical alloys with Ni and Co [14-16], including:
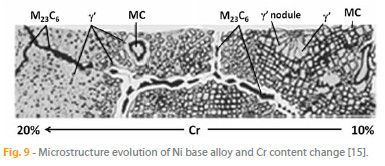
1. Strengthening of the austenitic y phase by solid solution: The matrix of the alloys consists in the y phase, which dissolves several metallic elements , such as Cr, Co, Fe, Mo, Ta, W or Re (in Ni base alloys) and Ni, Cr, Mo, W, Nb, Ta (in Co base alloys), randomly replacing the matrix atoms (Fig. 5 (a)). Due to size differences relatively to the matrix, the substituting solute atoms originate stress fields that impede dislocation motion, by interacting with the stress fields of dislocations, and therefore increase strength. This mechanism operates both in Ni and Co base superalloys.
2. Precipitates of various carbides within the grains and at the grain boundaries: Carbon, which is invariably present in the matrix, forms carbides with some of the alloying elements during processing and heat treatment. In Ni base alloys, MC type carbide is formed with Ti, Ta, W, Mo, Hf, Nb, whereas Cr, W and Mo lead to the formation of M23C6 type carbide; in Co base alloys, MC type carbide forms with Ti and M23C6 type carbide with Cr. Carbides within grains impede dislocation motion, thereby increasing strength. On the other hand, grain boundary carbides pin boundary movement, hindering grain growth and also impeding grain boundary sliding. The carbides together with the solid solution provide the primary mechanism for strengthening of Co base superalloys. In the case of Ni based superalloys, these mechanisms are secondary to y and y precipitation strengthening.
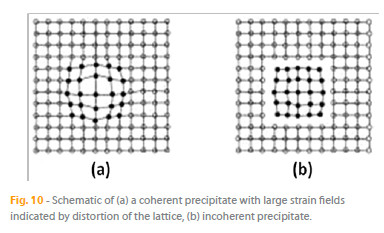
3. Coherent fcc y and bct y precipitates: The primary strengthening mechanism of Ni base superalloys is provided by precipitates of the ordered intermetallic compound of composition Ni3Al, known as gamma prime, y , with additional strengthening being achieved from solid solution and carbide precipitates. The Ni sites in the y crystal structure may also contain Co, Cr and Mo, while the Al sites may contain Ti and Nb. The unique characteristic of the y structure is that it is coherent with the y matrix. The matrix-precipitate interface for coherent and incoherent precipitates is schematically compared in Fig. 10. Because coherent interfaces have low energy, the coherent precipitates have little driving force for growth in size. This is the reason for the strength retention of y -strengthened superalloys over a wide high-temperature range. These fine and hard precipitates impede dislocation motion, resulting in a great increase in strength. Figure 11 shows cuboidal y precipitates in a y matrix [17].

The size and volume fraction of y precipitates are critical parameters regarding creep and fatigue strength of the alloys. The later is controlled by the Al+Ti content and ranges from 0.2 for wrought Fe- Ni superalloys to above 0.5 in Ni base superalloys. The size of the y precipitates ranges from 0.2 to 0.6 mm, with small precipitates having spherical morphology and larger sizes tending to be cuboidal.
In some superalloys containing Fe, such as Inco 718, the presence of y (a bct structured phase with composition Ni3Nb) is the predominant strengthening mechanism. The y phase is coherent with the y matrix and provides high strengths in the low to moderate temperature regime. However, above 65 ºC, strength significantly drops, as a result of the decomposition of y to either y or d.
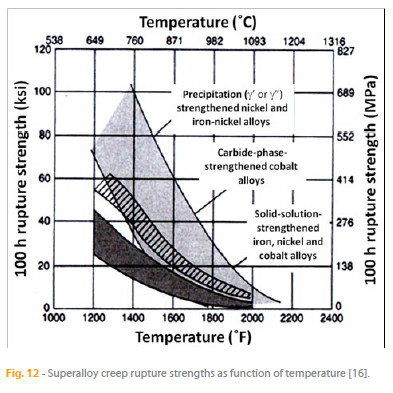
Fig. 12 presents the range of creep rupture strengths of superalloys strengthened by several of the mechanisms discussed above. Ni base superalloys in polycrystalline form are not acceptable for some creep-strength-critical applications, such as rotating turbine blades in modern gas turbine engines, because they tend to fail at grain boundaries oriented transversely to the centrifugal force of rotation, which is along the blade axis. These blades are fabricated by casting, which consists in melting of appropriate alloys in a furnace and pouring the melt into a ceramic shell. The creep properties of the alloys can be considerably improved by promoting columnar grains oriented parallel to the blade axis that eliminates undesired transverse grain boundaries. These microstructures are achieved through directional solidification, which consists in a modification of the casting process so that heat is removed from the melt by slowly pulling the ceramic shell from the furnace. Further increases in creep properties have been achieved by eliminating grain boundaries altogether in singlecrystal superalloys, which are obtained by using a grain selector or appropriate seeds in the casting process. Polycrystalline, columnar, and single-crystal structured turbine blades developed by Pratt & Whitney are compared in Figure 13.

Single-crystalline Ni-based superalloys with a high fraction (over 50 %) of y phase (Ni3Al) indeed exhibit a combination of excellent strength, creep properties and fatigue resistance at high temperatures, being currently used as blades in gas turbines and aircraft engines [18]. As mentioned, the microstructure of these alloys consists of sub-micrometer size cubical y precipitates in a matrix of y phase, an fcc super-saturated Ni solid solution. The size and volume fraction of the y phase dictate the width of the y channels, which can be in the nanometer range. Other microstructural features that may be present in Ni-based superalloy single-crystals include low angle boundaries, casting defects such as pores, and y/y eutectic pools due to incomplete solutionizing of the as-cast structure prior to the aging process. Hot isostatic pressing (HIP) was shown to be helpful in the closure of porosity and elimination of the eutectic pools, contributing for the increased strength of these single-crystalline Ni-based superalloys [19].
4. CONCLUSIONS
The development of structural alloys as discussed in this article has generally the main goal of achieving high strength, ductility, and producibility. However, obtaining materials that combine these properties with environmental resistance toward oxidation and high-temperature corrosion is not an easy task. As an example, increased Al and Cr contents improve oxidation and corrosion resistance of the resulting alloys, but beyond a certain level, these elements reduce creep strength. The separation of the two functions is one approach that is generally used in order to combine both strength and resistance to the environmental degradation. The method consists in protecting a substrate alloy, which provides the load capability, by the application of thin coatings with adequate Al and Cr, achieving the desired oxidation and corrosion resistance. The thickness of the coating is adjusted in a way that it does not carry any significant load.
The required coating for each of the above discussed alloys is dependent of the temperature of use and its compatibility with the properties of the substrate material, namely its composition and structure (modulus) and coefficient of thermal expansion, must be taken into consideration. This is a subject to be discussed in a following article.
REFERENCES
[1] M. F. Stroosnjder, R. Mevrel and M. J. Bennet, Mater. High Temp., 12, 53 (1994). [ Links ]
[2] S. Bose (High Temperature Coatings), Elsevier Butterworth- Heinemann, Amsterdam (2007). [ Links ]
[3] y . W. Meetham, Materials & Design, 9, 244 (1988). [ Links ]
[4] C. A. C. Sequeira (High Temperature Oxidation), in Uhligs Corrosion Handbook, 3rd Ed., (R. Winston Revie, ed.), John Wiley & Sons, New York, p. 247 (2011). [ Links ]
[5] C. A. C. Sequeira (High Temperature Corrosion in Molten Salts), Trans Tech Publications, Ltd., (2003). [ Links ]
[6] C. J. McMahon and L. F. Coffin Jr., Metall. Mater. Trans B, 1, 3443 (1970). [ Links ]
[7] M. J. Donachie Jr., (Titanium: A Technical Guide), 2nd Ed., ASM International, New York (2000). [ Links ]
[8] I. Gurrappa and A. K. Gogia, Mater. Sci. Tech., 17 (5), 581 (2001). [ Links ]
[9] C. Leyens, M. Peters, D. Weinem and W. A. Kaysser, Metall. Mater. Trans. A, 27, 1709 (1996). [ Links ]
[10] C. Leyens, M. Schmidt, M. Peters and W. A. Kaysser, Mat. Sci. Eng. A, 239-240, 680 (1997). [ Links ]
[11] R. W. K. Honeycombe (Steels: Microstructure and Properties), Edward Arnold and ASM International, New York (1982). [ Links ]
[12] J. Chipman, Metall. Mater. Trans. B, 3 (1), 55 (1972). [ Links ]
[13] E. E. Brown and D. R. Muzyka (Superalloys II), (C. T. Sims, N. S. Stollof and W. C. Hagel, ed.), John Wiley & Sons, New York, p. 165 (1987). [ Links ]
[14] C. R. Brooks (Heat Treatment, Structure and Properties of Nonferrous Alloys), ASM International, New York (1982). [ Links ]
[15] C. T. Sims (Superalloys II), (C. T. Sims, N. S. Stollof and W. C. Hagel, ed.), John Wiley & Sons, New York, p. 3 (1987). [ Links ]
[16] J. M. Donachie Jr. and S. J. Donachie (Superalloys: A Technical Guide), 2nd ed., ASM International, New York (2002). [ Links ]
[17] M. Gell, D. N. Dune, D. K. Gupta and K. D. Scheffler, JOM, 39 (7), 11 (1987). [ Links ]
[18] T. M. Pollock and S. Tin, J. Propul. Power, 22, 361 (2006). [ Links ]
[19] F. Ebrahimi and E. F. Westbrooke, Acta Mater., 56, 4349 (2008). [ Links ]
Artigo submetido em Abril de 2013 e aceite em Agosto de 2013













